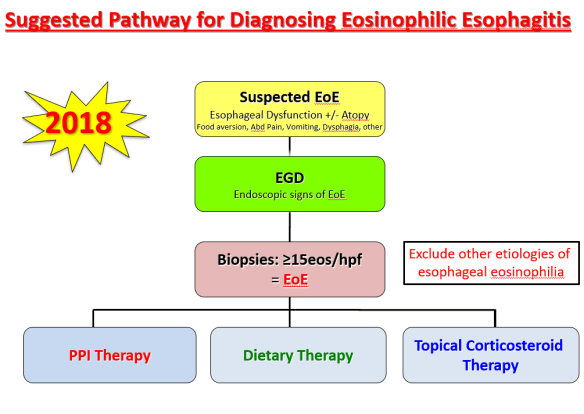
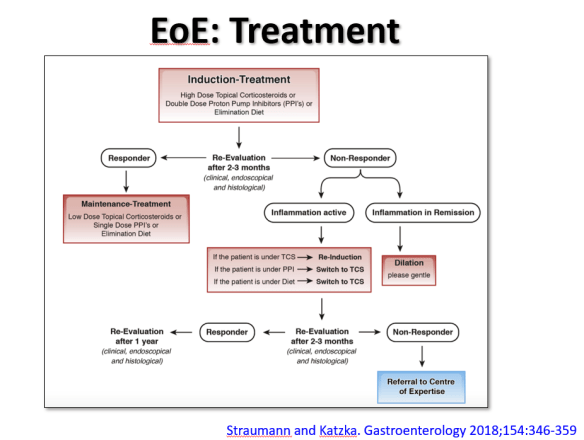
At a recent morning clinical conference, Dr. Seth Marcus provided a terrific update on eosinophilic esophagitis (EoE). I am placing some of the slides below and the following is a link to the full lecture: The Evolution of EoE -Seth Marcus
During this part of the presentation, there was a discussion regarding the role of allergists. Due to the poor predictive value (negative and positive) of allergy testing (skin tests and blood tests), the consensus is that routine allergy evaluation is NOT needed for children with EoE. However, IgE-mediated food allergies along with other atopic diseases are common in children with EoE and selected patients could benefit from allergy referral.
The slide above reviews the main treatment options: topical corticosteroids, proton pump inhibitor therapy, and elimination diet. While all of these are reasonable as first-line approaches, many in the group favored proton pump inhibitor (PPI) treatment as initial therapy. In those with a very good response (<5 eos/hpf at followup), this would allow lower dose PPI as a maintenance option. Another point of discussion was the fact that PPI responders tend to more favorably metabolize the PPIs to achieve higher therapeutic levels. It is anticipated that future treatment could be influenced by knowing the individual’s CYP2C19*17 Polymporphisms (#NASPGHAN17 EoE Session)
Related blog posts:
- Long-term Use of PPIs for EoE
- Do we still need PPI-REE?
- Nexium versus Fluticasone for EoE
- Guidelines for Eosinophilic Esophagitis
- Higher Doses of Topical Steroids for Eosinophilic Esophagitis …
- Putting in Place a Big Piece of the Eosinophilic Esophagitis …
- Eosinophilic Esophagitis Slide Set | gutsandgrowth
- MicroRNA signature for eosinophilic esophagitis | gutsandgrowth
- EoE: Drugs, Diets, Dilatation and PPI-REE