Recently Dr. Garza gave our group an excellent lecture. It is always great learning more about how the GI tract really works and what is going wrong when patients are having symptoms. I have taken some notes and shared some slides. There may be inadvertent omissions and mistakes in my notes.

Physiology:
- Air in the GI tract needs to either be expelled from mouth, be absorbed or be expelled rectally. Nitrogen gas is not well-absorbed. Bacteria in GI tract can contribute to gas production and can also absorb gas.
- Only about 23% of GI tract air is expelled rectally as gas. Most gas is absorbed and can be expelled from the lungs subsequently.
- GI tract makes adjustments after swallowing air to help with comfort. This includes raising of the diaphragm. Toddlers frequently have aerophagia but infrequently have symptoms due to distention. Symptoms may worsen in teens/older individuals due to not allowing gas passage

Bloating:
- Bloating is a sensation that can occur with and without distention.
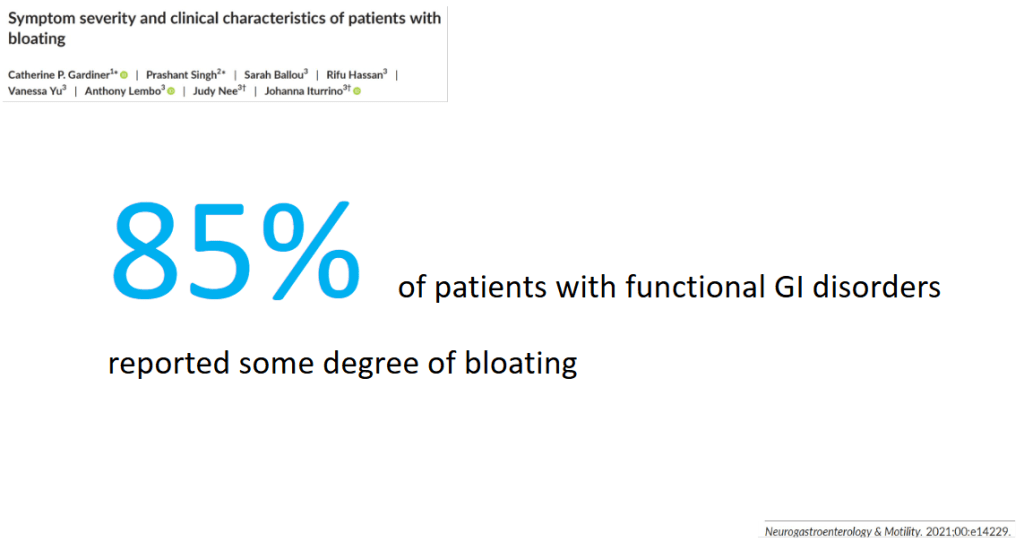
- Up to 85% of patients with DGBIs c/o bloating
- Reasonable to check for celiac disease and possibly other tests if alarm symptoms like bilious vomiting, weight loss and poor growth
- Increased air in the small bowel is rare and often indicative of dysmotility
- Elegant studies with CT scan have shown that the typical increase in excess gas during bloating symptoms is only 22 mL. With pseudoobstruction, excess volume of gas is around 3000 mL.
- pH-impedance is good at detecting aerophagia which often contributes to bloating. Aerophagia prevalence was 3.66% in one study
- The amount of air from bacterial overgrowth (SIBO) is usually NOT enough to cause most of the reported symptoms of bloating (though may be a contributing factor).
- A lot of bloating symptoms are due to increased sensitivity and ‘weird gas handling.’ The latter could include compression of diaphragm rather than elevation.
- Diets (eg lower fructan) can decrease gas but likely also work in other ways. Diets also have side effects and this needs to be carefully considered due to potential issues with eating disorders/ARFID
- Treat constipation IF PRESENT
- Diaphragmatic breathing, CBT, neuromodulators and peppermint oil are potential treatment options
- Increased activity helps with bloating and gas passage






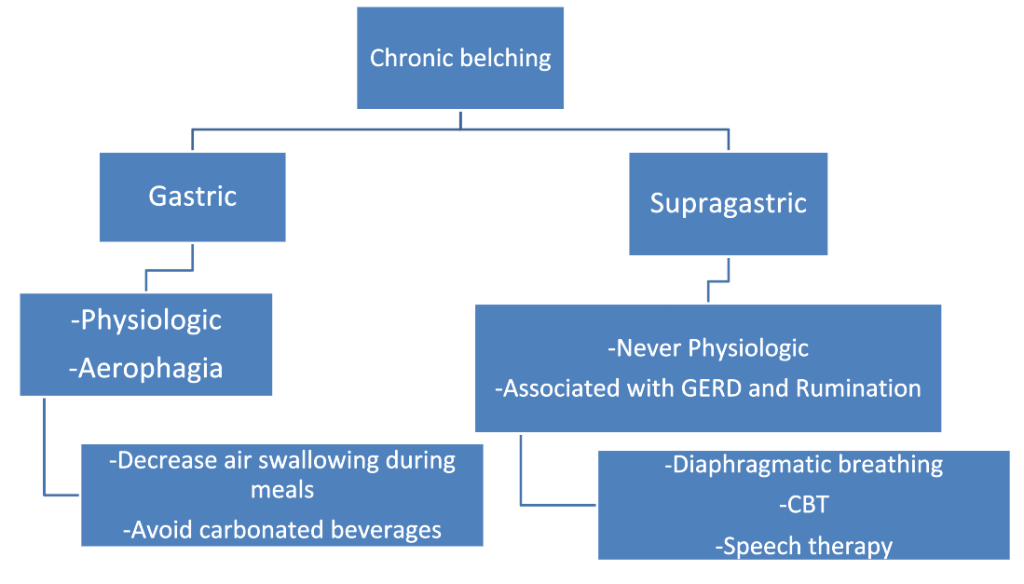
Belching:
- Descriptions of belching date back more than a hundred years
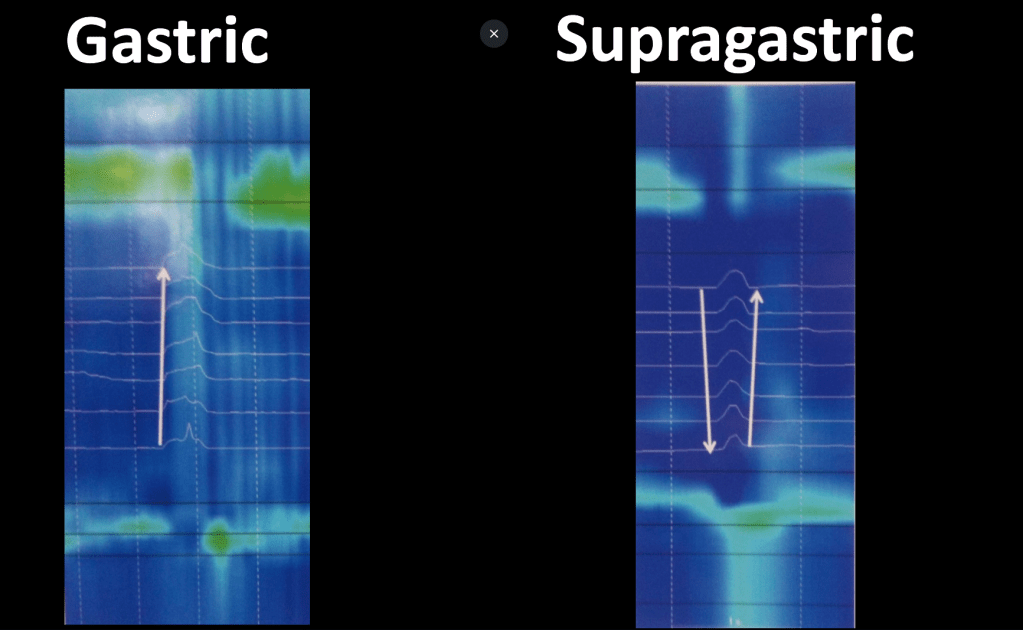
- Most belching is normal. Most belching is due to gastric belching and is physiologic
- Supragastric belching is abnormal. Hallmarks are frequent symptoms and can be associated with worsening reflux and rumination
- Differences between gastric and supragastric belching can usually be distinguished with clinical presentation (see below). Manometry findings are distinctive between the two. With supragastric belching, With supragastric belching, the air that is expelled is from the esophagus. With gastric belching, air that has reached the stomach is expelled.
- Main treatments for supragastric belching are diaphragmatic breathing, and CBT



Inability to Belch:
- In patients unable to burp, many have retrograde cricopharyngeus dysfunction. This is due to dysfunction of upper esophageal sphincter which had increased pressure and not allowing air in the esophagus to escape. This, in turn, causes discomfort and gurgling noises. While this disorder was reported in 1987, more widespread recognition has occurred since 2019
- Manometry should be done prior to botox therapy which results in improvement in most patients


Related blog posts:
- Bloating, Belching –Bowel Sounds Podcast with Dr. Jose Garza
- NASPGHAN Dysphagia Webinar: Dr. Khalil El-Chammas, Dr. Peter Osgood, and Dr. Jose Garza
- Jose Garza: What’s New in Motility (Part 1)
- Jose Garza: What’s New in Motility (Part 2)
- ANMS Virtual Symposia on Constipation
- Refractory Constipation -Terrific Update
Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.