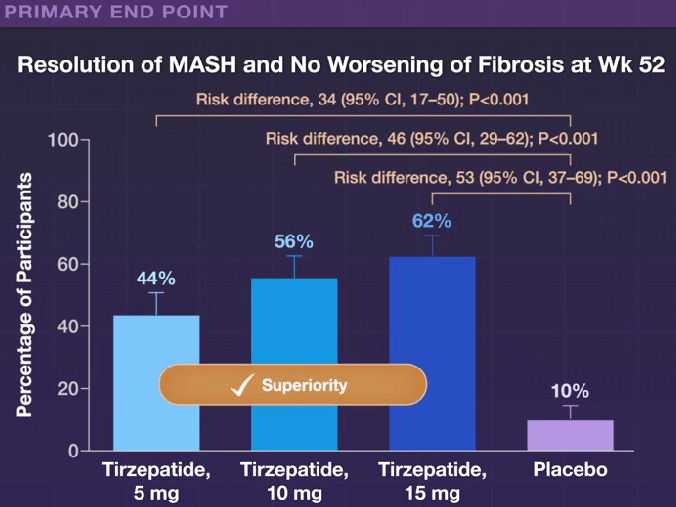
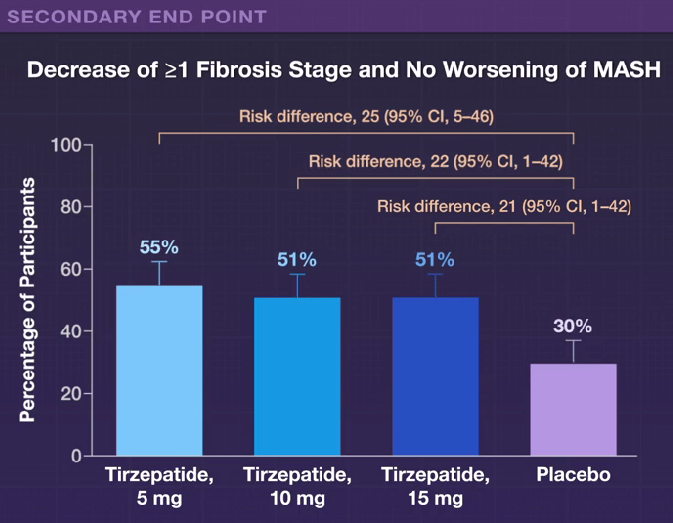
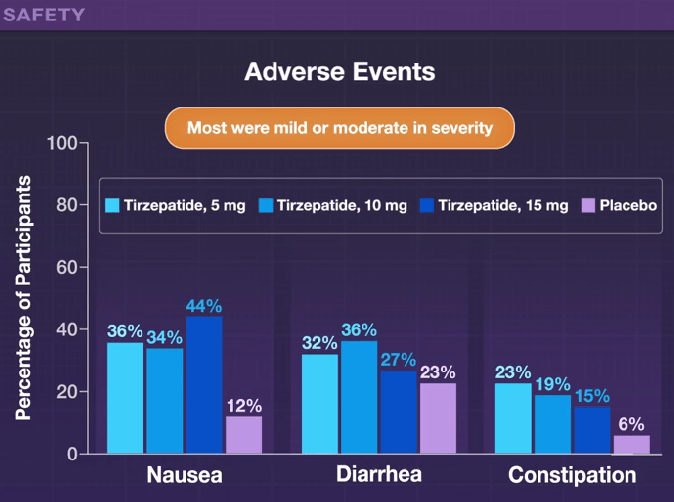
H Sato et al. Clin Gastroenterol Hepatol 2024; 22: 1531-1534. (Research Letter) Eosinophil Involvement Outside the Esophagus in Eosinophilic Esophagitis
The authors retrospectively studied a cohort of children (n=782) with eosinophilic gastrointestinal disorders (EGID) including 592 with isolated eosinophilic esophagitis (EoE), 190 with EGID which included esophageal involvement (“EI”) and 35 EGD without esophageal involvement (“Non-EI”).
Key findings:
- Abdominal pain was more frequent in EI than isolated EoE: 61% vs 45%, p=.002)
- EI patients had more dense esophageal eosinophil infiltrate (peak) than isolated EoE: 115 vs 92; P=.036) and higher peripheral blood eosinophil level (0.44 vs 0.38; p=0.027)
- 94-Gene expression profiles from esophageal biopsies from a 168 subgroup showed that EoE and EI were not significantly different. The heat map for upregulated and downregulated gene expression was different based on disease activity but not significantly different between EoE or EI in either state.
My take (borrowed from authors): The similar molecular transcriptome between EI and EoE are indicative of a shared pathogenesis.
Related blog posts:
- Updated Nomenclature for Eosinophilic Gastrointestinal Diseases
- Eosinophilic Colitis Is Not a Typical EGID or IBD
- Eosinophilic Gastritis -Pathogenesis Is Not Just the Eosinophils or Benralizumab Would Work
- How many eosinophils indicate eosinophilic gastroenteritis or colitis?
- Endoscopic Assessment of Eosinophilic Gastritis
- Two Studies for Eosinophiles
- New Treatment for Eosinophilic Gastritis and Duodenitis