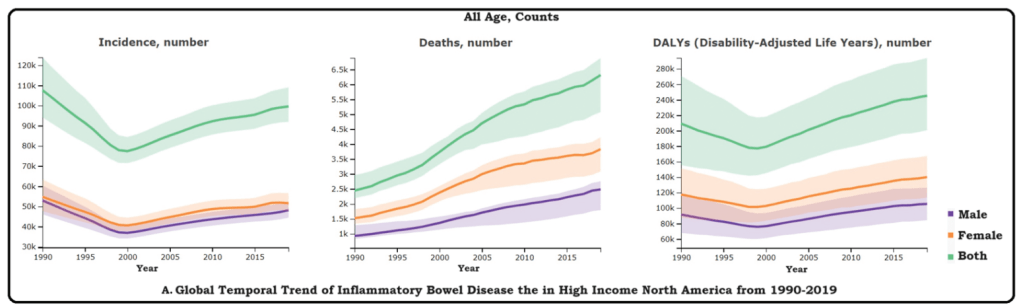
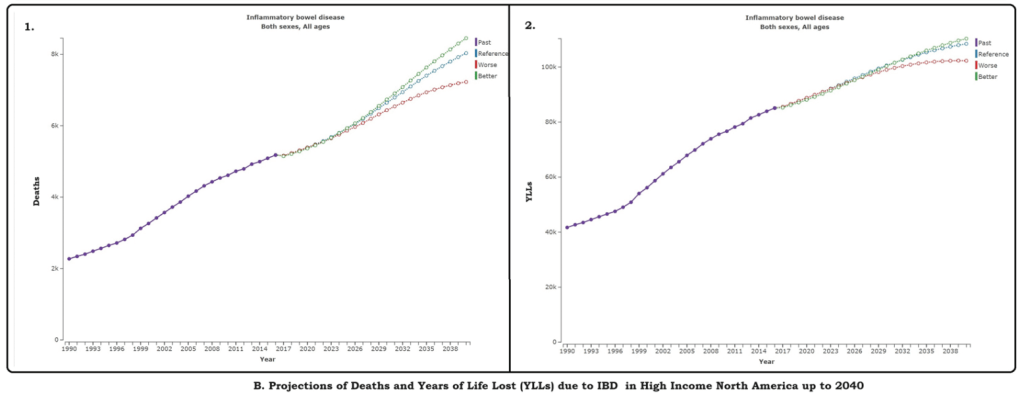
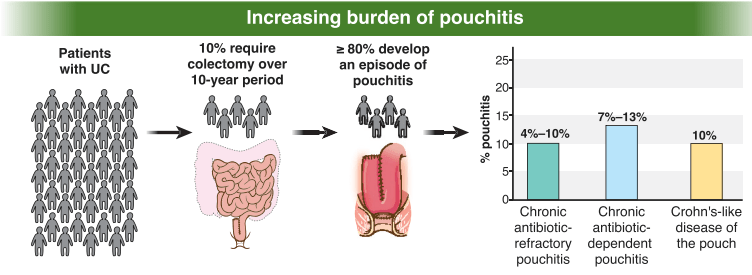
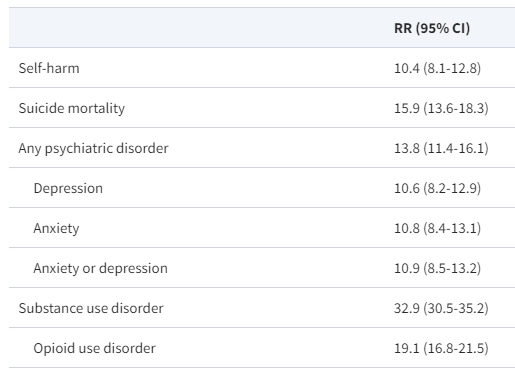
AAP News 2/13/24: FDA approves first oral therapy for EoE
Eohilia is given in doses of 2 milligrams twice a day for 12 weeks. The label notes it has not been shown to be safe and effective for longer.
The FDA accepted Takeda’s new drug application in December 2020. A year later, the FDA determined more study was needed. Takeda revised its application and resubmitted it in September 2023.
Takeda conducted two multicenter, randomized, placebo-controlled trials in patients 11 to 56 years and 11 to 42 years, respectively. The first found 53% of the treatment group achieved histologic remission compared to 1% receiving a placebo. The second found 38% of the treatment group achieved remission vs. 2% of the placebo group, according to Takeda…
Patients [need] to refrain from eating or drinking for at least 30 minutes after taking Eohilia. After 30 minutes, patients should rinse their mouth and spit to reduce the risk of developing thrush.
My take: Budesonide has been used effectively for EoE for a long time; it is good news that it is recognized by FDA with a specific EoE indication. However, it is a little concerning that the label indication is for 12 weeks when we know that this is chronic disease. Also, I am eager to see how much this formulation costs in comparison to the budesonide ampules.
Related blog posts:
- Briefly noted: Aerodigestive Medicine and Budesonide for Eosinophilic Esophagitis
- Eosinophilic Esophagitis -Increasing Incidence and Emergence of Biologic Treatments
- NASPGHAN YouTube Video for Eosinophilic Esophagitis
- Long-Term Treatment of Eosinophilic Esophagitis with Budesonide
- Phase 3 Trial of Budesonide for Eosinophilic Esophagitis & COVID-19 Deaths in U.S.
- Head-to-Head: Budesonide vs Fluticasone for Eosinophilic Esophagitis
- Orodispensable Budesonide Tablets for Eosinophilic Esophagitis
- Surprising Findings in Prospective Budesonide-Eosinophilic Esophagitis Study
- Practical Guide to Dietary Therapy for Eosinophilic Esophagitis
- When to Use Dupilumab for Eosinophilic Esophagitis: Multispecialty Guidelines
- Managing Adrenal Insufficiency in Eosinophilic Esophagitis