SR Gupta et al. JPGN 2019; 69: 544-50. This article reports on preliminary experience in 54 children who received external (non-hospital) infliximab infusions. The average age was 17.6 years. The authors noted no serious safety concerns. Prior to arranging these infusions, the authors insisted on the following:
- Infusion services had to guarantee pediatric trained nurses with PALS certification
- Emergency medications had to be available
- A plan for emergency communication was arranged
- Postinfusion communication would occur with each infusion
BN Limketkai et al. Inflamm Bowel Dis 2019; 25: 1828-37. This study, using Truven Health MarketScan database (2007-16) reviewed proactive or reactive mucosal monitoring after biologic initiation in IBD. Early (< 6 months) proactive monitoring (88% endoscopy-based) was performed in 11% (n=2195/19,899) of patients with Crohn’s and 12.8% (925/7247) of patients with ulcerative colitis.
- “Early proactive monitoring was associated with a reduction in disease-related complications for CD (aHR 0.90) and UC (aHR 0.87) and predominantly driven by a reduction in corticosteroid use.”
- Another interesting finding was that ~40% of patients had biologic therapy initiated without assessment of mucosal disease activity within 6 months.
- The authors state that disease monitoring is typically more useful in CD than UC because with the latter, cessation of bleeding and diarrhea appear to be adequate surrogates.
- This study was not able to assess whether a biomarker like fecal calprotectin would be suitable due to its low utilization.
RZ Cohen, BT Schoen, S Kugathasan, CG Sauer. JPGN 2019; 69: 551-6. In this chart review, the authors identified anti-drug antibodies (ADA) in 24.8% (n=58) of patients undergoing therapeutic drug monitoring (n=234) with both infliximab and adalimumab. 54% of this group had antibody suppression with dose optimization. Of note, 37 patients had detectable ADA at time of initial drug monitoring. Dose optimization was 10 mg/kg every 4 weeks with infliximab or 40 mg weekly with adalimumab. Patients who were switched to a second anti-TNF agent (n=23) were not more likely to develop ADA to the second agent (small sample size). Also, the authors caution that in the five patients with ADA levels (>10 U/mL), dose optimization failed and patients required a therapeutic switch. My take: This study provides some useful information about the frequency of ADA. My view is that the actual drug level is more critical than the presence of ADA; though, the presence of high ADA often precludes the ability to deliver a therapeutic drug level.
Related blog posts:

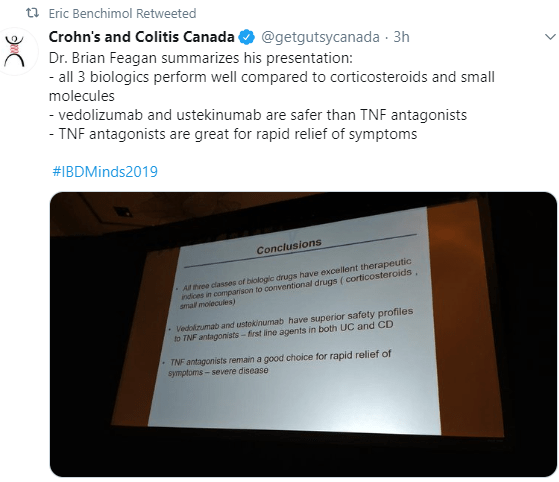
Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.













