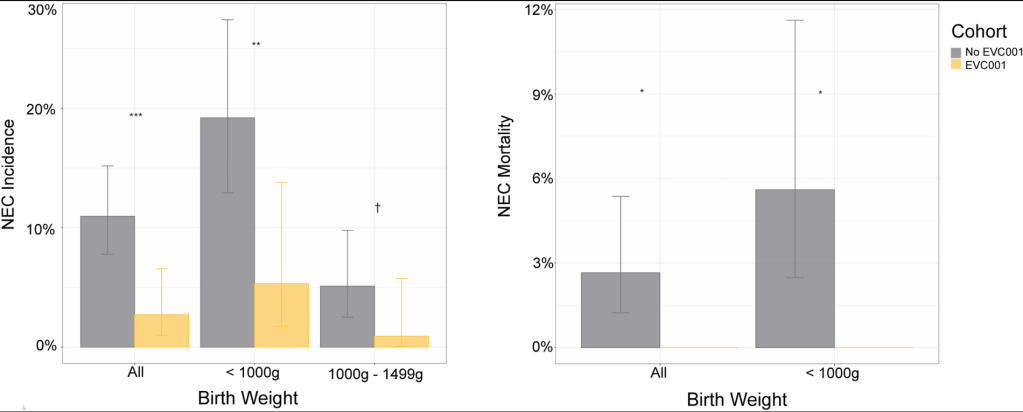
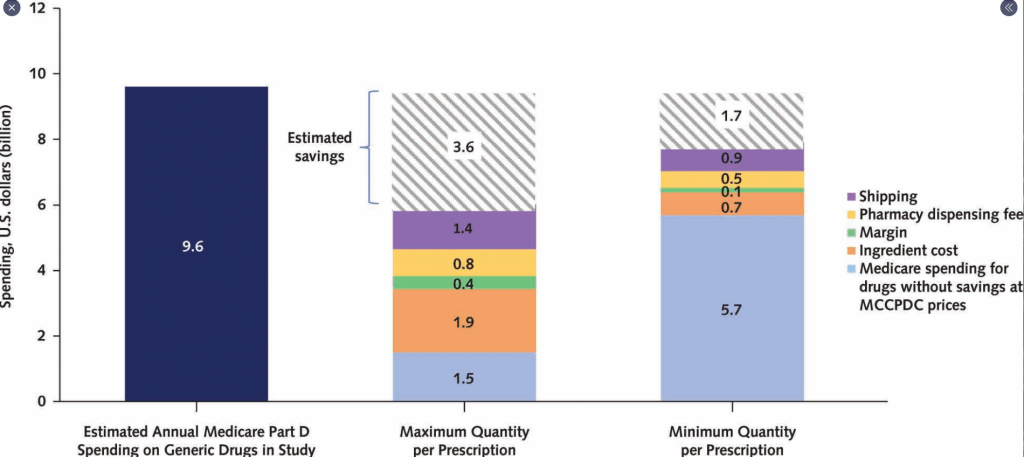
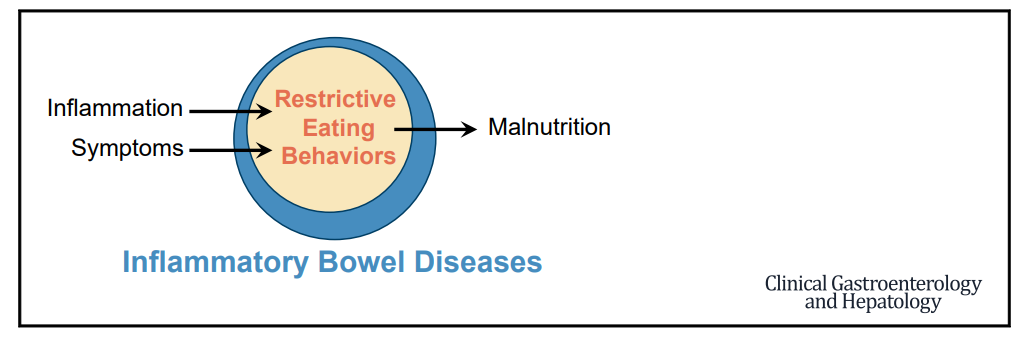
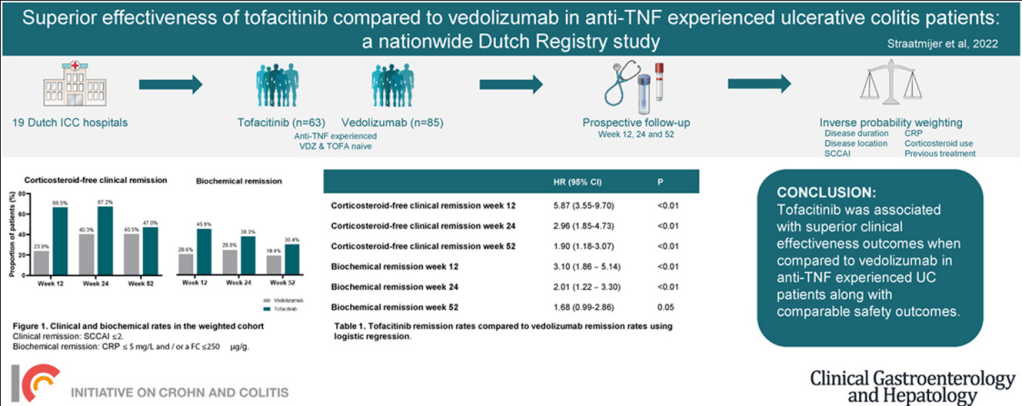
CMA de Bruijn et al. J Pediatr 2022; 244: 107-114. Open Access: Development of a Bowel Management Scoring Tool in Pediatric Patients with Constipation
This report describes the “newly developed and validated PBMST (Pediatric Bowel Management Scoring Tool) is a reliable tool for evaluating bowel management strategies in children with constipation.”
Key finding:
“This study shows that use of the PBMST (see below) can better guide management of childhood constipation, with its fair reproducibility indicating that it is stable over a specified time period. Indeed, consistent use of the PBMST can objectify the patient’s clinical condition over a longer period. Consequently, the score provides feedback regarding the effect of the applied bowel management strategy for each individual patient.”
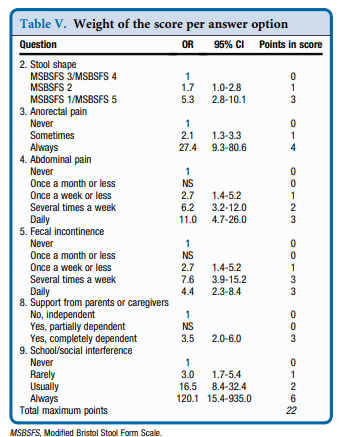
My take: 6 key questions for constipation visits: stool form, anorectal pain, abdominal pain, soiling, support from parents, and social limitations.
Related blog posts:
- Pictographic Constipation Action Plan
- ANMS Virtual Symposia on Constipation (link to video lectures)
- Expert 2017 Opinion: Miralax is (Still) First Choice Laxative for Children
- Stimulants for constipation
- Here we go again…Mirialax Safety
- Miralax Safety
- Kiwi for constipation
- Updated Pediatric Expert Constipation Guidelines
- Diagnostic tests hardly ever help patients poop
Modified Bristol Stool Form (see link: CAM Wegh et al. JPGN 2021; 73: 210-216. The Modified Bristol Stool Form Scale: A Reliable and Valid Tool to Score Stool Consistency in Dutch (Non)Toilet-trained Toddlers)