Thanks to all of you who provided articles, suggestions and encouragement to make this blog better this past year. This has been the 12th year for this blog and it has continued to gain more views each year. Here are links to many of my favorite posts from 2023 along with a few pictures:
GI:
- Which Diet is Best for Irritable Bowel Syndrome? A Randomized Trial
- Helicobacter Pylori Stool Susceptibility in Children –How Good Is It?
- Atlantis Study: Possibly Best Evidence That Tricyclics May Help Irritable Bowel
- Current Approach for FPIES
- Long Term Benefits of Helicobacter Eradication in U.S.
- Short Bowel Syndrome is a Full Time Job
- Stratifying the Risk of Asymptomatic Gallstones
- Radiographs and Constipation -Bad Practice and Good Study
- If a Gastroparesis Medication Works in the Forest But No One Sees It, Did It Really Work?
- Incredible Review of GERD, BRUE, Aspiration, and Gastroparesis
Eosinophilic Esophagitis:
- Practical Guide to Dietary Therapy for Eosinophilic Esophagitis
- Landmark Dupilumab Study for Eosinophilic Esophagitis
Endoscopy:
IBD:
- Why Do Children Taking Adalimumab Benefit from Methotrexate Dual Therapy?
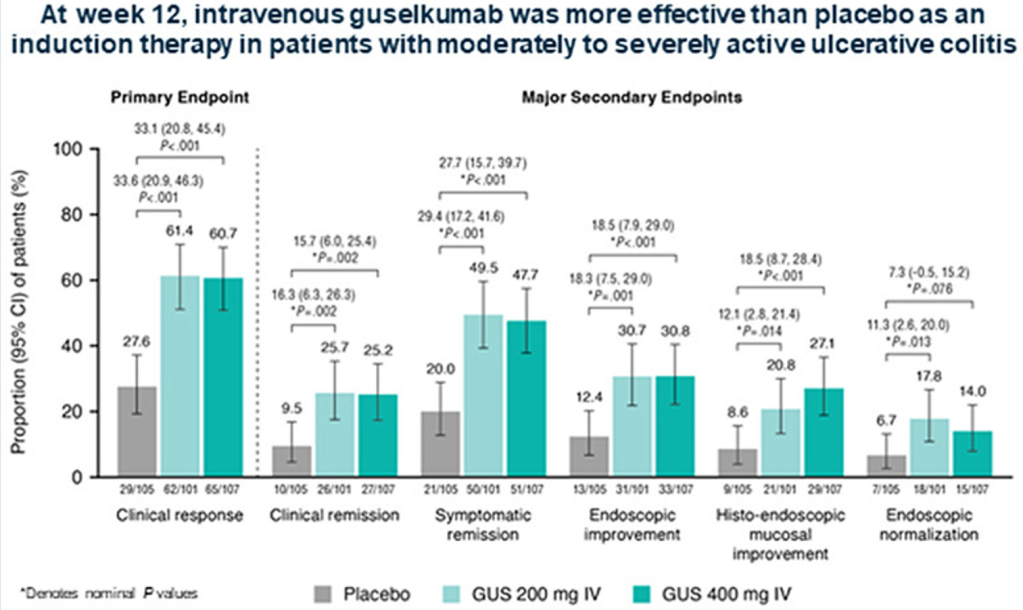
- Is Risankizumab More Effective for Crohn’s Disease Than Ustekinumab?
- Upadacitinib Works Quickly and with High Response
- CCFA 2023 (Atlanta) Part 4
- CCFA 2023 (Atlanta) -Part 1
- Do We Need to Reimmunize Patients (with IBD or Celiac) with Low Hepatitis B Surface Antibody Levels?
- Risk Factors for Inflammatory Bowel Disease: Ultra-Processed Food (Part 1)
- Landmark Study: Oral Biologic for Crohn’s –Upadacitinib
- Another Study Justifying Higher Infliximab Dosing in Pediatrics
- Immune Dysregulation and Inflammatory Bowel Disease
Liver:
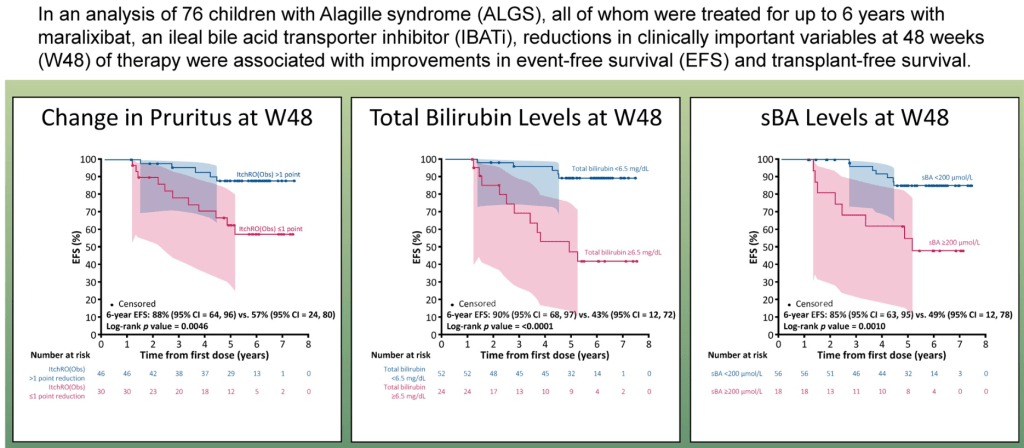
- GALA: Alagille Study
- Why CDC is Drafting New Guidelines for Screening Children for Perinatally-Acquired Hepatitis C Infection
- Customized Postoperative Therapy for Biliary Atresia -Does It Help?
Personal:

Humor:

General Health/Health Economics:
- Coding Correctly in 2023
- Poster Child for Gaming Pharmaceutical Regulations: Humira
- Maternal Mortality in Georgia
- Life Expectancy Dropping in U.S.
- ChatGPT Passes the Bar, an MBA exam, and Earns Medical License?