E Ramos Boluda et al. JPGN 2020; 71: 734-739. Experience With Teduglutide in Pediatric Short Bowel Syndrome: First Real-life Data
S Hill. JPGN 2020; 71: 697-698 (editorial) Use of GLP-2 May Herald a New Era of Improved Outcome of Short Bowel Syndrome-associated Intestinal Failure
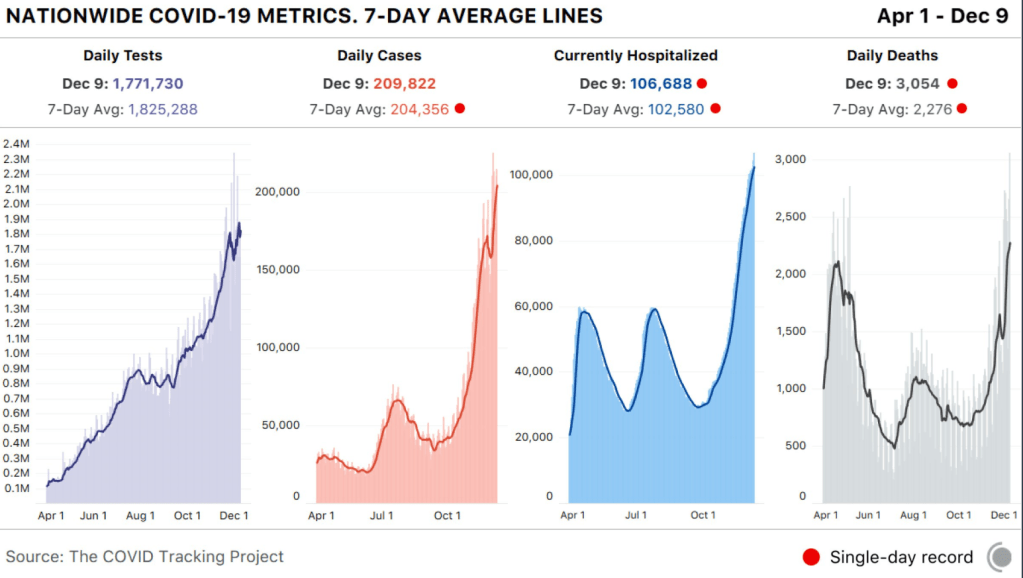
The study and associated editorial highlight the effectiveness of GLP-2 in a prospective cohort of 17 patients with short bowel syndrome. It is noted that Dr. Hill has received funding from the pharmaceutical manufacturer of the product.
Key findings:
- A total of 12 of 17 patients achieved parenteral independence: 3 patients after 3 months of treatment, 4 patients at 6 months, and 5 after 12 months.
- The percentage able to wean off parenteral nutrition was 17%, 44%, and 60% at 3, 6, and 12 months respectively. Only 1 patient did not exhibit improvement
- Plasma citrulline levels, a marker for enteral autonomy, increased from a baseline average of 20 micromol/l to 37.5, 46.75, and37.9at 3, 6, and 12 months respectively.
- Adverse reactions included abdominal pain 30%, nauseas 18%, injection-site reactions 22%, and headache 16%.
Both the editorial and the study comment briefly on the cost of the therapy. The editorial also notes the current recommendation for surveillance endoscopy in view of a hypothetical risk of malignancy.
My take: Is GLP2 Worth the Cost? It probably depends on who is paying and long-term safety data. Perhaps, we will develop tools to improve prediction of which patients will achieve enteral autonomy with GLP2 who would otherwise require ongoing parenteral nutrition.
Related blog posts:
- #NAPSGHAN19 Intestinal Failure Session
- Teduglutide for SBS
- FDA Approves Gattex (Teduglutide) for Pediatric SBS (3rd part of post)