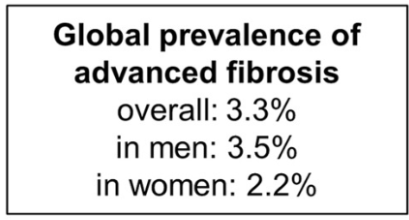
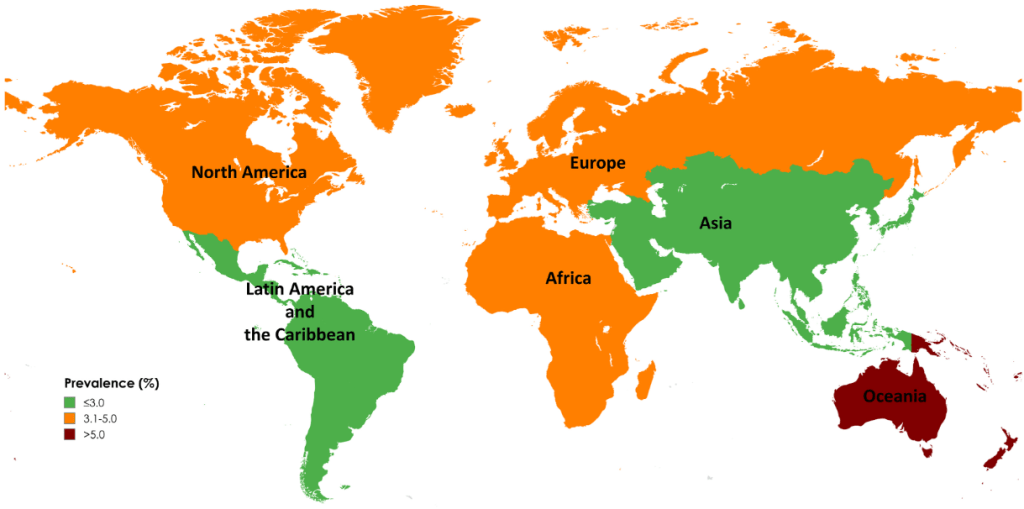
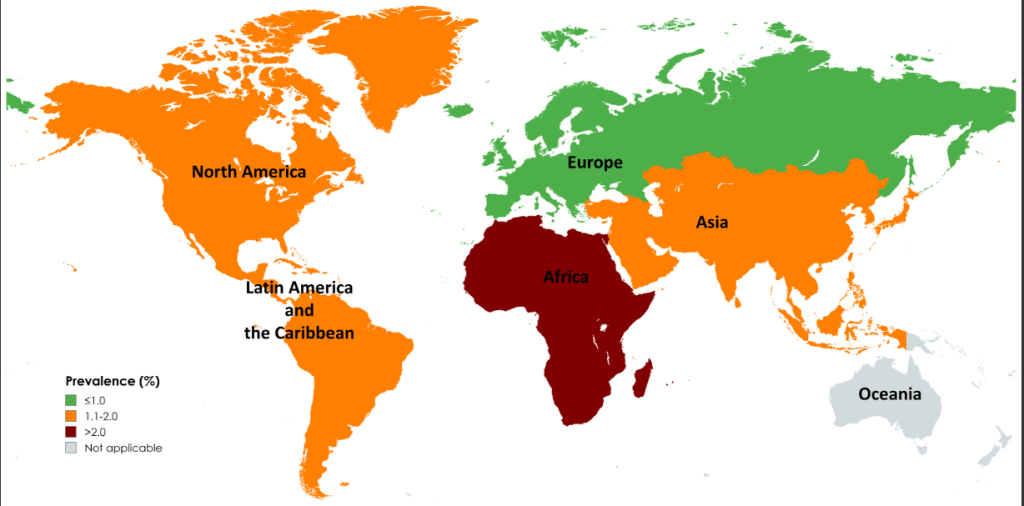
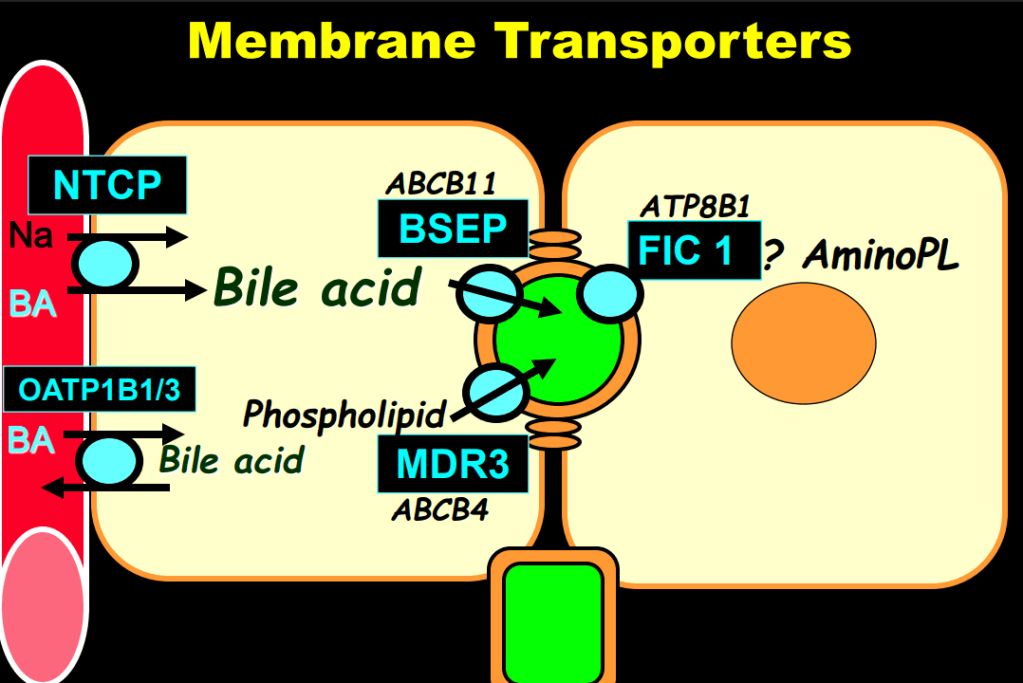
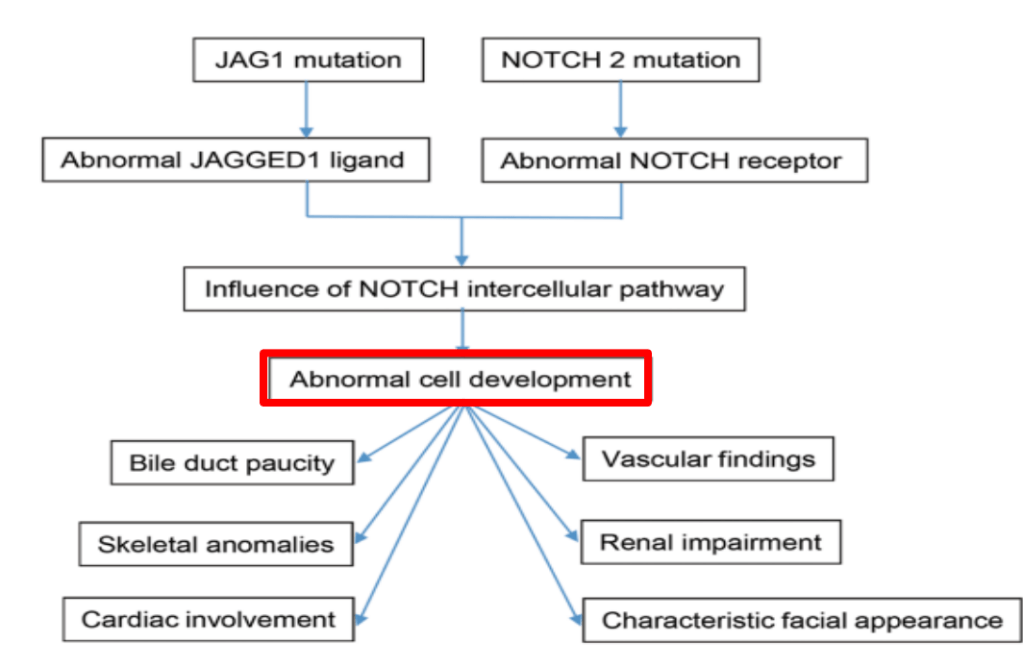
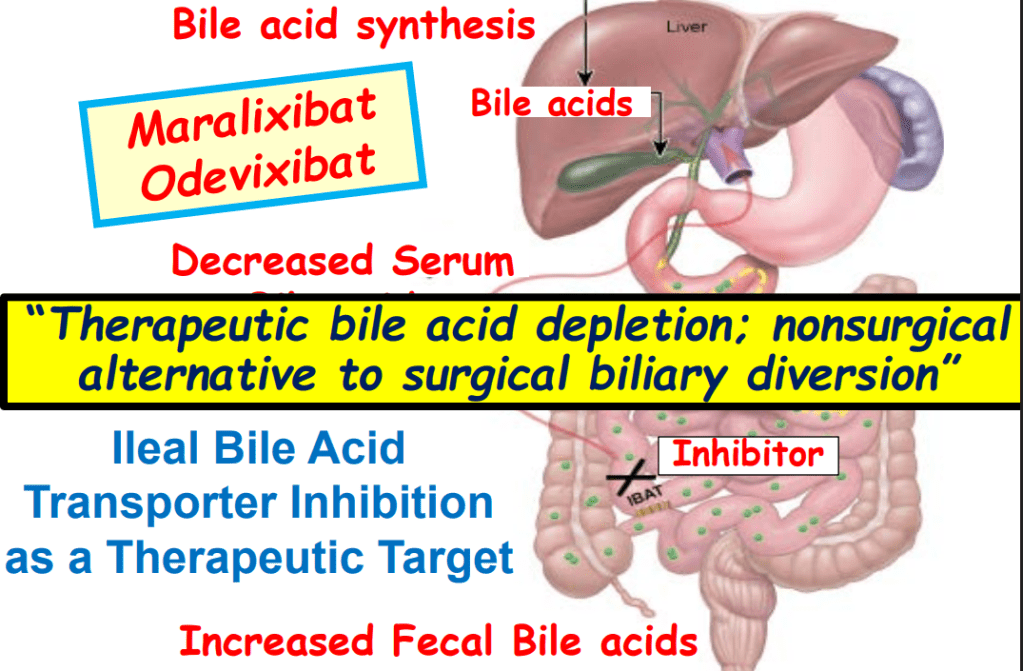
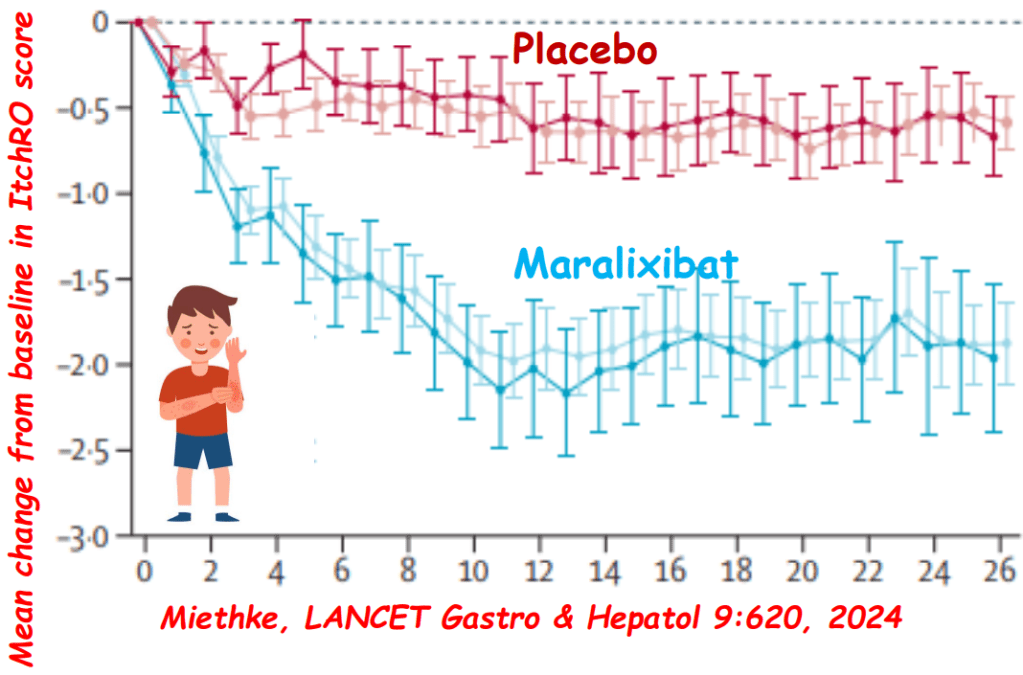
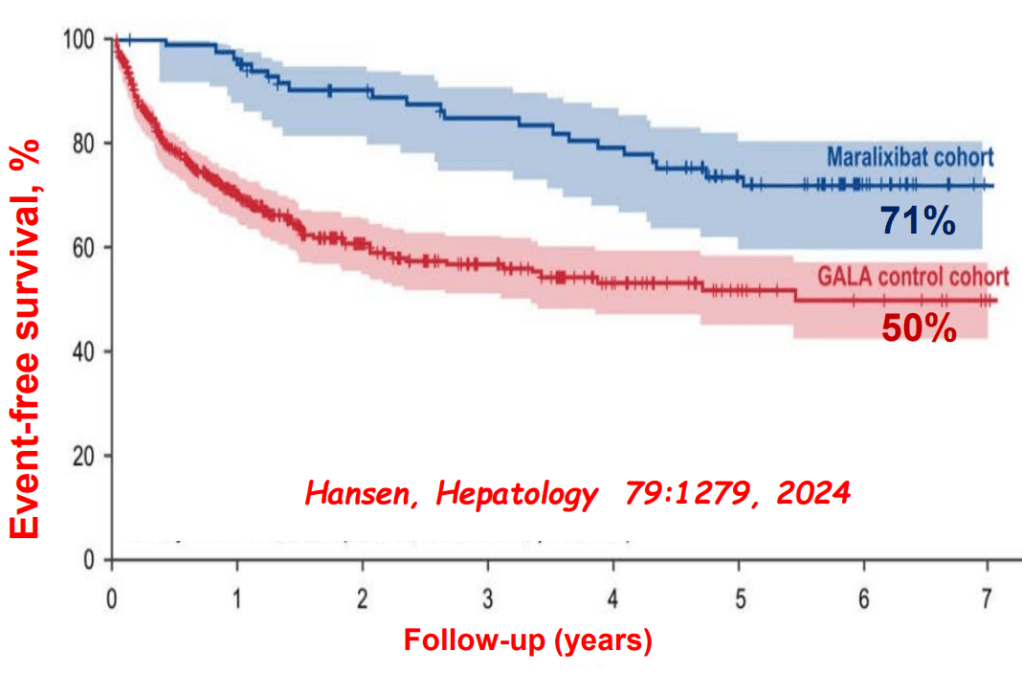
Link: Family reflections: research gives back childhood: a family’s experience with very early onset inflammatory bowel disease Pediatric Research; https://doi.org/10.1038/s41390-024-03506-8
An excerpt:
Very Early Onset Inflammatory Bowel Disease is so rare and individualized that no standard of care yet exists, and almost none of the interventions are approved for infants. After eliminating all dairy, prematurely ending breastfeeding, and moving exclusively to a prescription formula, we tried and failed multiple classes of medications, hoping each time that this medication would be the one that would ease our son’s suffering. Doctors are only now building a history of successful interventions to draw from, so treatment options come from very small studies from Very Early Onset Inflammatory Bowel Disease researchers and educated guesses. Treatment options generally ramp up in aggressiveness, which is hard enough for parents of a miserably sick infant to process and decide. Once these interventions fail, drug costs and insurance approval become major hurdles. This process of trial-and-error in treating Very Early Onset Inflammatory Bowel Disease patients is a nightmare. On a weekly or monthly basis, parents must make life-altering decisions with very little data for a patient too young to advocate for themselves.
Link: The VEO-IBD Foundation This foundation is still in its early stages, but anticipate it will be a resource for families with VEO-IBD.
Related blog posts:
- Biologics in Children with Very Early Onset Inflammatory Bowel Disease
- IBD Updates: Outcomes of VEO-IBD, PIANO Study Update, and Insurance-Disparity Relationship
- VEO-IBD -Useful “Position” Paper is Really a Review
- Targeted Therapy for Autoinflammatory Very Early Onset Inflammatory Bowel Disease
- How Very Early Onset-Inflammatory Bowel Disease is Different, Plus One
- IBD Update -Stem Cells for Perianal Disease, Medicine-Induced VEO-IBD, and Thalidomide for VEO-IBD
- Patterns and Puzzles with Very Early Onset Inflammatory Bowel Disease
- Comparative Evidence and Positioning Advance Therapies for Inflammatory Bowel Disease
- Immune Dysregulation and Inflammatory Bowel Disease