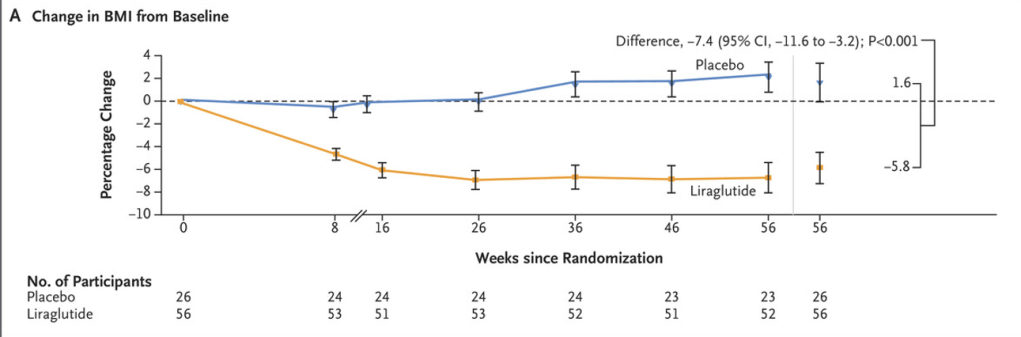
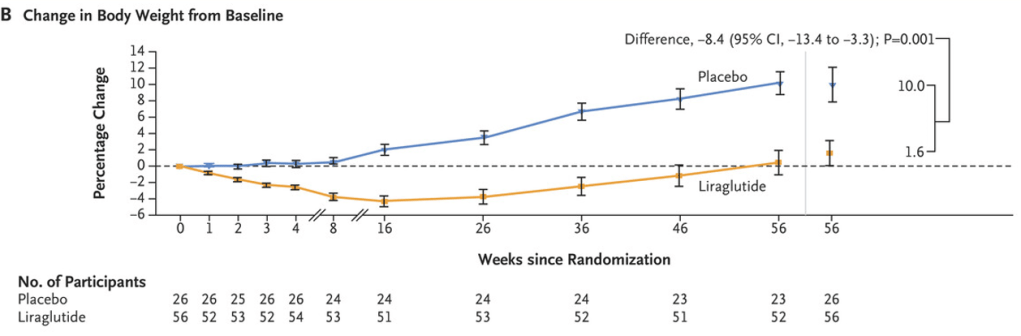
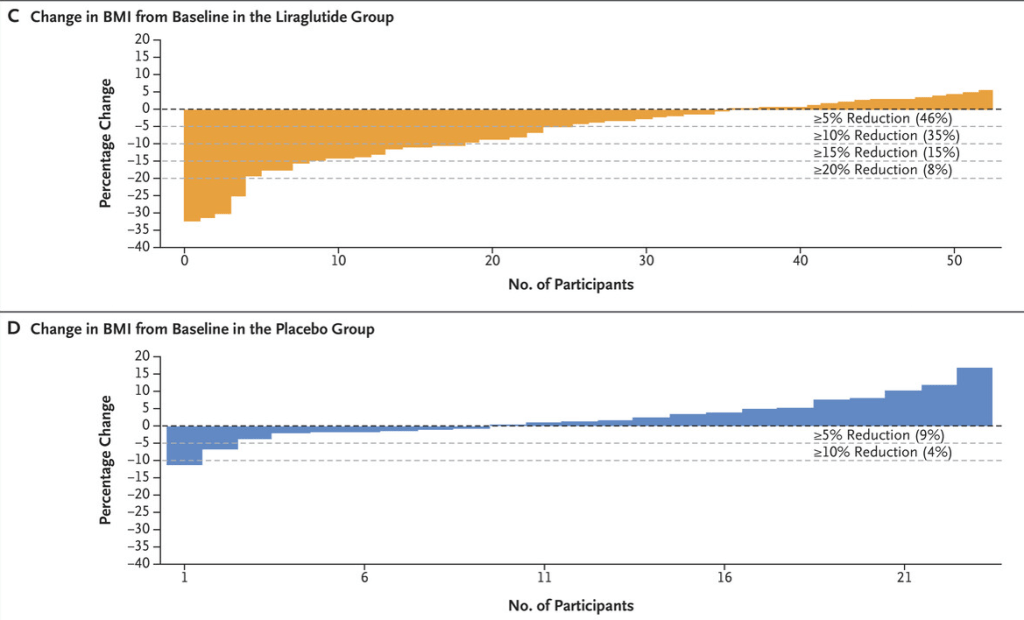
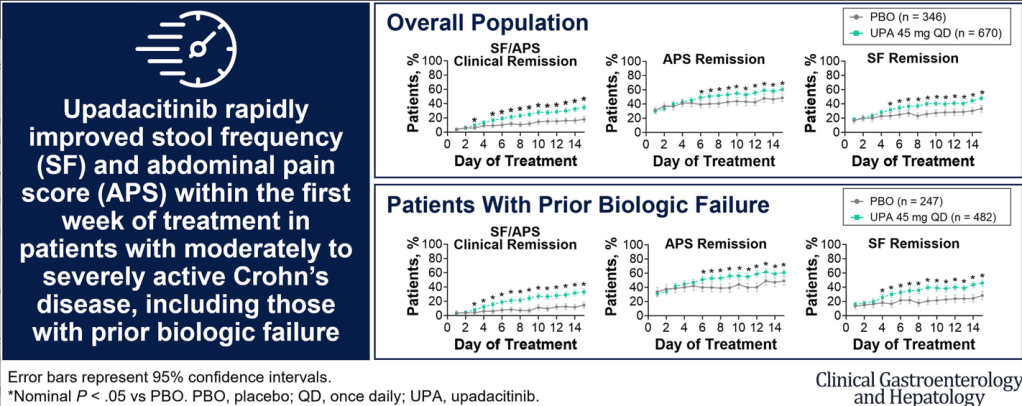
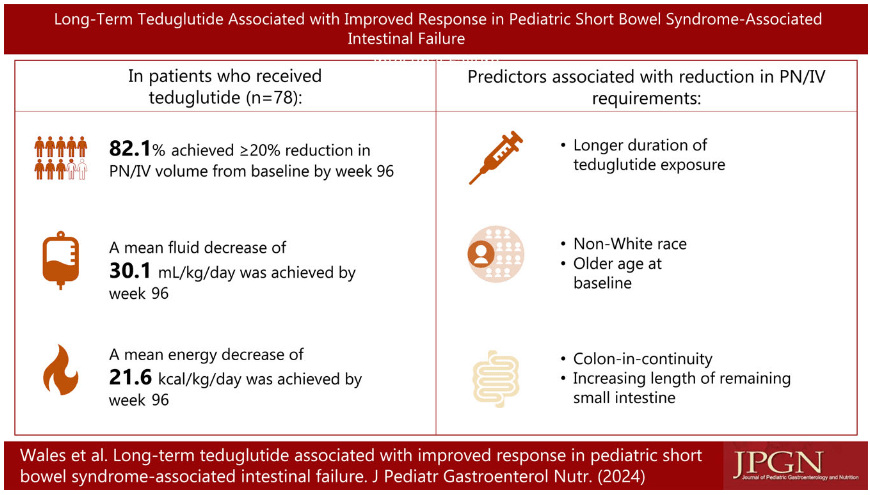
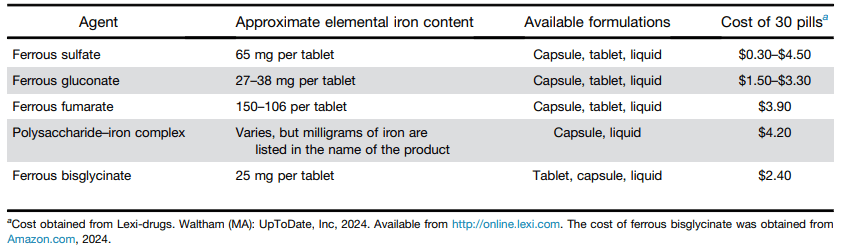
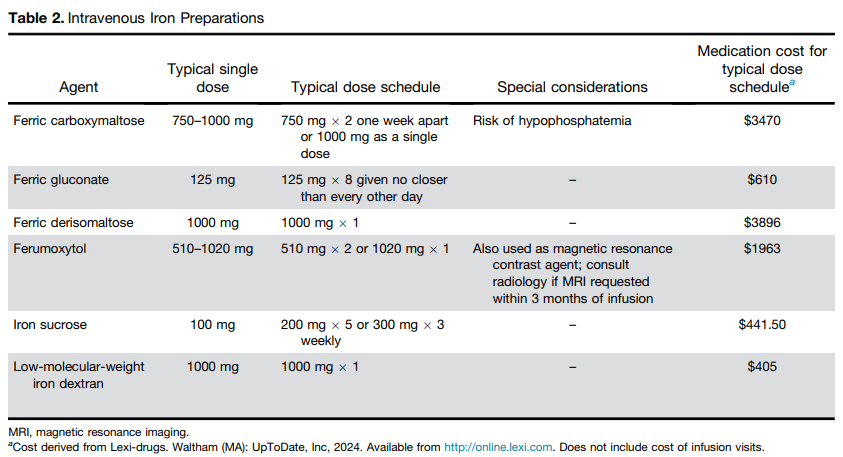
S.A Abrams, R.J. Shulman. The American Journal of Clinical Nutrition, https://doi.org/10.1016/j.ajcnut.2024.08.028. Open Access! What would happen in the United States if there were no cow milk-based preterm infant nutritional products: Historical perspective and evaluation of nutrient-related challenges
Background: “Recent legal action (1) has raised the possibility that preterm cow milk-based infant nutritional products (PCMBPs) may either not be produced and available in the United States or may require parental consent and warnings making them extremely difficult for caregivers to use and families to accept (2).”
Key points:
- “Evidence that unfortified human milk is inadequate to meet the nutrient needs of small preterm infants has been present for over 100 years (6). Until the late 1970’s and early 1980’s, there were limited widely available solutions to either fortify or replace human milk for preterm infants and these were often based on providing feedings of concentrated evaporated cow milk; now recognized as woefully inadequate and unsafe (7 ). At that time, specialized formulas that were high in energy, protein, and micronutrients were widely introduced into the market. Early studies confirmed faster growth of infants fed such products compared to unfortified human milk or full-term formula (8) and their use became widespread by by the mid-1980s.”
- “The availability of PCMBPs as well as a human milk-based human milk fortifier has led to relatively less frequent and severe nutritional failure in very low birth weight, less than 1500 g (VLBW) infants than in earlier eras. Nonetheless growth failure remains a current problem, especially in the smallest of infants, that is, those less than 1000 g birth weight and less than 25 weeks post-menstrual age at birth (12).”
- “Inadequate growth and mineralization can be expected to lead to long-term developmental disability, bone demineralization, and frank rickets”
- “Even with such improvements in policy to support breastfeeding and human milk donations, it is unlikely that an all human milk-based diet could be available for many years if ever for most of the preterm infants born in the US each year, either in the NICU or after hospital discharge.”
- “Supplements for infants who are over a year of age.. are not suitable for preterm infants, especially those in the first 6 to 9 months after birth. They have relatively high osmolality and nutrient levels, would pose substantial risks of intolerance, and would not meet the specific nutrient requirements of this population.”
- “The process of individualizing or creating multi-component fortification strategies would be daunting and carries risks of contamination, preparation errors, and tolerance issues. Without proper fortification, preterm infants are at a high risk of growth failure and its associated negative impacts on development and even survival.”
My take: Experts in the field of neonatal nutrition are worried that recent litigation will negate decades of nutritional advances in neonatal care. This is a very real threat. In addition to more complications, the litigation will result in much greater expenses for all infants/families.
Related blog post: Proliferation of Formula Lawsuits In Necrotizing Enterocolitis