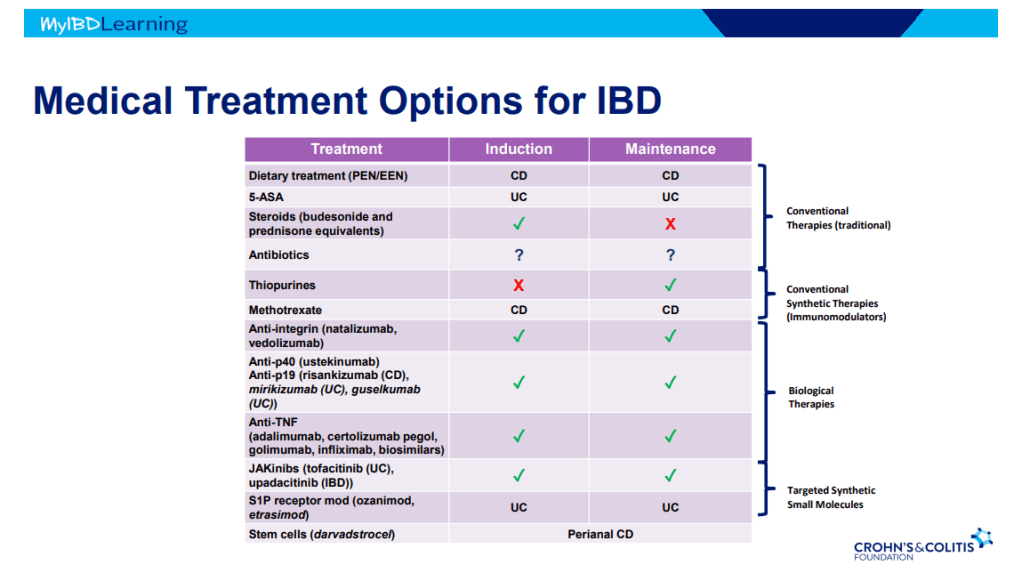
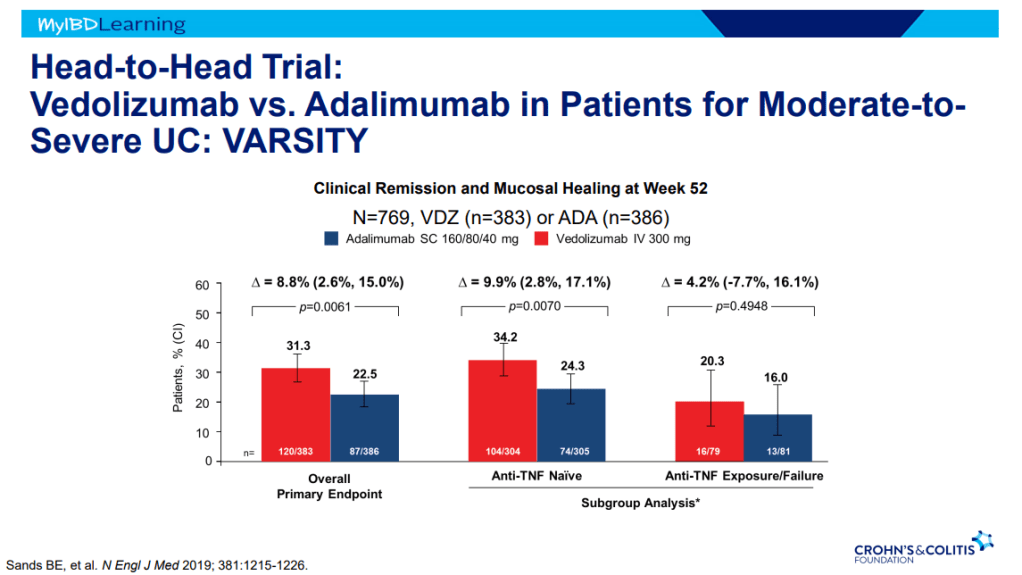
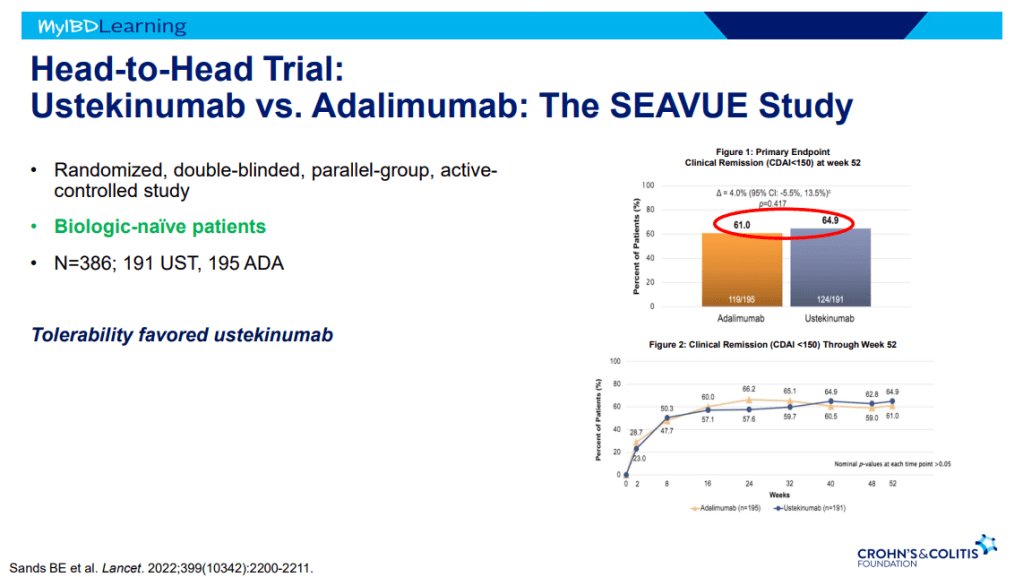
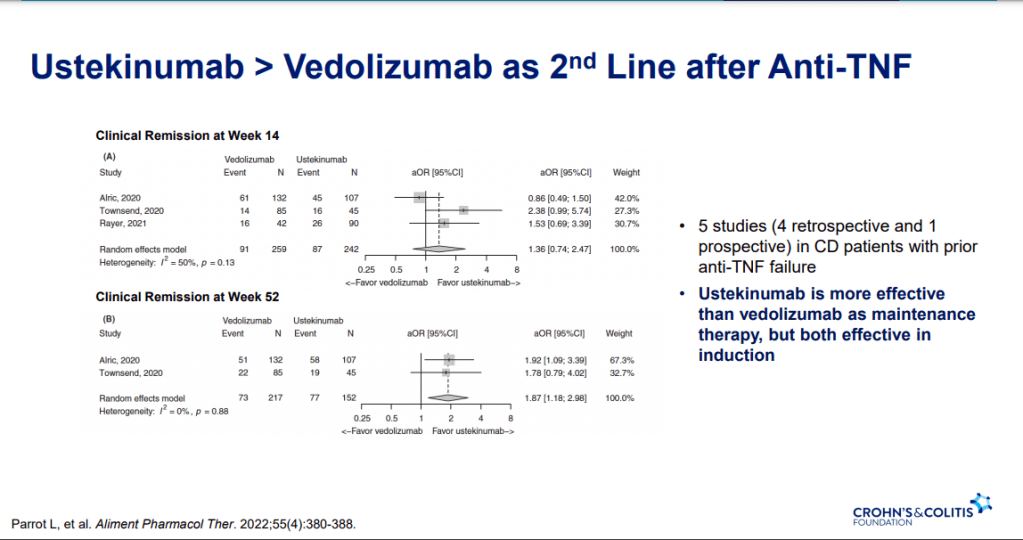
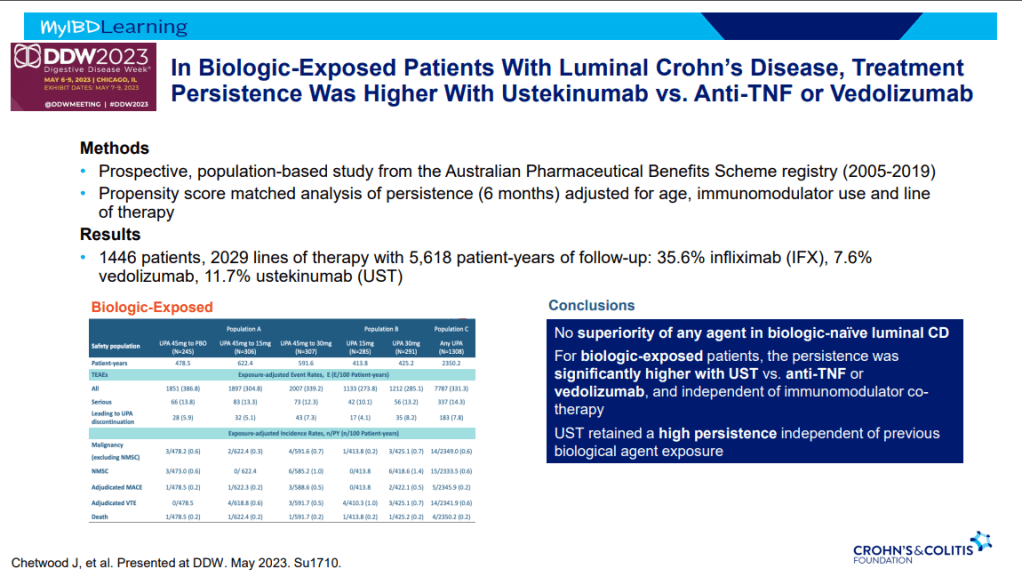
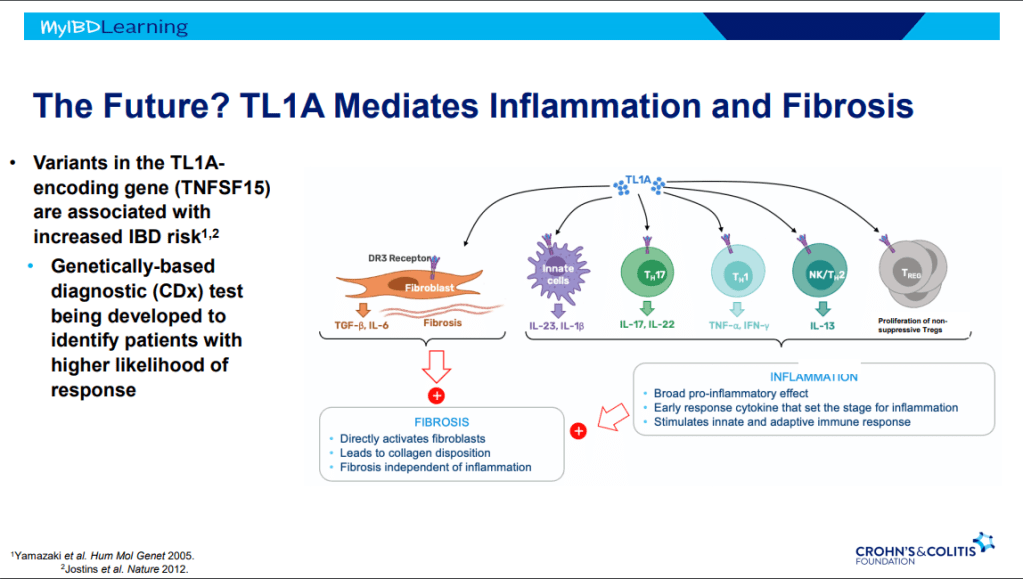
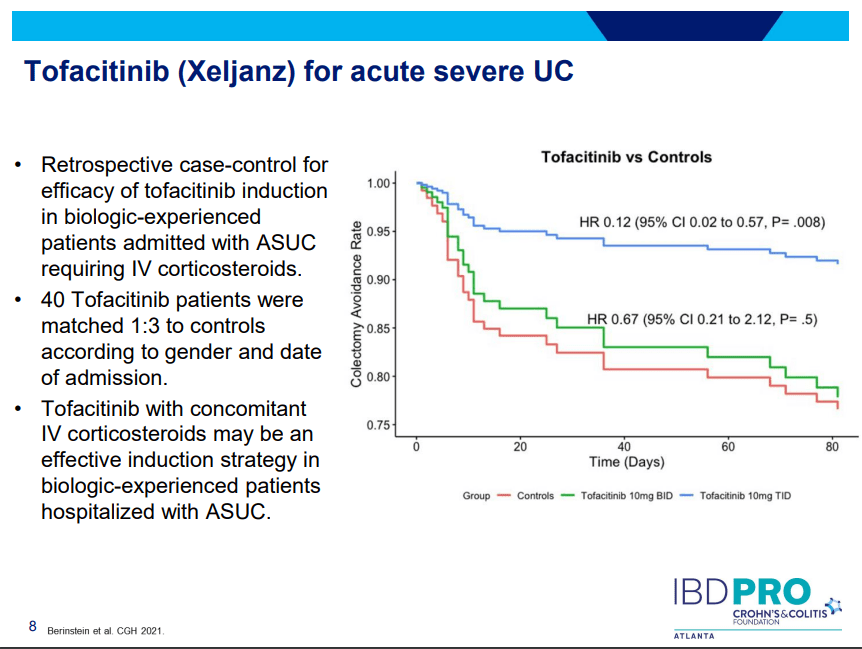
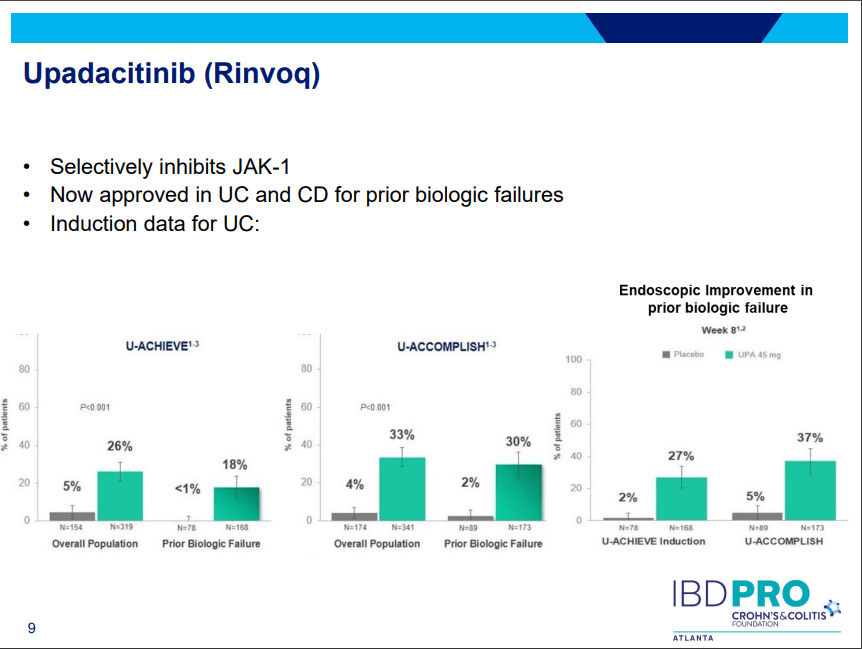
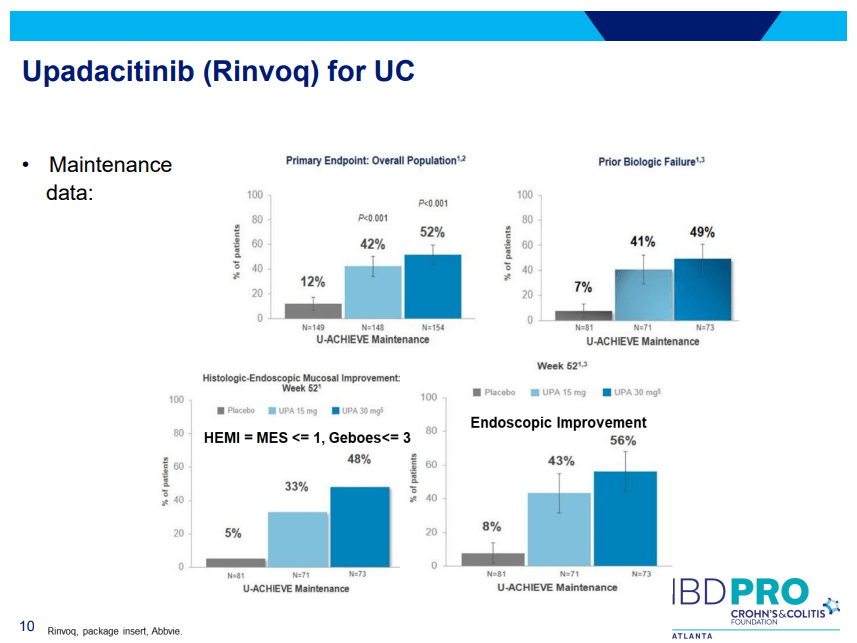
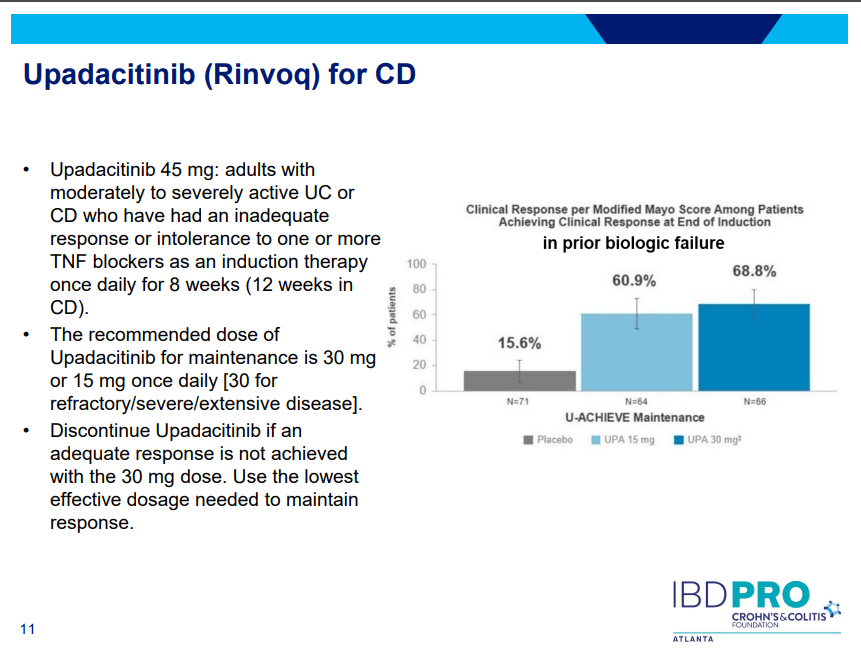
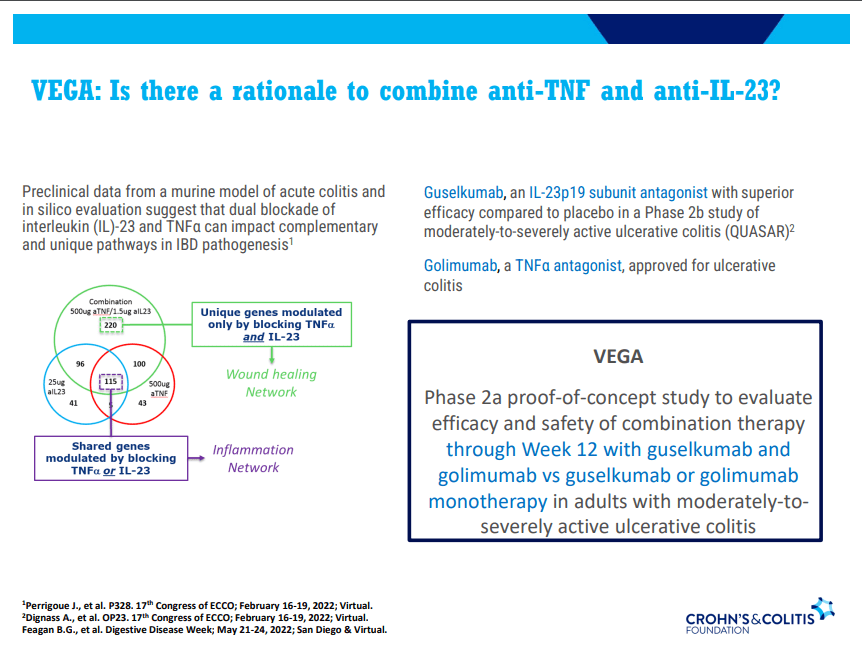
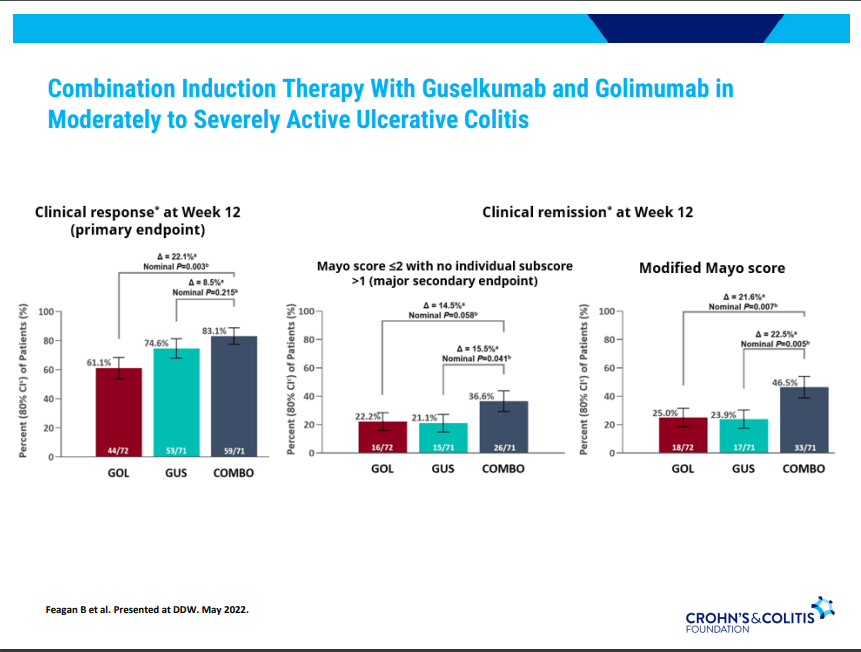
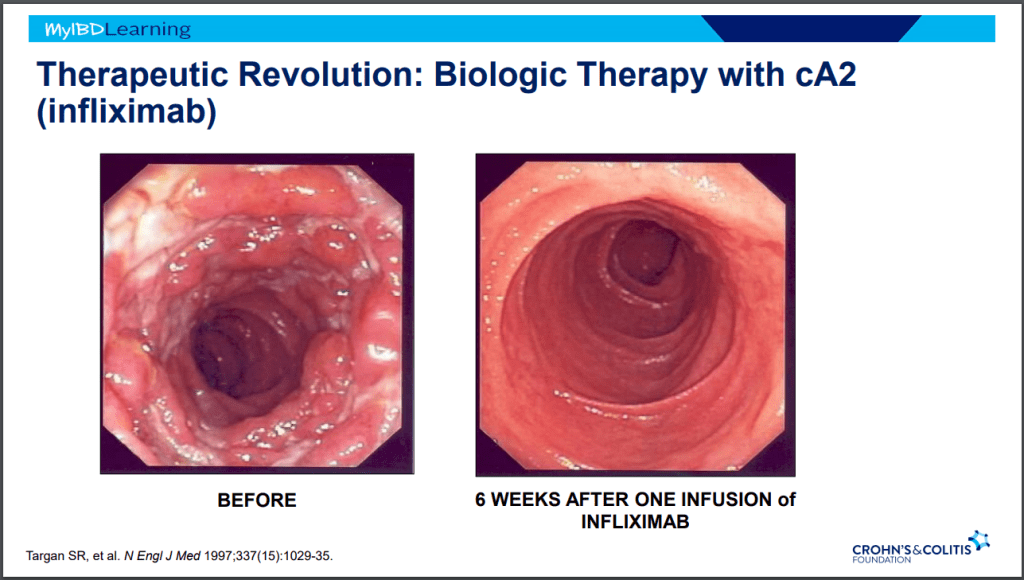
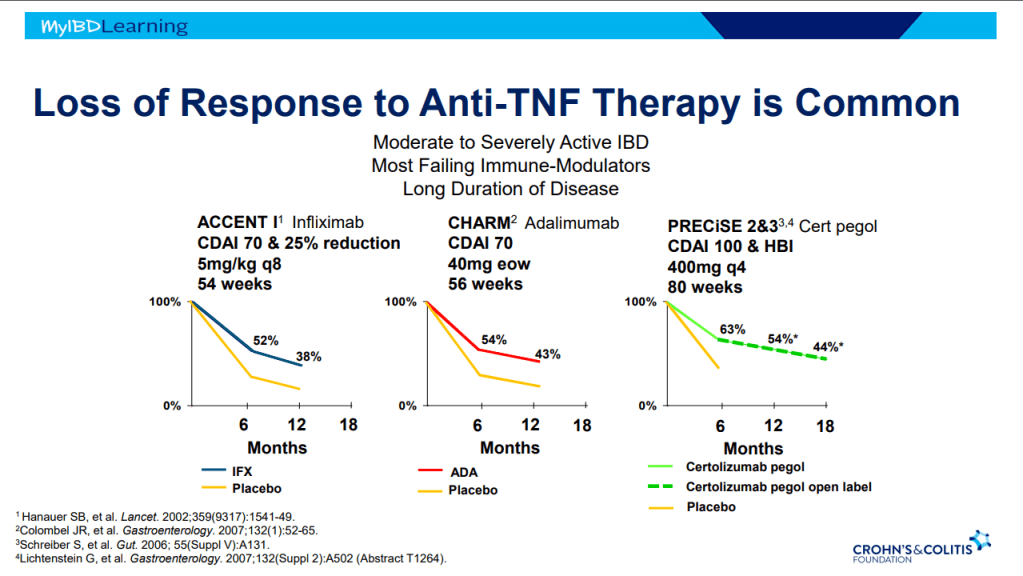
S Pandurangi et al. J Pediatr Surg 2023; 58: 1483-1488. Customized Postoperative Therapy Improves Bile Drainage in Biliary Atresia: A Single Center Preliminary Report
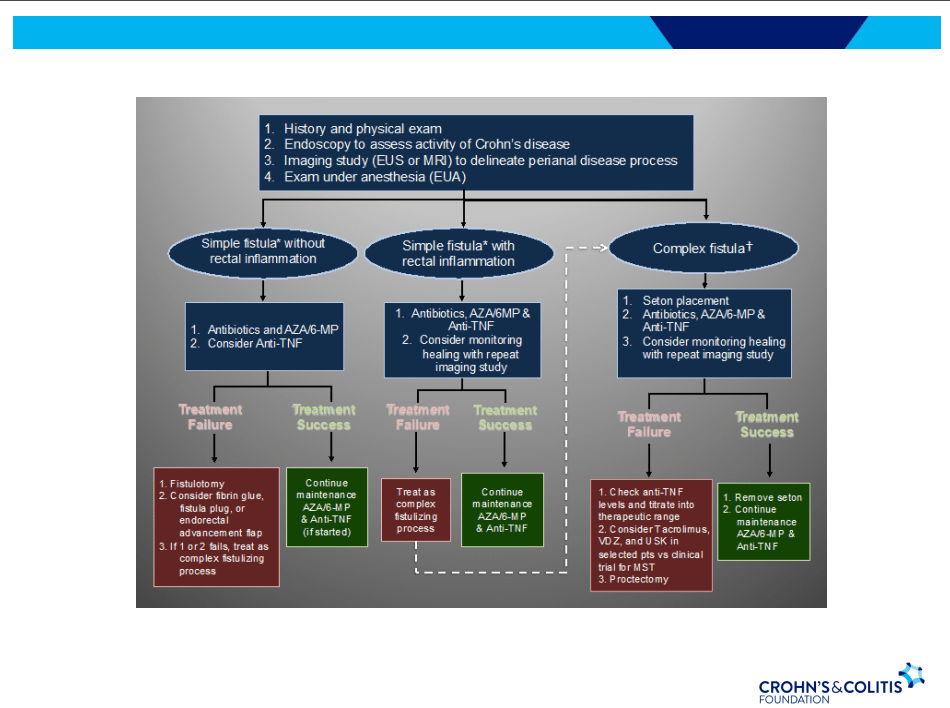
This single center retrospective study compared 20 consecutive infants underwent hepatoportoenterostomy (HPE) (beginning in 2017) for biliary atresia (BA) to a historical cohort. Analysis of successful biliary drainage 3 months after HPE (defined as serum total bilirubin (TB) <2 mg/dL) was the primary endpoint; survival with native liver at 2 years was the secondary endpoint.
Protocol:
- Cefoxitin was administered to all infants following HPE for 3-4 days.
- Standard protocol: If the stool color normalized (pigmented), the infant received “conventional” treatment with trimethoprim-sulfamethoxazole cholangitis prophylaxis, fat-soluble vitamin supplementation with DEKAsPlus or AquaADEKs (1 mL daily), and ursodeoxycholic acid (5 mg/kg twice daily).
- Customized protocol: If the stools were acholic (or not consistently pigmented) and </=45 days, the infants received intravenous cefoxitin or piperacillin-tazobactam and methylprednisolone, initial dose 5 mg/kg/day and decreased by 1 mg/kg/day each day thru day 5; then orally treated with dose dropped 0.25 mg/kg weekly. When switched to oral steroids, IV antibiotics were stopped and infant was placed on amoxicillin-clavulanate which was continued until TB <2 mg/dL or discontinuation of corticosteroids (whichever came first).
- If stools were acholic and infant was >45 days, then the same treatment was given if there was liver inflammation on histology.
Key findings:
- 8 had pigmented stools after HPE and received standard protocol.
- 12 had acholic/inconsistent stools. All of those >45 days had liver inflammation; thus, all 12 received the customized protocol. Two infants had two cycles of steroids/antibiotics who had initial response to treatment and then worsened.
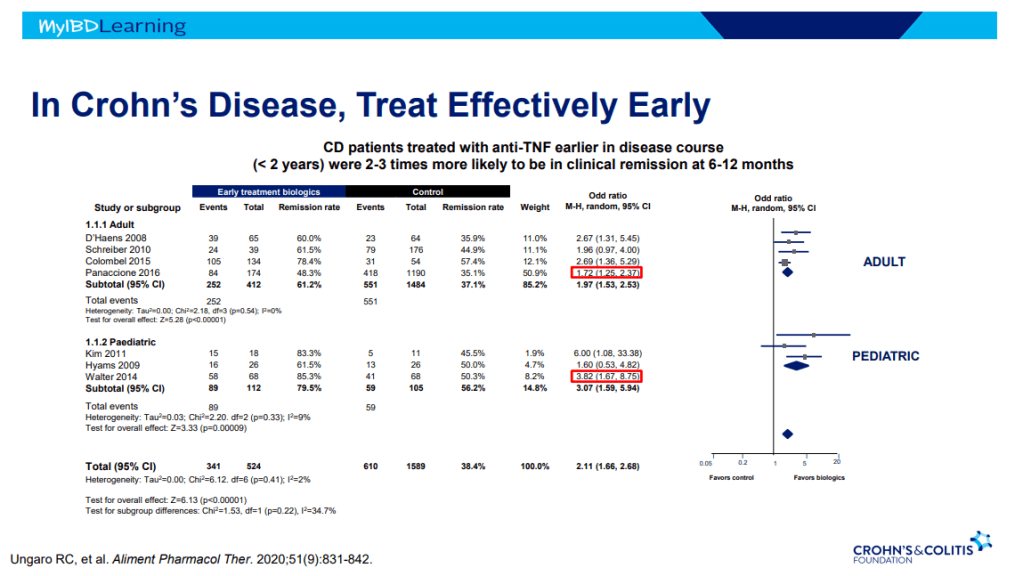
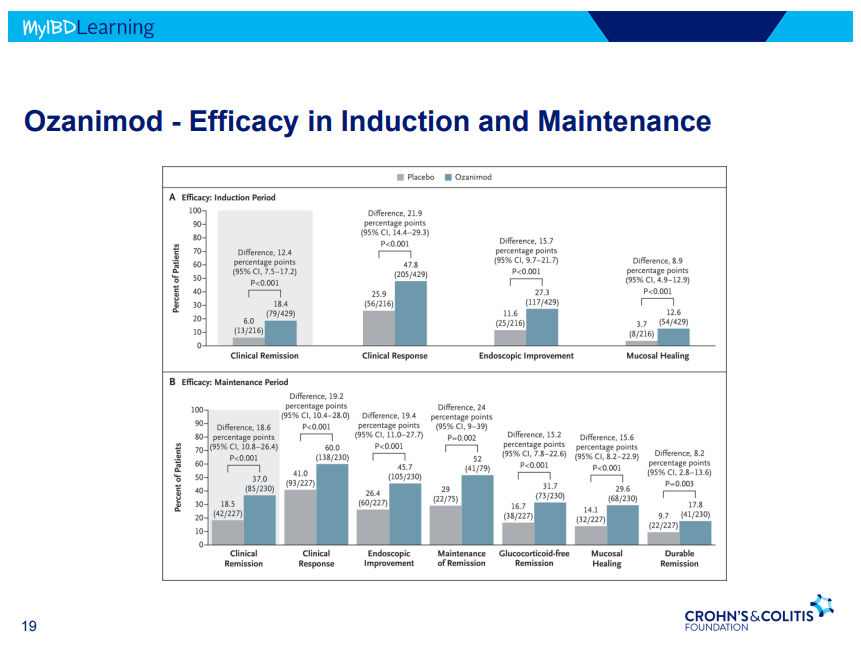
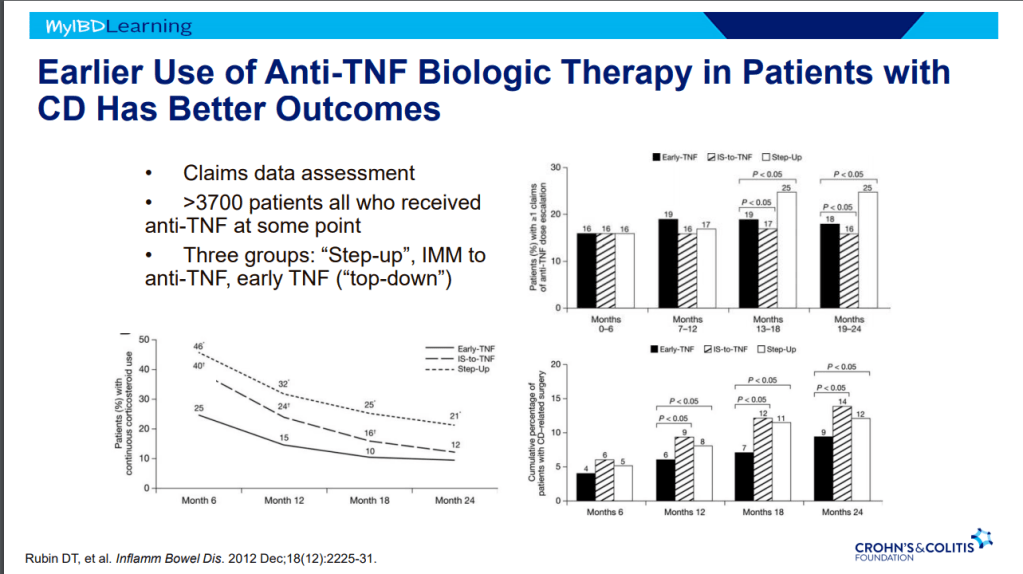
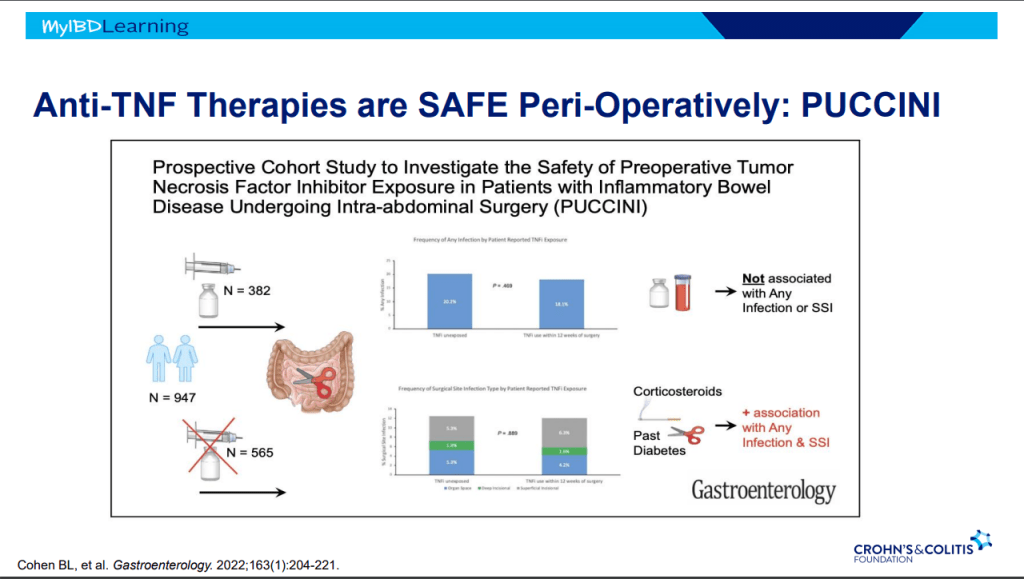
- Sixteen of 20 (80%) infants had successful bile drainage, compared to 8 of 20 (40%) infants in the historical cohort (P = 0.0225)
- Among the sixteen who have reached two years of age, 11 (68.8%) are alive with native livers versus 10 of 20 (50%) in the historical cohort (P = 0.0970). This did not achieve statistical significance.
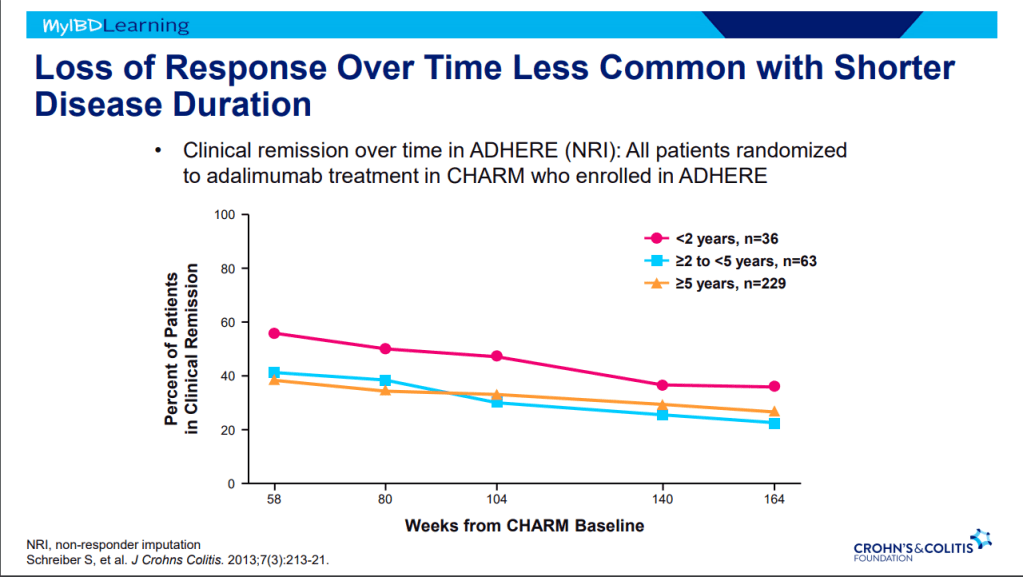
The authors established their protocol based on data from Kings College in 2016 suggesting that steroids appeared effective in younger patients who underwent HPE prior to 45 days (Peg Surg Int 2016; 32: 193-200). The START study showed no significant improvement in biliary drainage between patients receiving corticosteroids and placebo. However, in the group <70 days, 72% of infants receiving corticosteroids achieved biliary drainage compared with 57% of the placebo group (P=0.36).
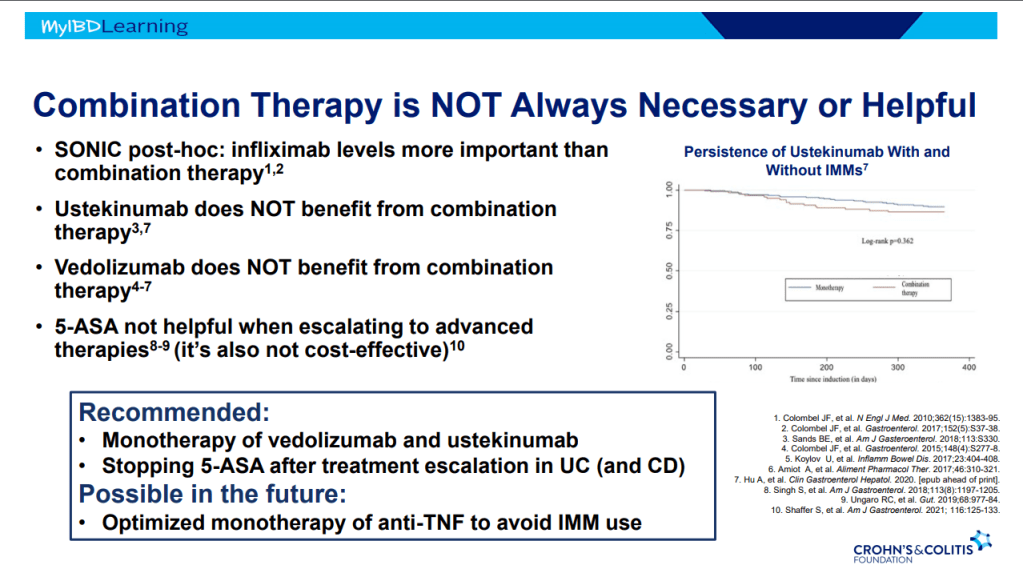
My take: This is a small sample size. Perhaps, this protocol will help improve outcomes. If so, we still don’t know which factor is more important —the IV antibiotics or the high dose steroids. If these agents are helpful, are there other predictive factors –microbiome? MMP-&?
Related blog posts:
- Cholangitis After Kasai Procedure for Biliary Atresia
- START Study: Steroids Not Effective For Biliary Atresia (After Kasai) In this study, the steroid intervention did not affect transplant-free survival which was 58.7% in the steroid group and 59.4% in the placebo group at 24 months of age. In addition, steroids were associated with an earlier onset of first serious adverse events.
- Bad News Bili
Pictures from the Villa Ephrussi de Rothschild