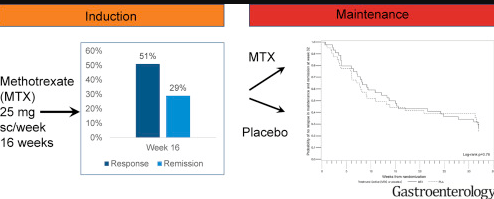
Button batteries –definitely more scary than Halloween. Here is a link to new guidelines from Poison Control: New Button Battery Guidelines 2018
A recent report from Nationwide Childrens, How Sweet It Is: Honey Attenuates Button Battery-Induced Esophageal Damage, highlights two important advances in button battery management that are now incorporated into the new button battery guidelines:
- At time of endoscopy, “a weak acetic acid rinse (sterile vinegar) can help neutralize tissue pH and protect the esophagus from continued tissue breakdown after battery removal. This irrigation concept has now been successfully used in children around the world with good clinical outcomes.”
- Prior to endoscopy, “both honey and sucralfate (Carafate®) were able to effectively neutralize the tissue pH and reduce visible injury.”
“More than 3,000 cases occur per year, mostly among children younger than age 6, and severe cases are on the rise. Lodged button batteries can cause rapid injury, including permanent bilateral vocal cord paralysis and even death.”
Guideline recommendations with regard to acetic acid:
After a battery is removed from the esophagus, inspect the area endoscopically for evidence of perforation. If none is evident, irrigate the injured areas with 50 mL to 150 mL of 0.25% sterile acetic acid (obtained from the hospital pharmacy). Irrigate in increments and suction away excess fluid and debris through the endoscope. For decades toxicologists have advised against neutralization for fear of causing a thermal injury. However, a recent study (Jatana, 2016) using piglet esophagus preparations after button battery removal, showed only a minimal increase in temperature (0-3 oC), effective tissue surface pH neutralization, and decrease in the visible injury using this neutralization strategy. The tissue surface pH neutralization may reduce the development of progressive, delayed-onset esophageal injury after battery removal.
From guideline with regard to honey:
Administer honey immediately and while en route to the ER, if:
- A lithium coin cell may have been ingested (if you don’t know what kind of button battery was swallowed, assume it is a lithium coin cell unless it is a hearing aid battery);
- The child is 12 months of age or older (because honey is not safe in children younger than one year);
- The battery was swallowed within the prior 12 hours (because the risk that esophageal perforation is already present increases after 12 hours);
- The child is able to swallow; and
- Honey is immediately available.
How to dose honey:
-
-
- Give 10 mL (2 teaspoons) of honey by mouth every 10 minutes for up to 6 doses. Do not worry about the exact dose or timing.
- Use commercial honey if available, rather than specialized or artisanal honey (to avoid inadvertent use of large amounts of honey produced from potentially toxic flowers).
- Honey is NOT a substitute for immediate removal of a battery lodged in the esophagus. Honey slows the development of battery injury but won’t stop it from occurring. Do not delay going to an ER.
Why give honey?
Honey is administered to coat the battery and prevent local generation of hydroxide, thereby delaying alkaline burns to adjacent tissue. Efficacy is based on a 2018 study (Anfang et al) assessing the in vitro protective effects of various liquids in the cadaveric porcine esophagus and in vivoprotective effects of honey and sucralfate (Carafate®) compared to saline irrigations of batteries placed in the esophagus of live piglets. Both honey and sucralfate (Carafate®) effectively prevented the expected battery-induced pH increase and decreased the depth of the resulting esophageal injury.
References:
- Anfang RR, Jatana KR, Linn RL, Rhoades K, Fry J, Jacobs IN. pH-neutralizing esophageal irrigations as a novel mitigation strategy for button battery injury. The Laryngoscope. 2018 Jun 11. [Epub ahead of print]
- Jatana KR, Rhoades K, Milkovich S, Jacobs IN. Basic mechanism of button battery ingestion injuries and novel mitigation strategies after diagnosis and removal. The Laryngoscope. 2017 Jun;127(6):1276-1282.
Related blog posts:

Disclaimer: These blog posts are for educational purposes only. Specific dosing of medications/diets (along with potential adverse effects) should be confirmed by prescribing physician/nutritionist. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.