DILI, DILI -Two Studies on Drug-Induced Liver Injury
A Benesic et al. Clin Gastroenterol Hepatol 2018; 16: 1488-94. This prospective study found that monocyte-derived hepatocyte-like (MH) cells isolated from patients could be used to test and identify drugs that triggered acute liver injury. Among 40 patients, 13 patients had 10 drugs identified which were toxic to MH cells. Overall, they reported the MH test as having a 92% sensitivity and 100% specificity.
I Medina-Caliz et al. Clin Gastroenterol Hepatol 2018; 16: 1495-1502. Using the Spanish DILI registry (1994-2016), the authors identified 32 of 856 cases of DILI that were due to dietary supplements. Patients were more often female (63%), and had a mean ALT level 37-fold above ULN. 3 patients (9.4%) progressed to acute liver failure. Many of these supplements were promoted as helpful for weight loss. The authors speculate that reported cases of DILI due to herbal supplements are ‘the tip of the iceberg’ due to under-reporting of cases.
Related blog posts:
- Advice on drug-induced liver disease
- Liver toxicity -where to look online | gutsandgrowth
- More data on DILI | gutsandgrowth
- Data on Drug-Induced Liver Injury | gutsandgrowth
- Predicting a Bad End in Drug-Induced Liver Injury | gutsandgrowth
- Liver Problems with Inflammatory Bowel Disease | gutsandgrowth
- Web is Better: Liver Toxicity from Herbs | gutsandgrowth
- Teaching an Old Drug New Tricks | gutsandgrowth
Do Biologics Alter the Natural History of Crohn’s Disease in Children?
An important recent study (B Kerur et al. Clin Gastroenterol Hepatol 2018; 16: 1467-73 & editorial C Ballengee S Kugasthasan 1398-1400) examined the impact of biologic therapies on Crohn’s disease progression and need for surgery in 1442 children (age, ≤16 y) between 2002-14. This study examined data from the Pediatric Inflammatory Bowel Disease Collaborative Research Group registry.
Key findings:
- Early use of biologics (n=145) was associated with slowing of disease progression (hazard ratio 0.85, CI 0.76-0.95). Those who received anti-TNF therapy within three months of diagnosis were less likely to develop stricturing (B2) or penetrating (B3) disease.
- Early anti-TNF therapy did not effect progression to surgery. Surgery rates were 4% at 1 year, 13% at 5 years, and 26% at 10 years.
- Of those who needed surgery, ~15% already had their first bowel-related surgery in the first 90 days after diagnosis.
- The study cohort at diagnosis included only 51 with B2 disease, 27 with B3, and 11 with both B2 & B3. Thus, these three disease phenotypes represented ~6% of the entire cohort.
In the editorial, the authors state that this study “is a sobering reminder that we apparently have not changed the long-term course of CD for our pediatric patients.” Though, at the same time, they explain how this study had some limitations which could have affected some of the conclusions.
- In contrast to the RISK study, this study classified patients as B1 who progressed to B2 or B3 in the first 3 months of diagnosis. Including these patients decreased the chance to show improvement with early biologic therapy.
- Also, this cohort included a lower percentage of African American patients compared to the RISK study (8% vs 13%). This also lowered the likelihood of identifying improvement; these patients are more likely to develop penetrating disease which can be prevented with early biologic therapy (RISK study: Kugasthasan S et al. Lancet 2017; 389: 1710-8).
Also, one other finding of the study was that there was a paradoxical increase in the risk of surgery in the first 5 years in the early biologic group. “This suggests that our practicing pediatric gastroenterologists may have selected the sicker patients to start biologics.”
My take: I think biologics do influence the natural history of Crohn’s disease in children. However, this study suggests that the magnitude of that alteration is suboptimal.
Related blog post: CCFA update 2017 -RISK study presentation
Vedolizumab and Extraintestinal Manifestations of Inflammatory Bowel Disease
A recent retrospective study( MC Dubinsky et al. Inflamm Bowel Dis 2018; 24: 1876-82) indicates that vedolizumab (VDZ) is likely to less effective than anti-TNF agents for extraintestinal manifestations of inflammatory bowel disease (IBD).
The authors used the MarketScan database (2102-2016). For Crohn’s disease (CD) this included 756 treated with VDZ and 19584 treated with anti-TNF. For ulcerative colitis (UC), this included 544 treated with VDZ and 8574 treated with anti-TNF.
Key findings:
- Compared to patients receiving anti-TNF therapy, VDZ-treated CD patients were 28% more likely to develop “any EIMs” with an adjusted rate ratio of 1.49. The adjusted rate ratio of developing specific EIMs: erythema nodosum 4.29, aphthous stomatitis 3.71, episcleritis/scleritis 2.51, arthropathy 1.45, primary sclerosing cholangitis (PSC) 7.79, and uveitis/iritis 2.89.
- VDZ-treated UC patients did not have a statistically-significant increase in general for EIMs; though when looked at individually, there was increased incident rate ratios for some: apthous stomatitis 3.67, pyoderma gangrenosum 4.42, and PSC 3.44.
The authors findings are counter to their hypothesis that VDZ-treated patients would not have a significantly higher incidence of EIMs and that the EIMs would parallel course with IBD. To explain their findings, the authors note the following:
- “EIMs may be more associated with systemic inflammation than previously thought.”
- “Alternatively, the correlation between specific EIMs and underlying intestinal disease activity may be less tight than previously described.”
- Anti-TNFs may control intestinal inflammation better than VDZ
- VDZ-treated patients may have had more severe disease
While EIMs are more likely to develop on VDZ therapy, this study and prior RCTs do not show whether VDZ is effective in resolving EIMs.
My take: This retrospective study indicates that EIMs, including PSC, are more likely to occur in patients receiving vedolizumab. It is unclear whether this is related to the gut-specific control of inflammation with VDZ or whether there are patient characteristics responsible for this observation.
Related blog posts:
Expansive View of Endoscopy from Porto IBD Group
The pediatric IBD Porto Group of ESPGHAN has updated endoscopy guidelines: S Oliva et al. JPGN 2018; 67: 414-430. In total, the authors make 17 recommendations –here are a few of them:
A) In non-emergency situations, the diagnostic evaluation for suspected IBD in children should include a combination of EGD and colonoscopy. Multiple biopsies from each segment are recommended even in the absence of macroscopic disease.
B) Endoscopic evaluation is recommended for the following:
- before major treatment changes
- in symptomatic patients when it is not clear whether the symptoms are inflammation-related
- in Crohn’s disease(CD) to ensure mucosal healing during clinical remission
- in Ulcerative colitis (UC) to ensure mucosal healing during clinical remission only if fecal calprotectin is elevated
C) 6-12 months after bowel resection to identify postoperative recurrence
D) Endoscopic surveillance in pediatric UC after 10 years from the onset of disease (as early as 8 years in older children (>16 years) with risk factors like extensive disease and strong family history
E) In patients with concurrent primary sclerosing cholangitis (PSC), surveillance colonoscopy may be considered every 1-2 years, starting from time of PSC diagnosis. However, in children <12 years of age, surveillance could be postponed based on individual risk factors.
In addition to discussions of conventional endoscopy, the authors favor evaluation of small bowel inflammation: “the choice to perform CE [capsule endoscopy], MRE or both, depends on local availability and expertise.” The authors caution to consider strictures and the potential need for patency capsule prior to CE.
Conclusion of authors: “Endoscopy in pediatric IBD provides a more definitive diagnosis and disease extent evaluation, assesses therapeutic efficacy and leads to targeted therapy, which lessens complications and progression.”
My take: While I agree that endoscopy increases our understanding of disease extent and response to treatment, I do have some concerns about the recommendations (under section B above) regarding assessment of mucosal healing. Part of the concern is that there is not a single accepted definition of mucosal healing. Also, as a practical matter, there needs to be a discussion of the costs and more proof that frequent endoscopy will improve outcomes; it is possible that increased use of endoscopy will lead to some detrimental outcomes in some patients based on the interpretation of the results (eg. dropping a therapy that may be helping and replacing with a less effective treatment)..
Disclaimer: These blog posts are for educational purposes only. Specific dosing of medications/diets (along with potential adverse effects) should be confirmed by prescribing physician/nutritionist. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.
Pediatric Fatty Liver Disease Study: New York City
A recent autopsy study (DM Fernandes et al. J Pediatr 2018; 200: 174-80) examined nonalcoholic fatty liver disease (NAFLD) in a pediatric cohort of 582 (2-19). Approximately 75% were in 14-19 years of age and 50% were black; black pediatric patients (n=290) were over-represented in this sample only 25% of the New York population is black or African American based on the 2010 census.
Key findings:
- Causes of death: 49% homicides, 31% accidents, 10% acute illness, 9% suicide, 1% other
- Overall, NAFLD was present in 4.5%; this low overall prevalence was due in part to the low rate of NAFLD in black children; only 3 of 290 (1%) had NAFLD and none had nonalcoholic steatohepatitis (NASH)
- The rate of NAFLD was 7.9% in hispanics and 8.3% in white patients.
- In this cohort, 36% were overweight or obese. In this subgroup, 14.1% of hispanics and 14.8% of whites had NAFLD.
- Overall, NASH was present in 1.7% of the entire cohort. NASH and fibrosis have been shown in prior studies as the best predictors of disease progression
My take: If black children are not killed by homicide or accidents, it is unlikely that they will die from NAFLD due to its low prevalence.
Related blog posts:
- Pediatric NAFLD: You Don’t Have to be Obesity/Overweight to Have Fatty Liver Disease (but it helps)
- NAFLD Guidance from American Association for the Study of Liver Diseases
- Pediatric NAFLD Guidelines 2017
- Concise Review: Fatty Liver in Pediatrics
- Ultrasound Unreliable to Exclude Fatty Liver
- A liver disease tsunami | gutsandgrowth
- Increasing prevalence of pediatric NAFLD | gutsandgrowth
Is There Good Evidence for Proactive Drug Monitoring of Anti-TNF Therapy?
A recent clinical review (X Roblin et al. Inflamm Bowel Dis 2018; 24: 1904-9) examines the utility of proactive drug monitoring of anti-TNF therapy in inflammatory bowel disease.
The authors note that several observational trials suggested that proactive drug monitoring would help optimize the effect of anti-TNF therapy, especially infliximab. However, two randomized controlled clinical trials, TAXIT (n=273) and TAILORIX (n=122), were not able to show long-term benefit from proactive therapeutic monitoring.
At the same time, the authors note that a recent trial (Ungar B et al. Clin Gastroenterol Hepatol 2016; 14: 550-7, e2) has shown that infliximab trough levels >5 mcg/mL and adalimumab levels >7.2 mcg/mL identified mucosal healing with 85% specificity. Higher cutoffs showed only minimal further increase in mucosal healing rates.
My take: To this point, controlled trials have not shown that proactive drug monitoring of anti-TNF therapy is beneficial; this review explains the design and limitations of these studies. My personal view is that more studies are needed to know if proactive drug monitoring is worthwhile. Proactive drug monitoring may be more useful in children/adolescents than adults due to much greater variation in size and dosing.
A recent commentary on therapeutic drug monitoring (from KT Park Twitter Feed): Therapeutic Targets in IBD
Related blog posts:
- CCFA Conference Notes (Part 1): Preemptive Therapeutic Drug Monitoring Not That Helpful
- AGA 2017 Guidelines on Therapeutic Monitoriing Proactive drug monitoring: “careful and selective use of proactive TDM could be beneficial, but current evidence for its routine use is limited and its overall benefits remain uncertain”
- “Silent” Crohn’s Disease | gutsandgrowth
- Treating to Target | gutsandgrowth
- New Target Drug Levels in Inflammatory Bowel Disease | gutsandgrowth
IBD Shorts -September 2018
S Sridhar et al. Inflamm Bowel Dis 2018; 24: 2086-92. This retrospective pediatric study with 409 patients examined dermatologic manifestations on anti-TNF therapy. 47 (11.4%) had dermatologic findings recorded including 33 with psoriasis, 28 with infections, and 10 with eczema (some had multiple skin findings). The majority were able to continue with current anti-TNF regmimen, including 60% of those with psoriasis.
Related blog posts:
- Pattern of Skin Reactions to Anti-TNF Agents | gutsandgrowth
- Immune-Mediated Reactions to Anti-TNFs and What to Do About Them | gutsandgrowth
- Characteristics of Skin Lesions Associated with Anti-Tumor Necrosis Factor Therapy | gutsandgrowth
- IBD Shorts -Skin, Adalimumab Kinetics | gutsandgrowth
NA Rozette et al. Inflamm Bowel Dis 2018; 24: 2007-14. This study with 50 subjects showed good safety of rapid versus standard infliximab infusions. One interesting aspect of their study which included a retrospective arm (standard infusion) and a prospective arm (rapid infusion) was a declining use of premedication, though even in their prospective group 60% received premedication including the combination of acetaminophen, benadryl, and methylprednisolone in 30%. There were two patients in the rapid infusion with mild reactions who reverted to standard infusion rates.
CJ Moran, JL Kaplan, HS Winter. Inflamm Bowel Dis 2018; 24: 2048-52. This study with 199 subjects with active Crohn’s disease (CD) (21-86 years) –had their BioBank blood tested for 5 common CRP genetic variants. Some specific variants, rs2794520TT & rs1800947, were associated with lower CRP levels. This study helps explain why CRP is not a useful marker in some patients with CD.
More Sensitive Detection of Gluten Free Diet Adherence
A recent cross-sectional study (K Gerasimidis et al. JPGN 2018; 67: 356-60) examined the use of fecal gluten immunogenic peptide (GIP) to assess for adherence with gluten free diet (GFD) in biopsy-proven celiac disease (CD).
GIP reflects recent gluten consumption. There is a commercially-available kit available (Ivydal GIP Testing) –though I am uncertain about how its reliability compares to the GIP measured in this study.
In the study, the authors note that GIP positivity can occur with as little as 100 mg of gluten/day ingestion. GIP is a 33-mer peptide from α2-gliadin that is stable against breakdown by gastric, pancreatic, and intestinal brush border enzymes.
Key findings of this study:
- GIP was detectable in 16% of patients with previous CD diagnosis (N=67)
- GIP was detectable in 95% of newly-diagnosed CD patients (n=19) and was detectable in 27% at 1 year afterwards.
- When compared with traditional indicators of GFD adherence (eg. TTG levels, Biagi score, clinical assessment), 4 out of 5 children with detectable GIP were missed
My take: Fecal GIP for celiac disease adherence has similar potential as a biomarker as calprotectin has for IBD. A normal GIP appears to be much more sensitive at detecting gluten ingestion.
Related blog posts:
- Followup biopsies in Pediatric Celiac disease?
- Are followup biopsies necessary for Celiac disease? Look beyond the headlines
- Closer followup for Celiac disease & pediatric guidelines
- How Accurate is Serology at Predicting Mucosal Healing in Pediatric Celiac Disease?
- Expert review: Celiac disease
- Never Too Old for Celiac Disease
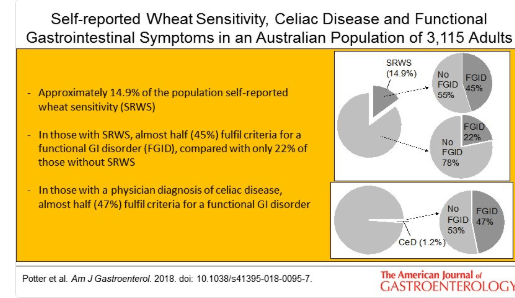
Wheat Intolerance –Self-Reported in 15%!
A recent study (MDE Potter et al. Am J Gastroenterol 2018; 113: 1036-44 -thanks to Ben Gold for this reference) examined the frequency of wheat intolerance and chronic gastrointestinal symptoms in a randomly selected population of 3542 in Autstralia via a mail survey.
Key findings:
- Self-reported wheat sensitivity was 14.9%
- Prevalence of celiac disease (CD) was 1.2%
- A doctor-diagnosis of CD was associated with functional dyspepsia with an odds ratio (OR) of 3.35.
- Self-reported wheat sensitivity was independently associated with irritable bowel syndrome with an OR of 3.55 and almost half (45%) have an underlying functional GI disorder.
In a related editorial (pgs 945-8), Imran Aziz makes several useful points:
- Gluten-free industry has boomed in U.S. with retail sales going from $0.9 billion in 2006 to ~S24 billion in 2020.
- While previous studies have shown that gluten can induce symptoms in the absence of CD (Biesiekierski JR et al. Am J Gastroenterol 2011; 106: 508-14), more recent rigorous studies have indicated that “gluten-per-se accounts for 1-in-6 cases with the remaining majority either due to fructans (a type of FODMAP or a nocebo effect.”
- There are no accurate biomarkers of wheat intolerance
- Dr. Aziz also cautions against adopting a gluten-free diet without proper counseling. “The greatest concern is whether these diets are safe in the long-run, given the emerging data suggesting cardiovascular, nutritional, metabolic, and microbial changes.”
My take: This study shows that about 1 in 10 individuals have self-reported wheat intolerance; gluten, though, is the actual culprit in less than 20%.
Related Blog Posts:
- Wheat Intolerance Syndrome?
- What to make of FODMAPs | gutsandgrowth
- An Unexpected Twist for “Gluten Sensitivity” | gutsandgrowth
- Food choices, FODMAPs, and gluten haters | gutsandgrowth
- UVA Links | gutsandgrowth
- Mechanism for FODMAPs diet
- Low FODMAPs Diet in Pediatric Irritable Bowel | gutsandgrowth