RS Dalal et al. Inflammatory Bowel Diseases, Volume 30, Issue 3, March 2024, Pages 395–401. 1-Year Comparative Effectiveness of Tofacitinib vs Ustekinumab for Patients With Ulcerative Colitis and Prior Antitumor Necrosis Factor Failure
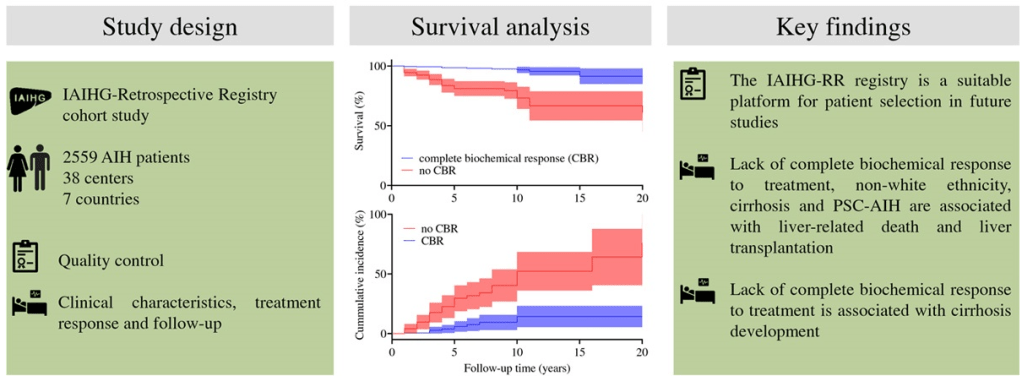
In this real-world cohort of anti-TNF-exposed patients with ulcerative colitis, tofacitinib (n=69) and ustekinumab (n=97) demonstrated similar effectiveness in achieving steroid-free clinical remission (SCFR) at 12 and 52 weeks. The median follow-up was 88.0 and 62.0 week, respectively. 35 of 66 in the tofacitinib cohort had dose reduction from the starting dose of 10 mg twice daily. This reduction occurred at a mean of 144 days. 59 of 97 in the ustekinumab cohort received either Q4W dosing (n=43) or Q6W (n=16).
Key findings:
- 53% of patients receiving tofacitinib and 32% of patients receiving ustekinumab achieved SFCR at 12 weeks. Tofacitinib-treated patients had higher baseline Mayo endoscopic subscores and CRPs.
- At 52 weeks, approximately 50% of patients in both treatment groups achieved SFCR. There were also high proportions (>60%) of patients in both treatment groups who had endoscopic response within 52 weeks.
- Both drugs were well-tolerated, as only 1 patient in each treatment group discontinued therapy due to an AE during >260 patient-years of follow-up.

My take: This shows similar response to either tofacitinib and ustekinumab in a cohort that had refractory disease as patients were anti-TNF failures and most had prior vedolizumab as well.
Related blog posts:
- Comparative Efficacy: Infliximab vs. Ustekinumab
- When is the Right Time to De-escalate Dose of Tofacitinib for Ulcerative Colitis?
- Head-to-Head (Sort of): Infliximab vs Ustekinumab for Crohn’s Disease
- Ustekinumab vs Adalimumab: Head-to-Head Study
- How Much Ustekinumab (Stelara) Is Needed to Get a Good Response
- IBD Updates: Treat-to-Target Uptake, Long-Term Data on Ustekinumab Intensification, and Low Rates of C diff with Tofacitinib (& Clinical Pearl)