M Bohm et al. AP&T: 2020; July 2020 https://doi.org/10.1111/apt.15921 Full text: Comparative safety and effectiveness of vedolizumab to tumour necrosis factor antagonist therapy for Crohn’s disease
Thanks to Ben Gold for this reference.
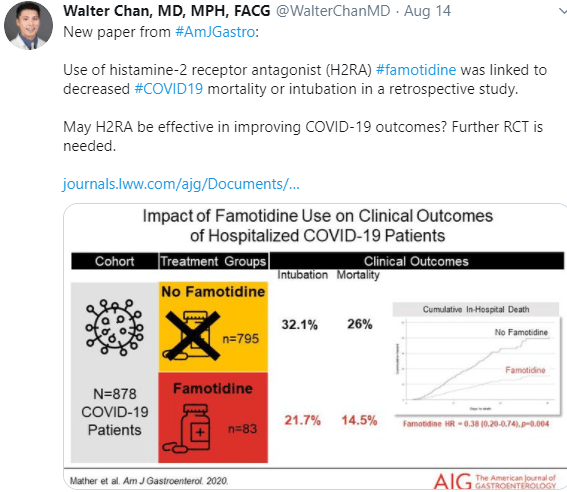
Methods: Retrospective observational cohort (May 2014–December 2017) propensity score‐weighted comparison of vedolizumab vs TNF‐antagonist therapy (infliximab, adalimumab, certolizumab) in CD. This study included 1266 patients (n = 659 vedolizumab).
Key findings:
- Rates of non‐infectious serious adverse events (odds ratio [OR] 0.072, 95% confidence interval [CI] 0.012‐0.242) were significantly lower with vedolizumab vs TNF‐antagonist therapy.
- These events included severe arthralgias in 3 vedolizumab-treated patients. For anti-TNF recipients, events included hypersensitivity or infusion reactions (n = 6), drug‐induced psoriasis (n = 6), drug‐induced lupus (n = 5), severe liver function test abnormalities (n = 3), skin rash (n = 2), lung cancer (n = 1) and jaw or hip necrosis (n = 2).
- Rates of serious infections (OR 1.183, 95% CI 0.786‐1.795), were NOT significantly lower with vedolizumab vs TNF‐antagonist therapy.
- “The risk of serious infections with biologic therapy is largely driven by disease activity and concomitant use of immunosuppressive agents…. the higher concomitant use of steroids among the vedolizumab‐treated patients in our cohort may therefore help to explain the lack of observed difference in risk for serious infections between agents.”
- No significant difference was observed between vedolizumab and TNF‐antagonist therapy for clinical remission (hazard ratio [HR] 0.932, 95% CI 0.707‐1.228), steroid‐free clinical remission (HR 1.250, 95% CI 0.677‐2.310) or endoscopic remission (HR 0.827, 95% CI 0.595‐1.151).
- “Our observational cohort study was not designed to be a noninferiority study, and the safety and effectiveness comparisons were exploratory in nature.”
- The efficacy of vedolizumab in this study is more impressive given that 91% of the patients had prior anti-TNF therapy.
- “Exploratory subgroup analyses suggested that vedolizumab might be superior to subcutaneous TNF‐antagonist therapy for the achievement of clinical remission and steroid‐free clinical remission in TNF‐antagonist–naïve patients.”
- TNF‐antagonist therapy was associated with higher treatment persistence compared with vedolizumab.
My take: This article shows that clinical experience with vedolizumab is quite good and compares favorably with anti-TNF agents. Randomized head-to-head studies are needed, though, to truly determine efficacy in similar populations.
Related blog posts:
- Expert Guidance on Current IBD Mgt (2020)
- Real-World Vedolizumab: Better Than Expected
- Predicting Response to Vedolizumab and Ustekinumab for Inflammatory Bowel Disease
- Ustekinumab Over Vedolizumab for Crohn’s Disease as a 2nd Line Agent for Crohn’s disease
- IBD Highlights from Recent Meetings with Commentary by Dr. Sandborn Explains Why Vedolizumab Should Be Considered a First Line Agent (2019)
- VICTORY Consortium Data for Vedolizumab 2018
- Vedolizumab More Effective Than Adalimumab for Ulcerative Colitis (Part 2)
- Vedolizumab More Effective Than Adalimumab for Ulcerative Colitis
- Vedolizumab vs Adalimumab for Infliximab Failure in Ulcerative Colitis –Which is Better?
- Getting the Most Out of Vedolizumab
- Enthusiasm for Vedolizumab
- Expert Guidance on Inflammatory Bowel Disease (Part 3)
- Expert Guidance on Inflammatory Bowel Disease (Part 2)