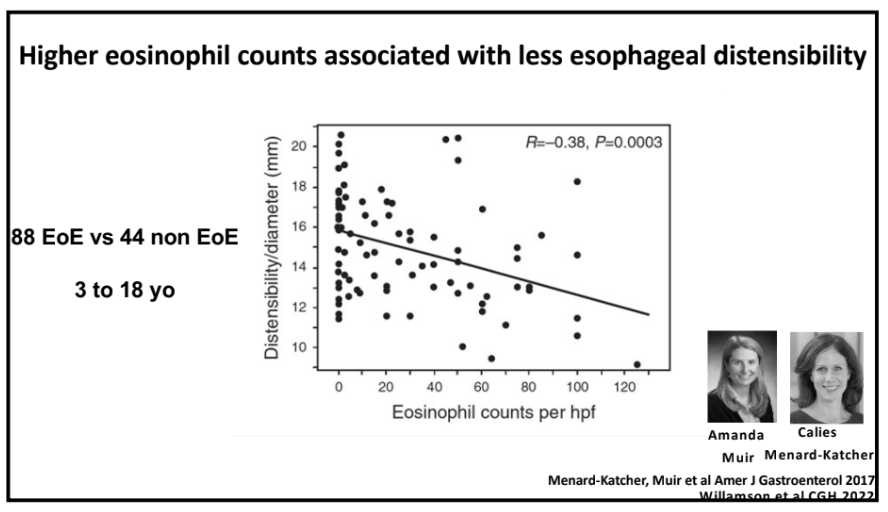
D Geem et al (Senior author: Subra Kugasthasan). Clin Gastroenterol Hepatol 2024; 22: 368-376. Progression of Pediatric Crohn’s Disease Is Associated With Anti–Tumor Necrosis Factor Timing and Body Mass Index Z-Score Normalization
Congratulations to my colleagues at Emory who led/participated in this study.
This study examined 5-year longitudinal data from the pediatric multicenter RISK cohort (n=1075). RISK=risk stratification and identification of immunogenetic and microbial markers of rapid disease progression in children
Key findings:
- For children with a low BMIz at diagnosis (n = 294), BMIz normalization within 6 months of diagnosis were associated with a decreased risk for surgery (HR 0.47). Patients without BMIz normalization were enriched for genes in cytokine production and inflammation.
- Unsurprisingly, baseline B2 (stricturing disease) and B2+B3 (stricturing and penetrating disease) were associated with increased risk of surgery with HR, 4.20 and HR, 8.24 respectively
- Earlier anti-TNF therapy was associated with a lower hazard rate (HR) of needing surgery
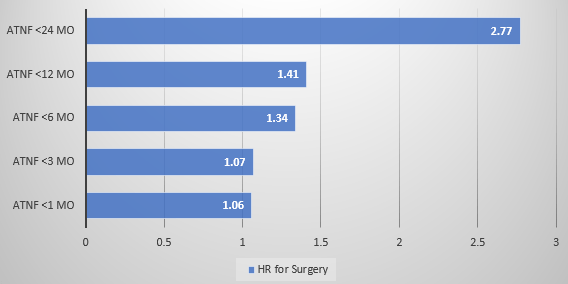
My take: It appears that early anti-TNF therapy lowers the risk of surgery. Improved BMI with treatment is another good prognostic variable. There may be an early window in which effective treatment prevents long-term damage to the GI tract in pediatric patients with Crohn’s disease.
This study has overlapping findings (also with RISK cohort) by Adler et al showing early treatment preventing perianal fistulas. Blog post: Early Treatment Can Prevent Fistulas in Pediatric Crohn’s Disease
Related article: JC McCurdy et al. Clin Gastroenterol Hepatol 2024; 22: 377-385. Open Access! Comparative Effectiveness of Biologic Therapies in Preventing Penetrating Complications in Patients With Crohn’s Disease
In this observational retrospective study with 40,693 patients: 93% anti-TNF, 3% UST (ustekinumab), and 4% VDZ (vedolizumab), “Anti-TNF therapy was associated with a lower risk of LPD and PPD [luminal and perianal penetrating disease] compared with VDZ, and lower risk of LPD compared with UST.”
Related blog posts: