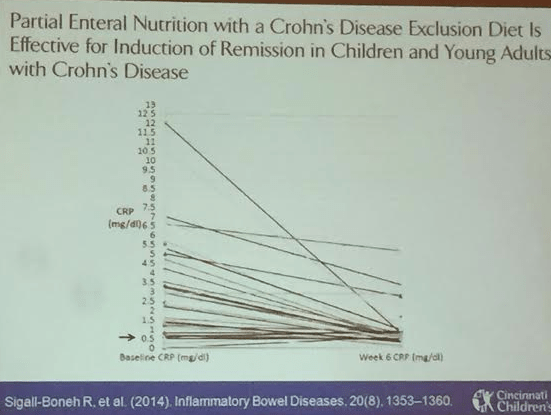
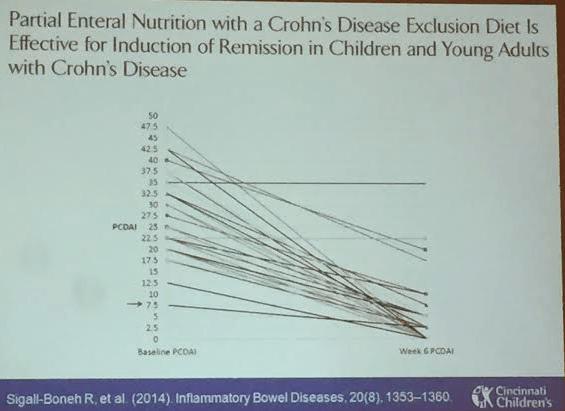
This blog entry has abbreviated/summarized this terrific presentation; most of the material has been covered in this blog in prior entries (can use search function to find additional relevant material) but still this was a useful review. Though not intentional, some important material is likely to have been omitted; in addition, transcription errors are possible as well.
Emerging Therapies in IBD –Dr. Gary Lichtenstein
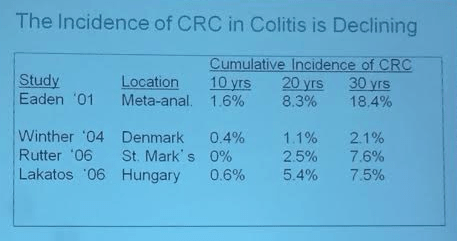
Background: This lecture started with a review of current therapies. We have learned how to use our current therapies better. There still remain a large number of patients that face surgery with IBD; though there has been improvement (?50% reduction).
Issues with thiopurines were reviewed. May take 2-6 months to take effect, though monotherapy with thiopurines are fairly ineffective for Crohn’s disease as initial therapy.
Leukocyte Trafficking Agents:
- Natalizumab
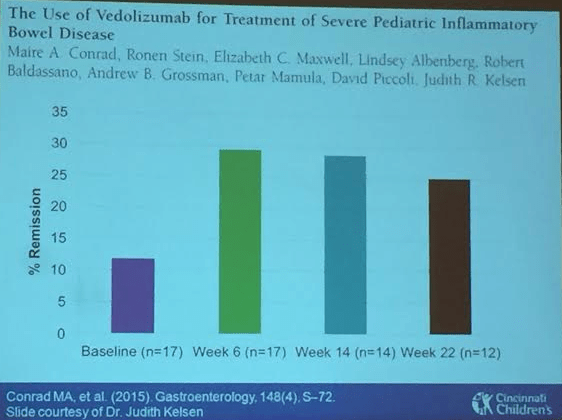
- Vedolizumab
- AJM300 –oral agent. Initial safety data were fine.
- AMG 181
- Etrolizumab (Vermeire S Lancet 2014) –low rates of endoscopic healing, but better than placebo
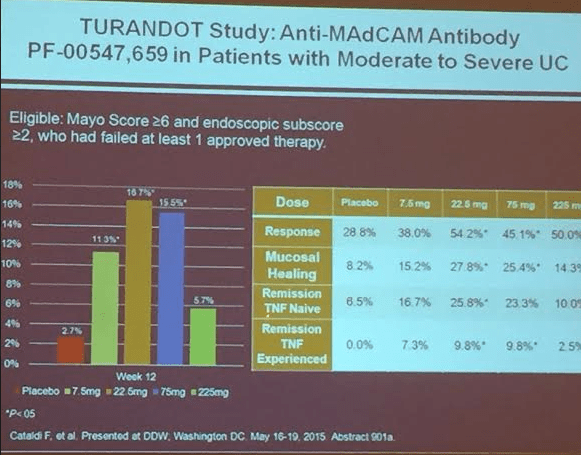
PF-00547,659
S1P Modulators:
Fingolimod
Ozanimod (RPC1063) (oral agent, fairly rapid onset) causes S1P-r on lymphocytes to be internalized –more selective than Fingolimod. Good safety has been noted thus far. No notable cardiac problems. Infrequent elevations of transaminases; this issue will need to be followed.
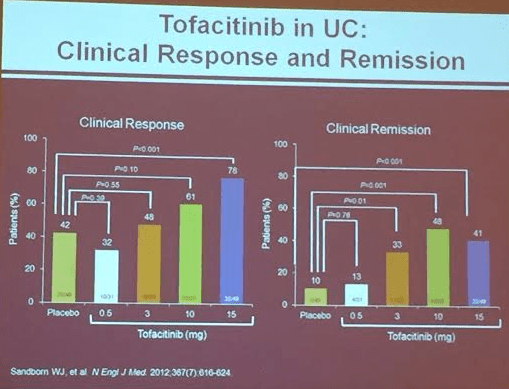
Tofacitinib oral Janus Kinus (JAK) Inhibitor (Sanborn WJ et al. NEJM 2012; 367: 616-24). Dr. Lichtenstein thinks 10 mg will be recommended dose. Follow lipids. For UC
Mongerson related post: Mongerson -Phase II Data Available in NEJM | gutsandgrowth
Ustekinumab
Related article from GI & Hep News: Ustekinumab for complex Crohn’s from ECCO conference/UNITI-1 Study (n=741)
FMT. Further studies are needed