K Mullin, RM Rentea, M Appleby, PT Reeves. Pediatrics in Review 2024; 45: 210-224. Gastrointestinal Ostomies in Children: A Primer for the Pediatrician
Like yesterday’s article on GTs, this is another terrific review with plenty of helpful images and advice regarding ostomy management.
- Background: “There are upwards of 1 million people living with ostomies (ostomates) in the United States.” “Approximately 75% of all ostomies in the pediatric population are created in the neonatal or infant patient.”
- Table 1 lists the purposes and types of ostomies including gastrostomy, jejunostomy, ileostomy, appendicostomy (Malone), cecostomy, colostomy and urinary diversions (eg. Mitrofanoff).
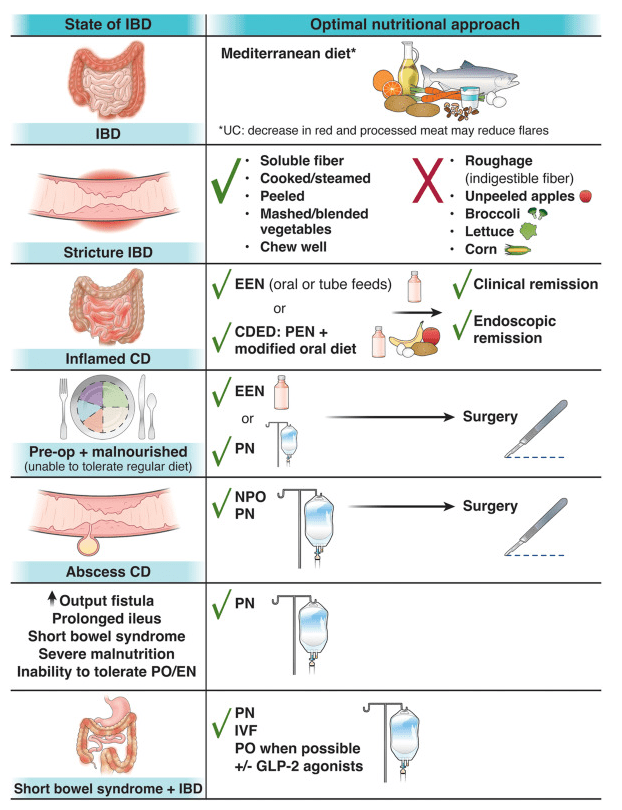
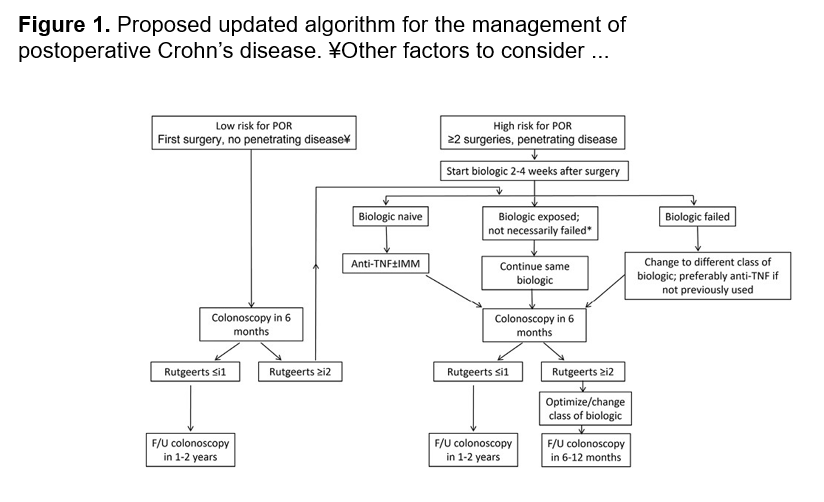
- Surgical considerations are reviewed including optimizing nutrition preoperatively and minimizing corticosteroids. Biologics: “The most recent evidence does not support a delay in gastrointestinal surgery for children with IBD receiving biological therapy…[and] typically, biological therapy can be resumed 14 to 28 days after the operation.” For oral small molecules (with short half-lives), these may be restarted sooner if indicated.
- Table 2 provides pictures of the lower ostomies. For example:


- Postoperative care is discussed including healing times, need for wound ostomy nurse input, and addressing self-image. Patients with motility disorders are “more likely to experience postoperative complications”
- Table 4 details the products for pouch care including pouching systems, skin barriers, pouch liner, gas vent, pouch lubricant, pouch covers, and adhesive remover.
- Table 5 summarized ostomy-related complications and treatments. Complications include stomal necrosis, stomal bleeding, stomal retraction, mucocutaneous separation, parastomal hernia, stoma prolapse (can apply cool compresses, apply osmotic agent (sugar) or manually reduce), stoma stenosis, and dermatitis.
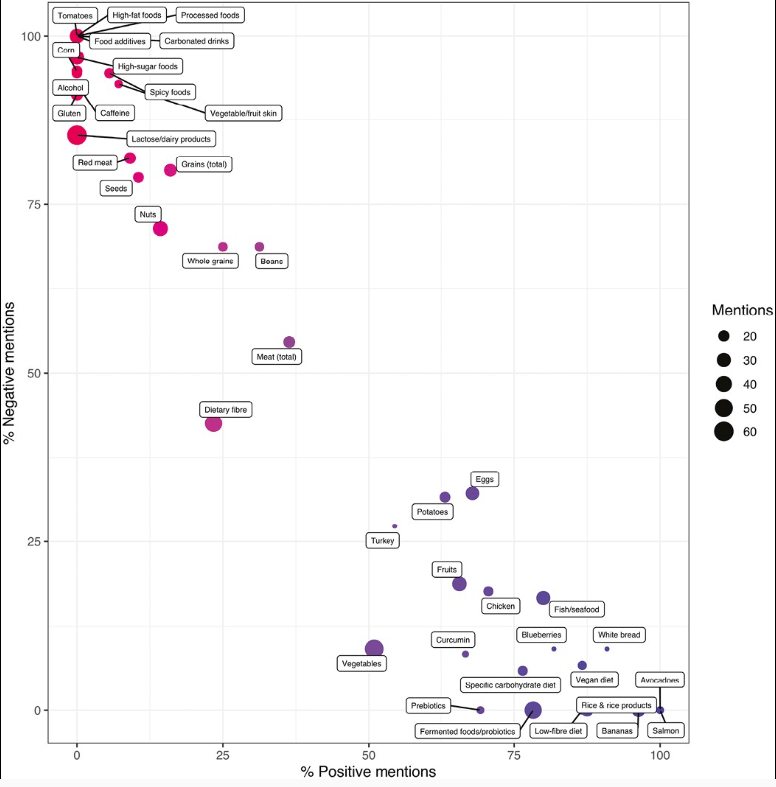
- Table 6 addresses medical management issues like odor, blockage, diarrhea, and constipation. This table also provides recipes for antegrade enemas (see below) and links including a very useful bowel management guide for families (28 pg from Boston Children’s) and enema ingredients and supplies (2 pg from Seattle Children’s); the latter has some overlapping information with the former.

- At the conclusion of the article, there is further discussion of systemic and ostomy-related complications (much of which is summarized in Table 5). The article references the Ostomy Skin Tool as a metric to follow the clinical state of the ostomy. The United Ostomy Associations of America (ostomy.org) is listed as a good resource (which it is!).
My take: This is a very useful resource. Even a quick read will make clinicians appreciative of having the assistance wound ostomy nurses.
Related blog posts:
- Management of Ostomies
- Practical Intestinal Rehabilitation (Part 1)
- Practical Intestinal Rehabilitation (Part 2)
Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.