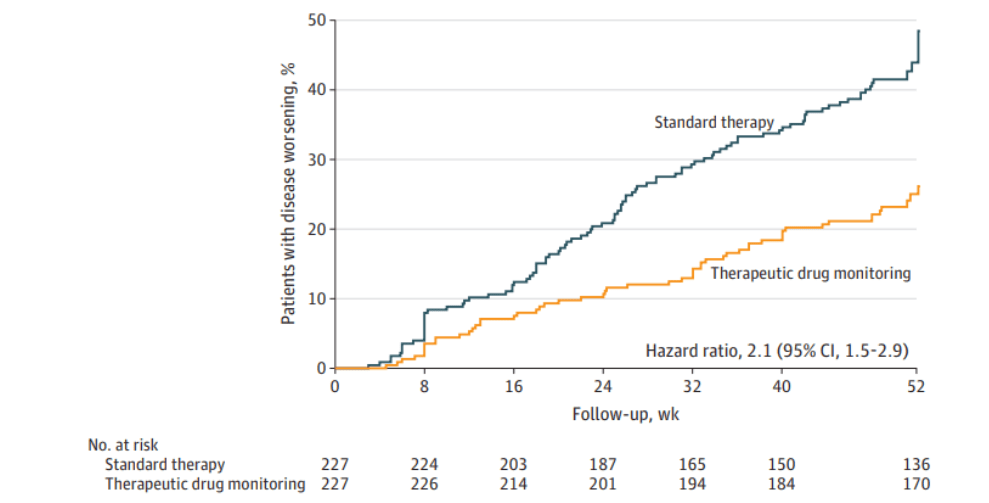
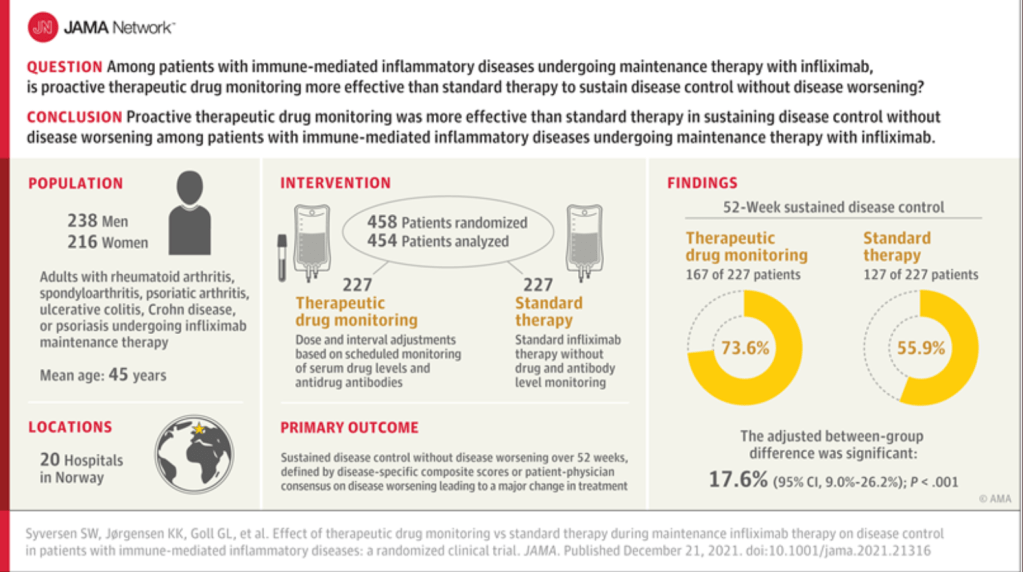
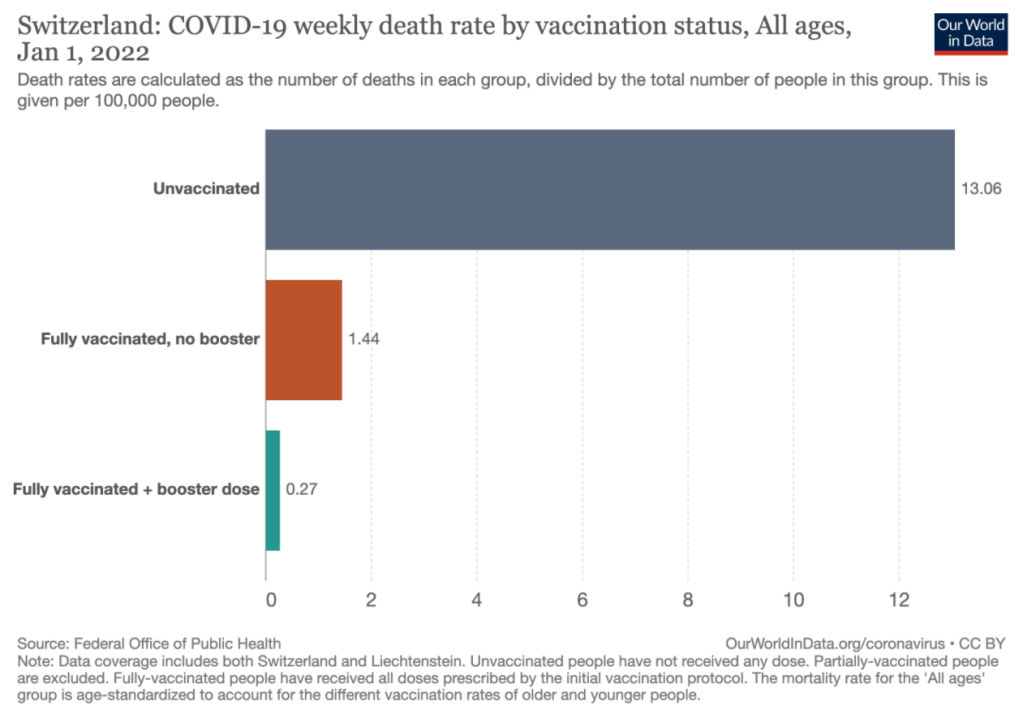
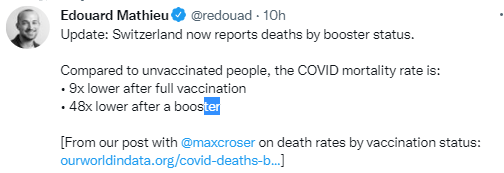A recent commentary (PJ Zettler et al. NEJM 2021; 385: 2409-2411. A Divisive Ruling on Devices — Genus Medical Technologies v. FDA) highlights another challenge facing the FDA’s role in regulating devices, especially as the definition of devices and medications becomes more murky.
Key points/excerpts:
- In April 2021, however, the U.S. Court of Appeals for the District of Columbia decided, in Genus Medical Technologies v. FDA, that products meeting the FDCA’s definition of a device “must be regulated as devices”…The Genus case concerned a challenge to the discretion of the FDA to classify Genus Medical Technologies’ Vanilla SilQ line of diagnostic barium sulfate contrast agents as drugs rather than as devices…FDCA’s drug and device definitions overlap.
- Ultimately, the D.C. Circuit Court held that products meeting both definitions must be regulated as devices because the FDCA’s “text unambiguously forecloses the FDA’s interpretation.” The court did not give the FDA so-called Chevron deference, a doctrine under which courts defer to an agency’s reasonable interpretation of a statute that it implements, if the statute is ambiguous.
- Because technological innovation often outpaces legal change, we believe it could serve both public health and industry for the FDA to retain some discretion regarding the most appropriate regulatory pathways for anticipated and as-yet-unforeseen device classes… overly restricting the FDA’s flexibility to use its expertise could endanger important public health priorities, especially when there are compelling reasons why Congress permitted flexibility.
- The Genus decision comes at the end of a decade of growing challenges to the FDA’s authority to regulate. From court decisions in the early 2010s that were sympathetic to arguments that FDA policies regarding off-label drug promotion violate the Constitution’s First Amendment, [and] to the political (and legislative) success of right-to-try laws
My take: This commentary shows the difficulties the FDA faces as the judiciary curtails its discretion. While the legislative branch could codify FDA authority, this is unlikely. As such, this will limit FDA oversight in some new medical products. When problems arise, it is likely that the FDA (not the courts) will be blamed despite the fact that their ability to regulate has been undermined.
Related blog posts:
- Is An Unproven Medication Worth More Than the EPA or NASA?
- Can the FDA prohibit free speech?
- FDA Postapproval Studies: ‘You can have pie as long as you do your homework later’
- Can the FDA stop snake oil salesmen?