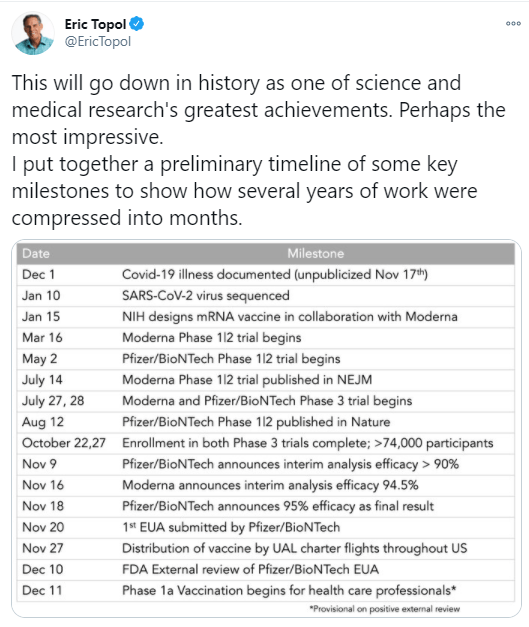
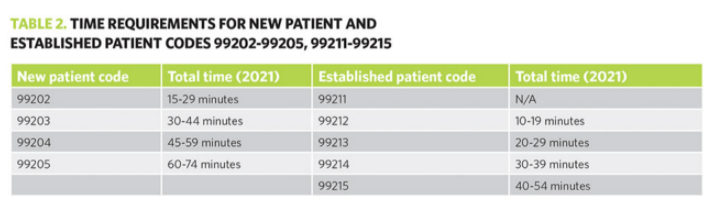
M Thanhaeuser et al. J Pediatr 2020; 226: 142-148. A Randomized Trial of Parenteral Nutrition Using a Mixed Lipid Emulsion Containing Fish Oil in Infants of Extremely Low Birth Weight: Neurodevelopmental Outcome at 12 and 24 Months Corrected Age, A Secondary Outcome Analysis
This study evaluated neurodevelopmental outcomes using Bayley Scales. the authors provided a secondary outcome analysis of a double-blind randomized trial of 206 extremely low birth weight infants. Participants received either SMOFlipid or soybean oil-based lipid. Lipids were dosed at </+ 3 g/kg/day.
Key findings:
- Parenteral nutrition using a mixed lipid emulsion (SMOF) containing fish oil did not improve neurodevelopment of extremely low birth weight infants at 12 and 24 months corrected age
- At 24 months of age, specifically, there was again no significant differences in any of the following areas (median values):
- cognitive: SMOF: 95 & soybean oil: 95
- language: SMOF: 89 & soybean oil 89
- motor scores: SMO 94 & soybean oil: 94
Limitations: One of the reasons why this study did not find any difference is that it was not powered for assessment of neurodevelopmental outcomes. The authors provide other potential reasons:
- DHA in SMOFlipid provided 43 mg/kg/d, while more than the soybean-lipid, is at the lower end of published fetal accretion rates (40-67 mg/kg/day)
- DHA deficits may not have been pronounced enough in this study to see an effect of SMOFlipid on neurodevelopement
- Full feeds were reached after 23 days (IQR, 17-37 days); thus, it is possible that infants with longer term dependency on parenteral nutrition would benefit more
My take: SMOFlipid has not been proven to have more favorable long-term neurocognitive effects than intralipid. However, for children with prolonged need for parenteral nutrition, SMOFlipid is more likely to allow full dosing which in itself may be an important contributor to better outcomes. That is, soybean-lipid emulsions are more likely to be reduced due to cholestasis and this could lead to nutritional deprivation.
Related blog posts:
- SMOFlipid vs Intralipid for Intestinal Failure Patients
- Double-Blind Randomized SMOFlipid Study
- Nutrition Week: SMOFlipid
- Optimizing lipids to minimize cholestasis
- Neonatal Nutrition Lecture -What We Know Right Now …
- Improving Outlook in Neonatal Nutrition (Part 1)
- Current Mortality from Being Born Premature | gutsandgrowth
- Visual Acuity and LCPUFA | gutsandgrowth
- Weight Gain in Preemies, Neurodevelopmental Outcomes, and Reverse Causation
- Medical Progress for Intestinal Failure Associated Liver Disease