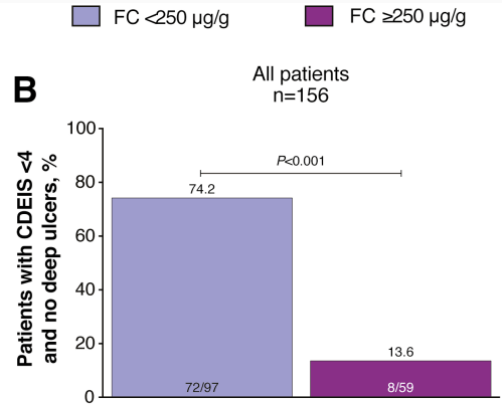
W Reinisch et al. Inflamm Bowel Dis 2020; 1562-1571.Full Text: Association of Biomarker Cutoffs and Endoscopic Outcomes in Crohn’s Disease: A Post Hoc Analysis From the CALM Study n=244.
- The proportion of patients who achieved the primary end point CDEIS <4 and no deep ulcers was significantly greater for those with FC <250 µg/g (74%; P < 0.001)
- Fecal calprotectin <250 µg/g, CRP <5 mg/L, and CDAI <150 gave a sensitivity/specificity of 72%/63% and positive/negative predictive values of 86%/42% for CDEIS <4 and no deep ulcers 48 weeks after randomization
My take: Fecal calprotectin levels are useful for monitoring mucosal healing. Levels less than 250 are encouraging. Levels less than 100 are better.
Related blog posts:
- What is the calprotectin threshold for disease progression in Crohn’s disease?
- Expert Guidance on Inflammatory Bowel Disease (Part 2)
- Prospective Monitoring of Calprotectin for Crohn’s Disease
- An Insurance Company Doing the Right Thing (with Calprotectin)
- Keep the Stool Cool for More Reliable Calprotectin Testing
- Calprotectin in Triaging Potential Pediatric IBD Cases
- Biomarkers identify patients who benefit and how
- Fecal Calprotectin Monitoring for IBD Relapse
- Best Fecal Marker for Crohn’s Disease: Calprotectin | gutsandgrowth
- Value of Calprotectin | gutsandgrowth…
- What Treat-to-Target Could Look Like in Crohn’s Disease
- Treating to Target | gutsandgrowth
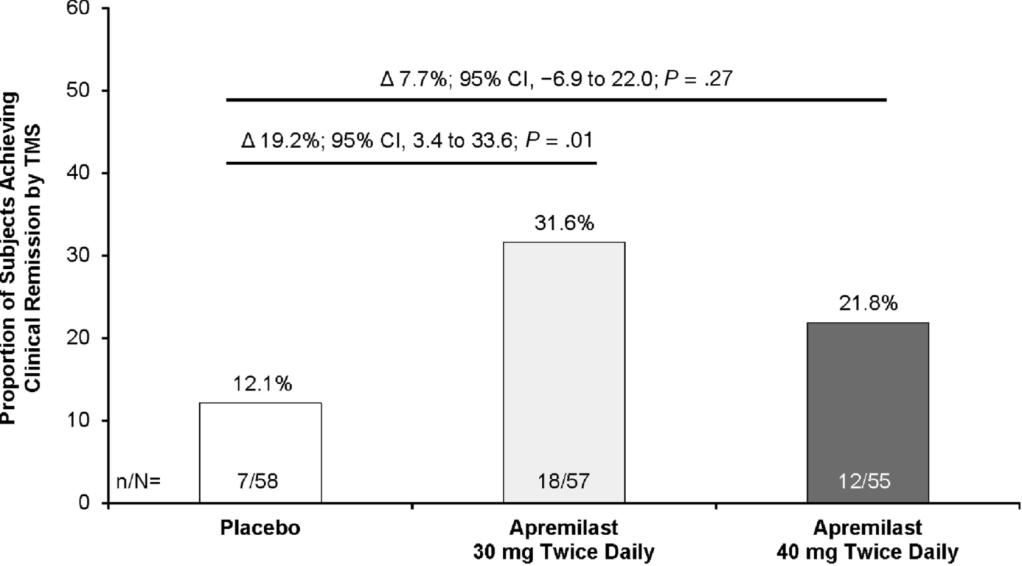
S Danese et al. Clin Gastroenterol Hepatol 2020; 18: 2526-2534. Full text link: Effects of Apremilast, an Oral Inhibitor of Phosphodiesterase 4, in a Randomized Trial of Patients With Active Ulcerative Colitis “We performed a double-blind, phase 2 trial of adults with active UC for 3 months or more who were naïve to biologic therapy or had been failed by, could not tolerate, or had contraindications to conventional therapies.” n=168. Key findings:
- Clinical remission was achieved at week 12 by 31.6% of patients in the 30 mg apremilast group and 12.1% of patients in the placebo group (P = .01). However, only 21.8% of patients in the 40 mg apremilast group achieved clinical remission at week 12 (P = .27 compared with placebo)
- At week 52, clinical remission was achieved by 40.4% of patients initially assigned to the apremilast 30 mg group and 32.7% of patients initially assigned to the apremilast 40 mg group.
X Zhuang et al. Inflamm Bowel Dis 2020; 26: 1636-1647. Full text: Fecal Microbiota Alterations Associated With Clinical and Endoscopic Response to Infliximab Therapy in Crohn’s Disease
Methods: Microbiota was prospectively analyzed in 49 patients with active CD at baseline, week 6, and week 30
Key Findings:
- Increased proportions of Lachnospiraceae and Blautia were associated with IFX efficacy; the combined increase of these taxa at week 6 showed 83.4% and 84.2% accuracy in predicting clinical response at weeks 14 and 30, respectively, with a predictive value of 89.1% in predicting endoscopic response at week 30
- IFX diminished CD-related gut microbial dysbiosis by modifying microbiota composition and function