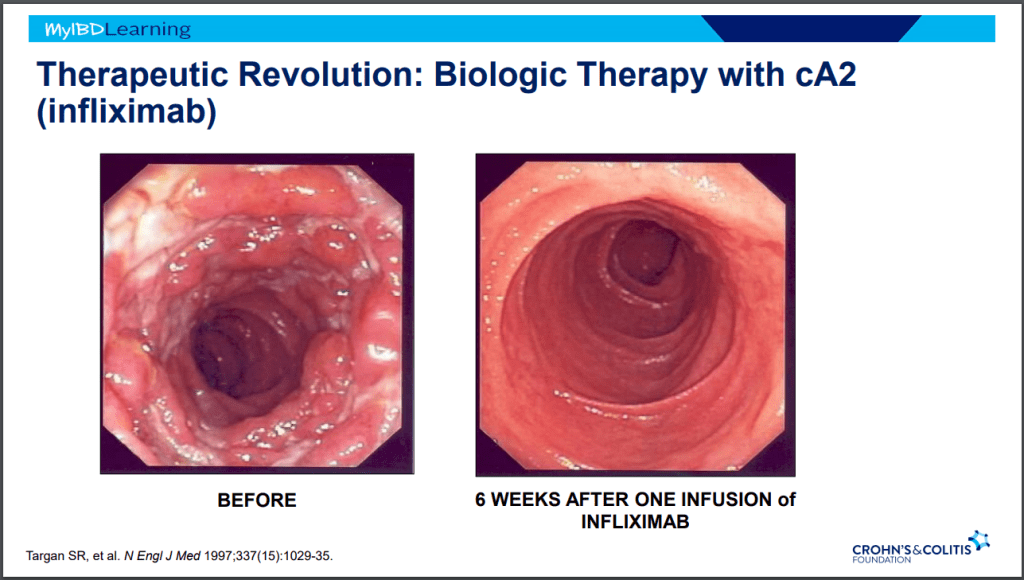
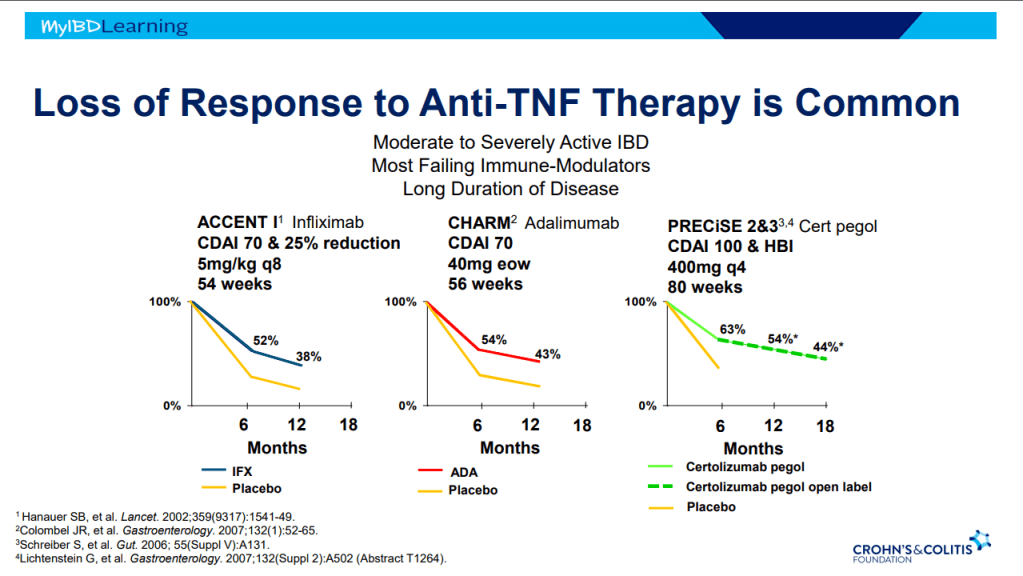
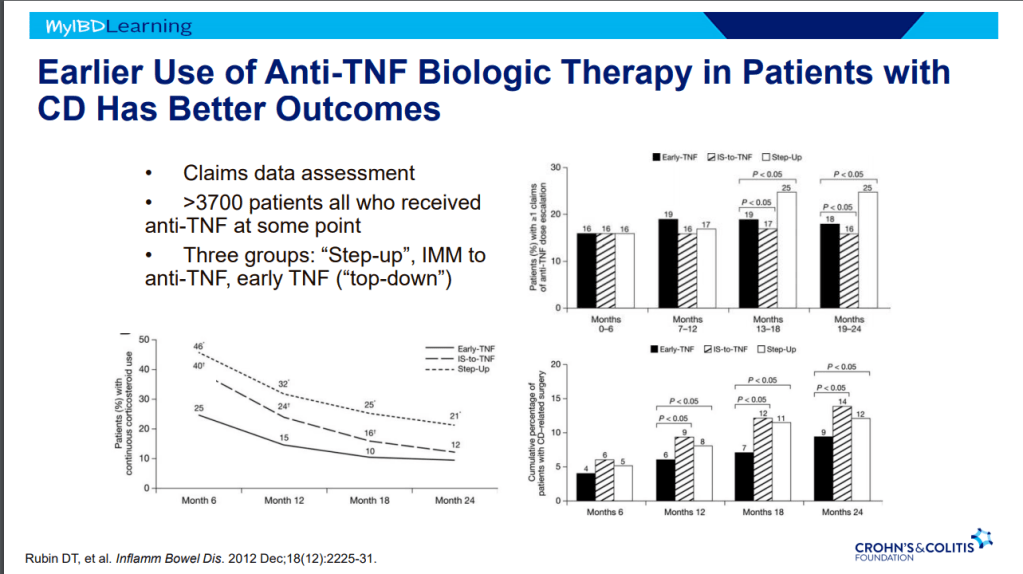
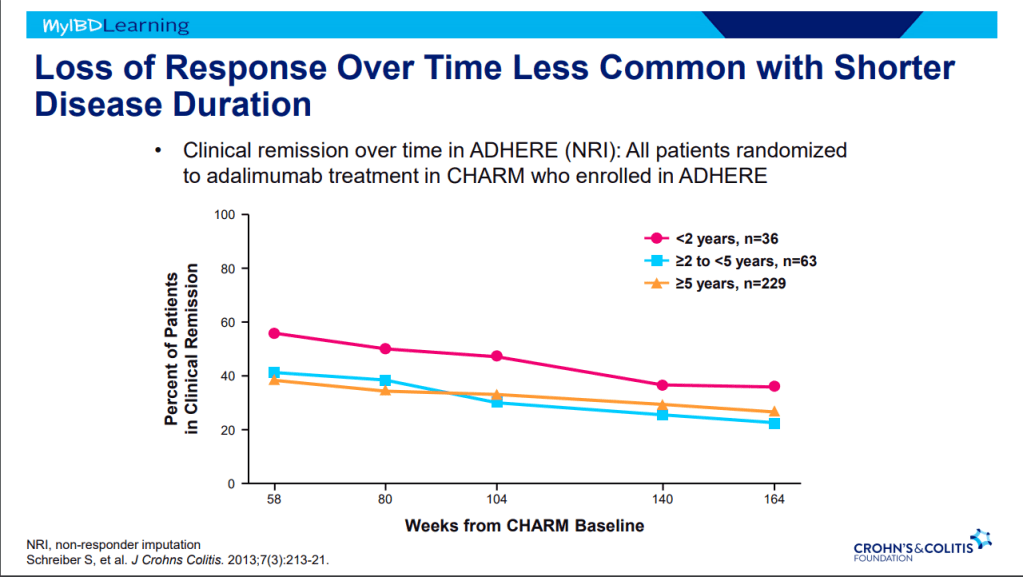
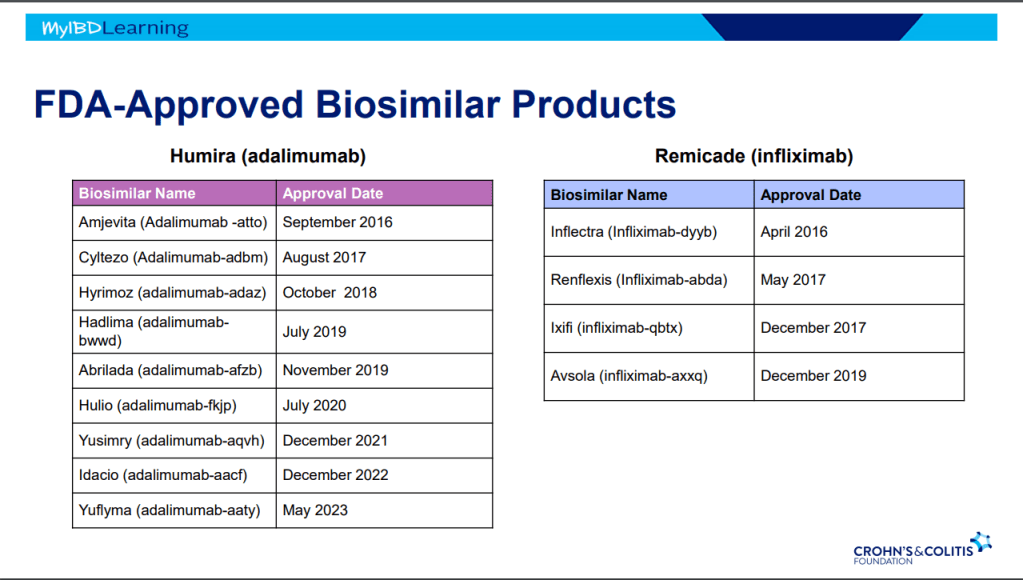
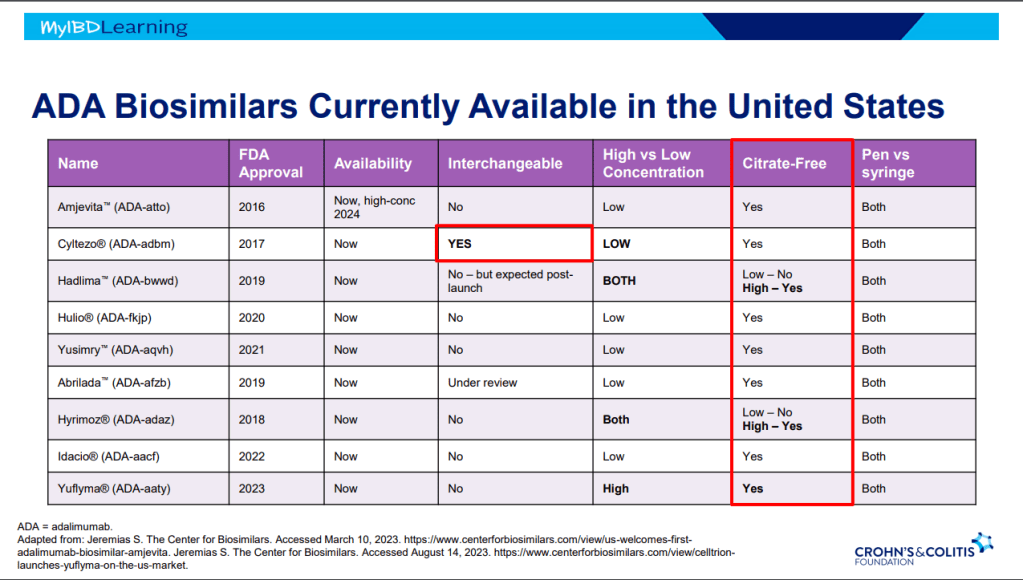
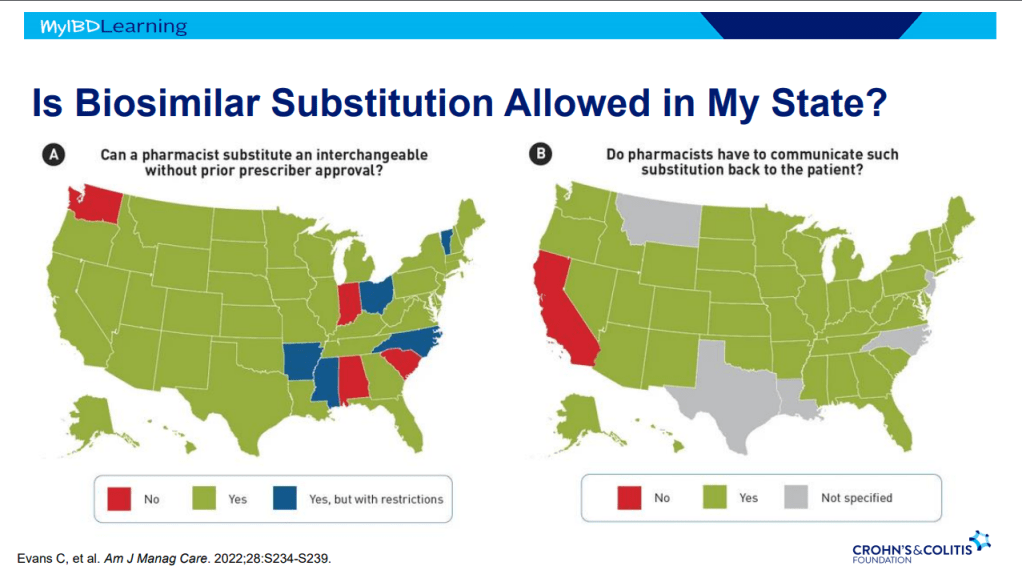
Another terrific lecture from David Rubin: Positioning of Advanced IBD Therapy. Here are my notes and some slides which could include errors in transcription and omission. Can get access to all 46 slides here: IBD Pro CCFA 2023 Atlanta
Many challenges in sequencing treatment -heterogeneity of diseases, loss of response, challenges in interpreting data, understudied issues (perianal disease, extra-intestinal manifestations, psychological health).
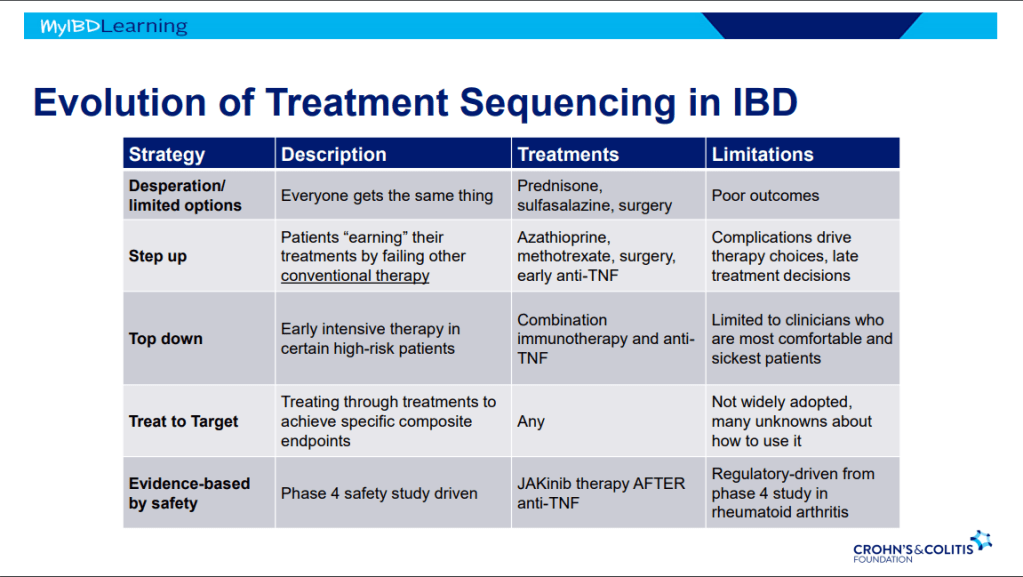
Evolution of therapies: desperation era -limited options, step up era, top down era with anti-TNFs, treat to target era and currently evidence-based by safety era.
- It is possible to avoid steroids in many patients with upcoming plans to use an advanced therapy -this is probably beneficial. This may result in patients remaining symptomatic until these therapies can be started.
- JAK inhibitors cannot be given as first-line agent in U.S. (but is done in other countries).
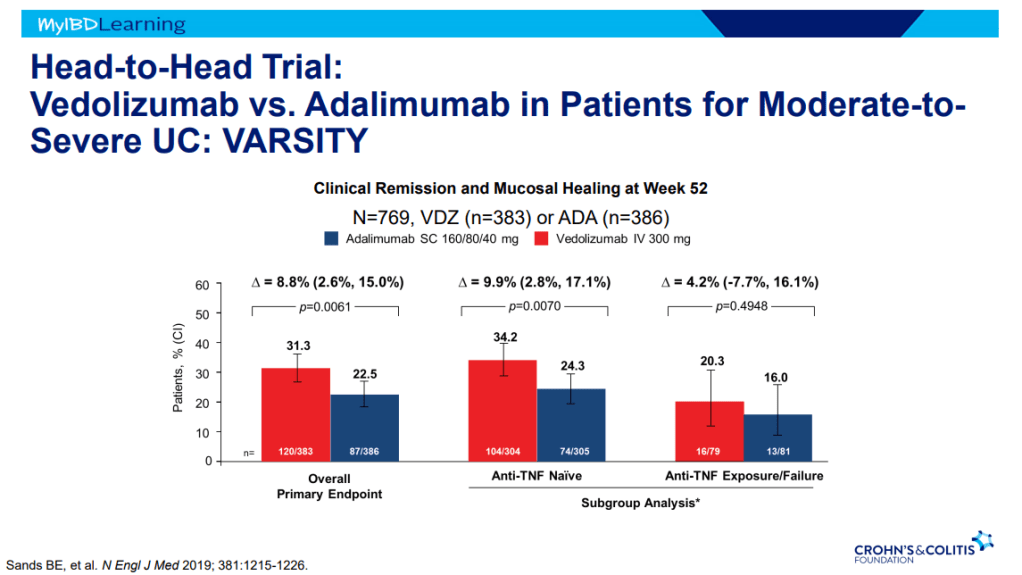
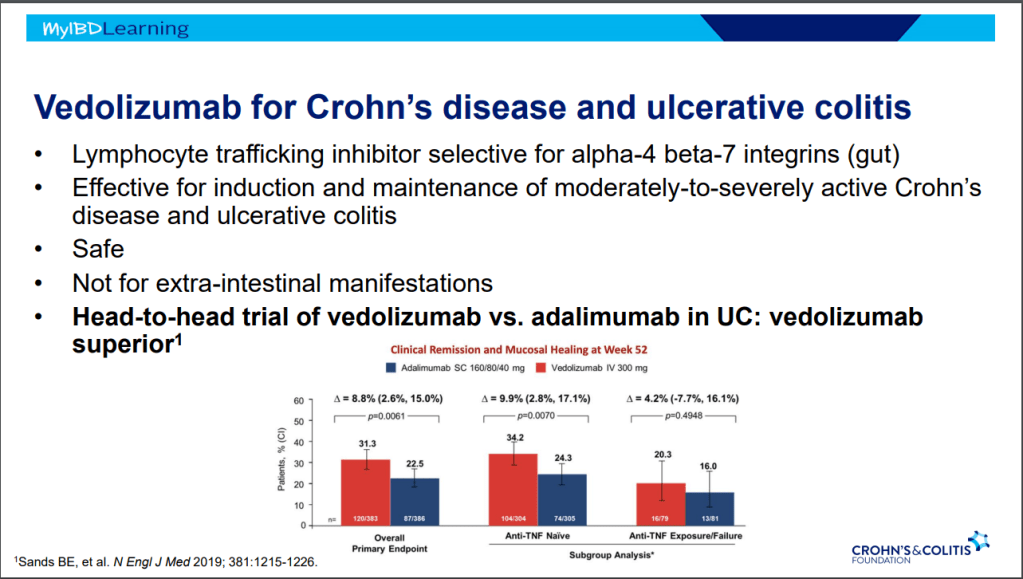
- VARSITY trial enrolled patients with prior anti-TNF exposure (but not adalimumab) which biased the study against adalimumab vs vedolizumab
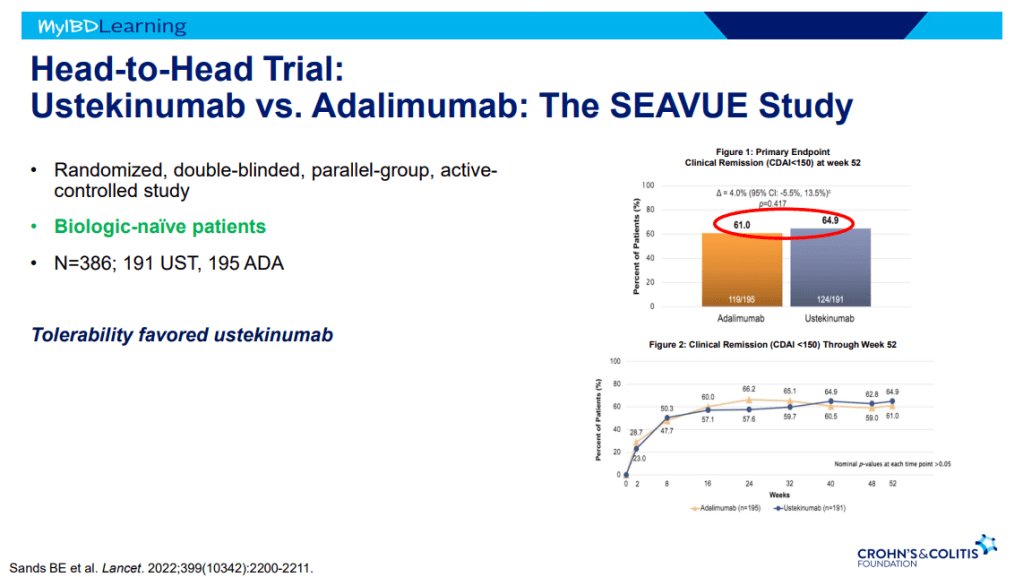
- SEAVUE study ~65% in clinical remission at 1 yr with both adalimumab and ustekinumab; though, ustekinumab had better tolerability
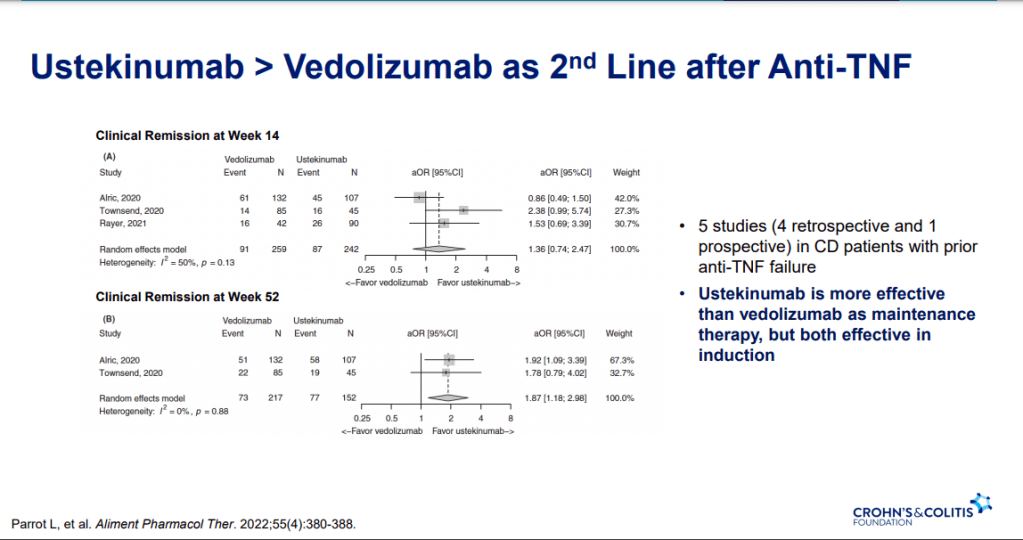
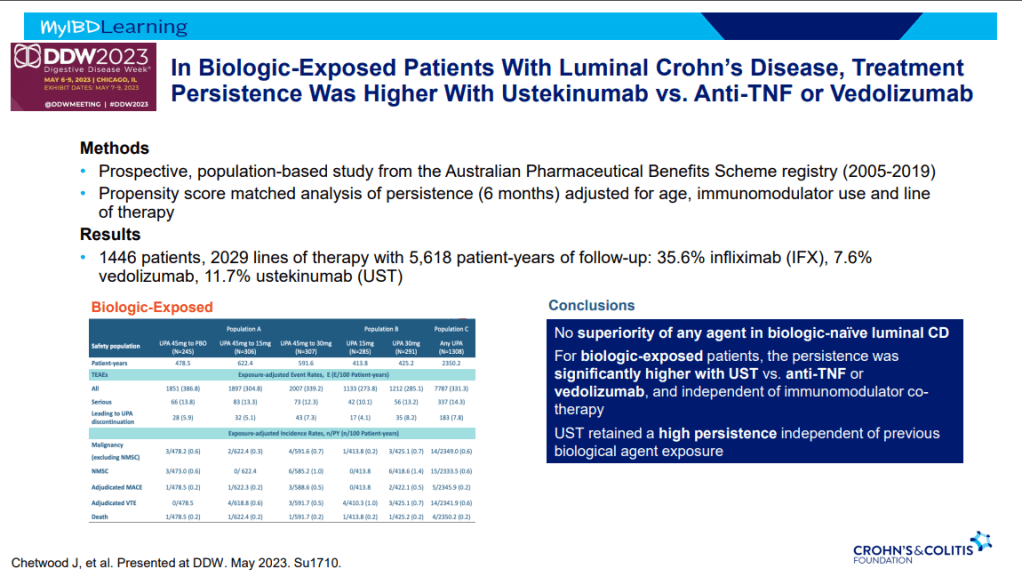
- Ustekinumab better 2nd line treatment for CD after anti-TNF (Parrot et al. AP&T 2022; 55: 380-388
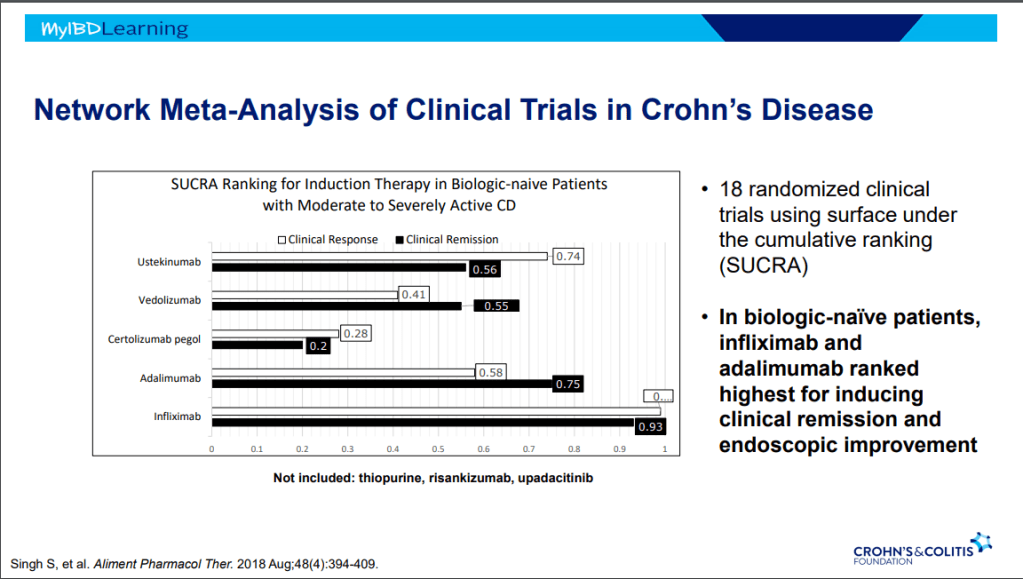
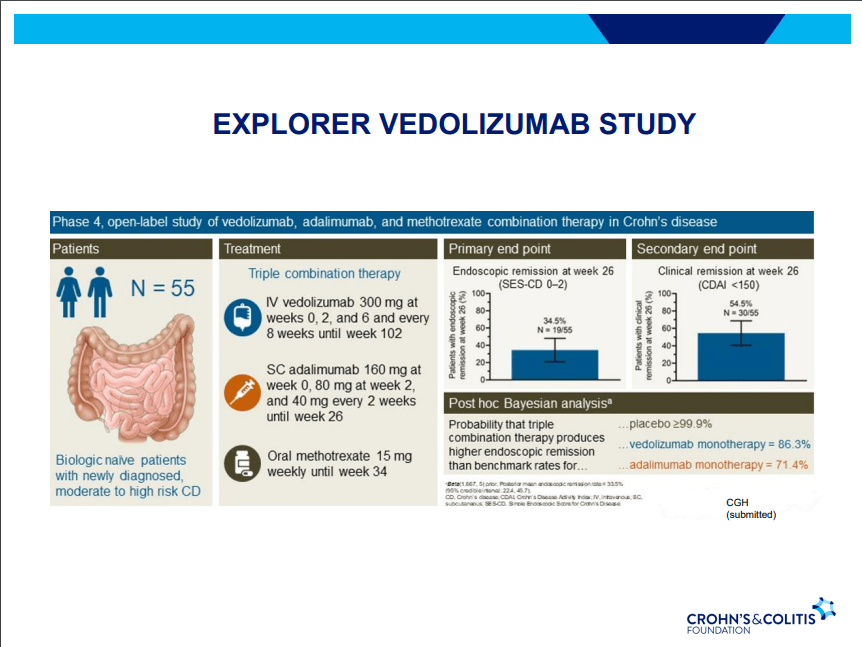
- Vedolizumab is a good therapy for CD, especially in biologically-naïve
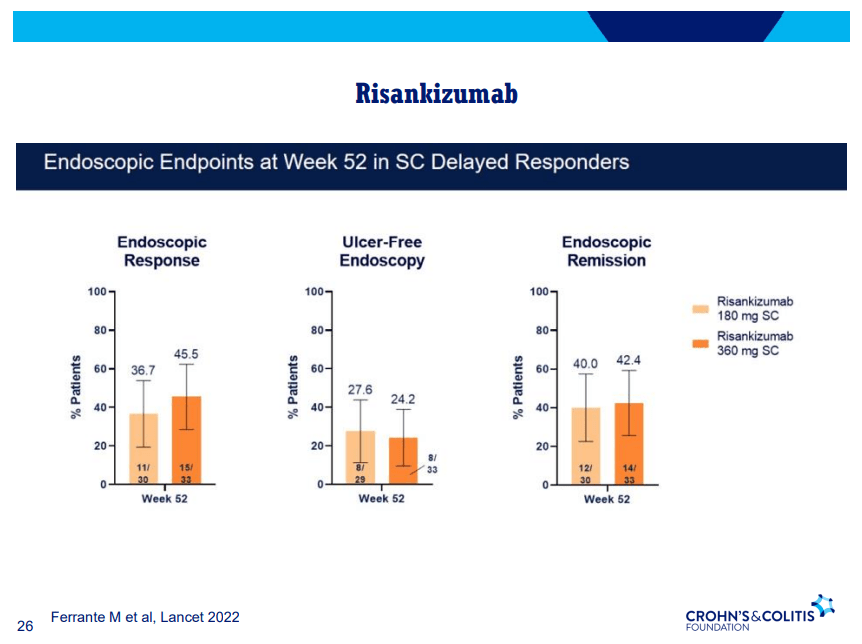
- Risankizumab good for moderate-severe CD both in biologically-naïve and biologically-experienced
- Dose reduction in maintenance of Jak inhibitors can result in LOR and 50-75% can achieve response after resumption of higher dosing