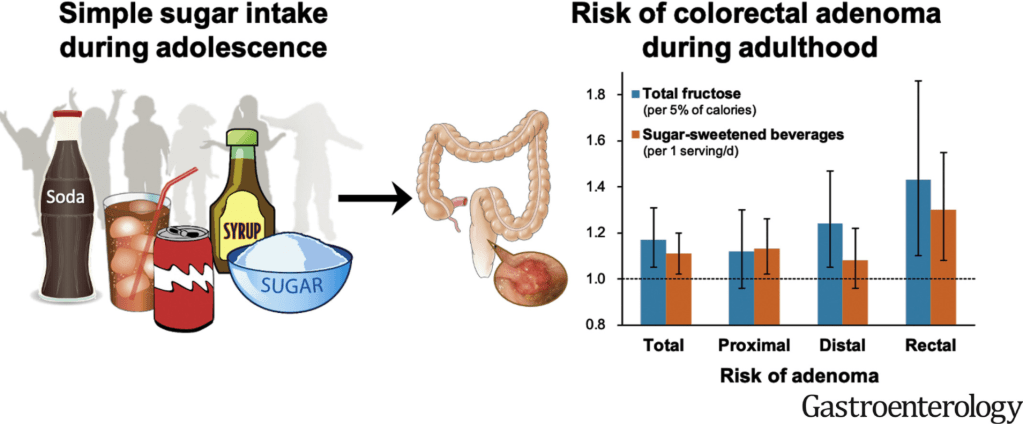
G D’Arcangelo et al. JPGN 2021; 73: 242-246. Is Fecal Calprotectin a Useful Marker for Small Bowel Crohn Disease?
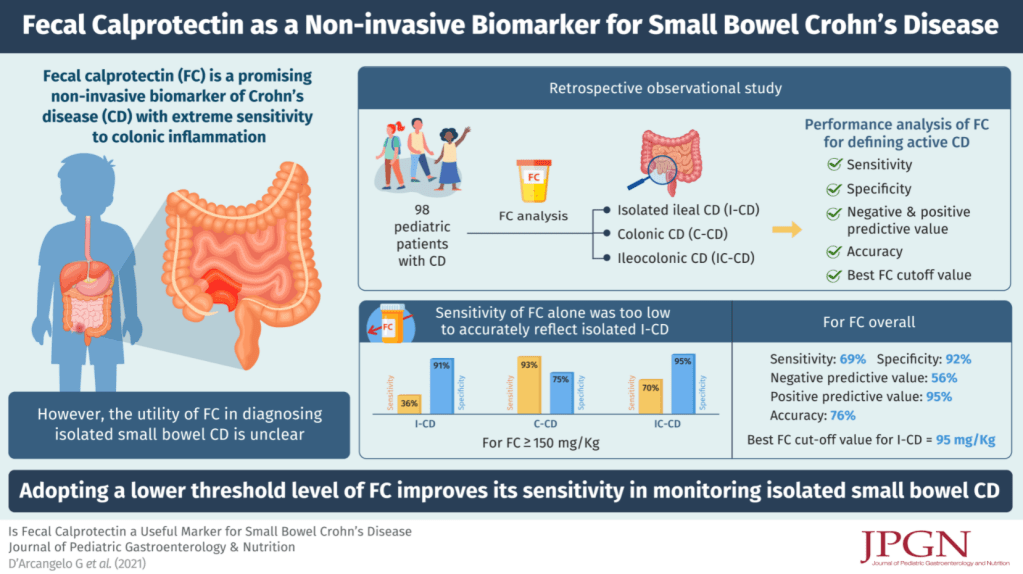
In this retrospective study with 98 patients, the authors examined the sensitivity and specificity of fecal calprotectin (FC) at a cutoff of 150 mcg/g in comparison to findings of ileocolonoscopy and MRE in those with isolated ileal CD (L1, n=14), colonic CD (L2, n=10) and ilecolonic CD (L3, n=74) . Note: the abstract erroneously states the cutoff as 50 mcg/g.
Key findings:
- The sensitivity and specificity of FC for L1 CD were 36% and 91%, respectively, compared to 93% and 75% for L2 and 70% and 95% for L3.
- An FC of 95 mg/kg was identified as the best cut off for identification of active isolated ileal disease, with a sensitivity of 77% and a specificity of 56%
My take: Though this study had only 14 patients with isolated ileal disease, it is likely that a calprotectin level is less reliable as a biomarker in these patients.
Related article: Jukic A, Bakiri L, Wagner EF, et al Calprotectin: from biomarker to biological functionGut Published Online First: 18 June 2021. doi: 10.1136/gutjnl-2021-324855. Thanks to KT Park for this reference. Open Access- Full text: Calprotectin: from biomarker to biological function
Related blog posts:
- What is the Calprotectin Threshold for Crohn’s Disease Progression?
- An Insurance Company Doing the Right Thing (with Calprotectin)
- Keep the Stool Cool for More Reliable Calprotectin Testing
- Calprotectin in Triaging Potential Pediatric IBD Cases
- Biomarkers identify patients who benefit and how
- Fecal Calprotectin Monitoring for IBD Relapse
- Best Fecal Marker for Crohn’s Disease: Calprotectin | gutsandgrowth
- Value of Calprotectin | gutsandgrowth
- Prospective Monitoring of Calprotectin for Crohn’s Disease …
- What Treat-to-Target Could Look Like in Crohn’s Disease