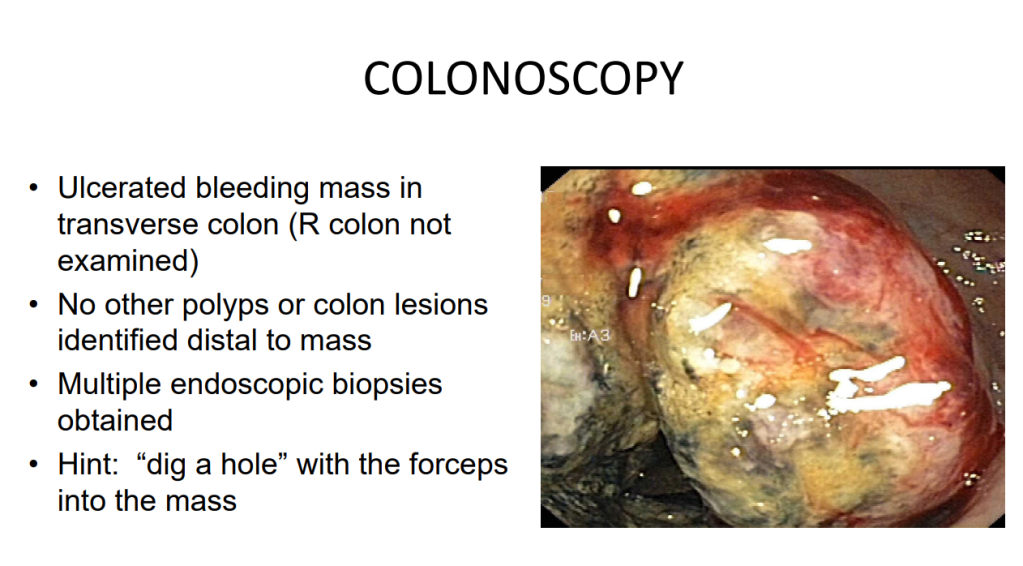
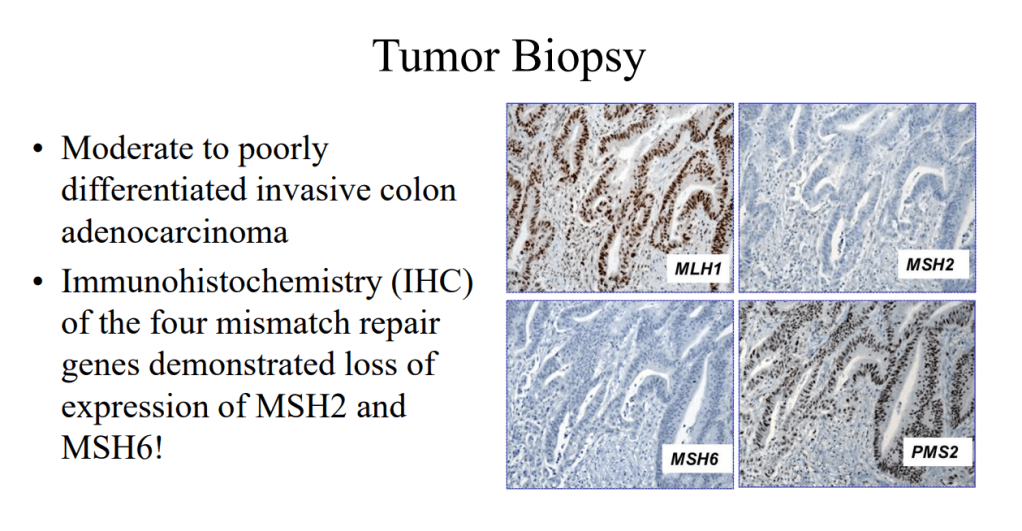
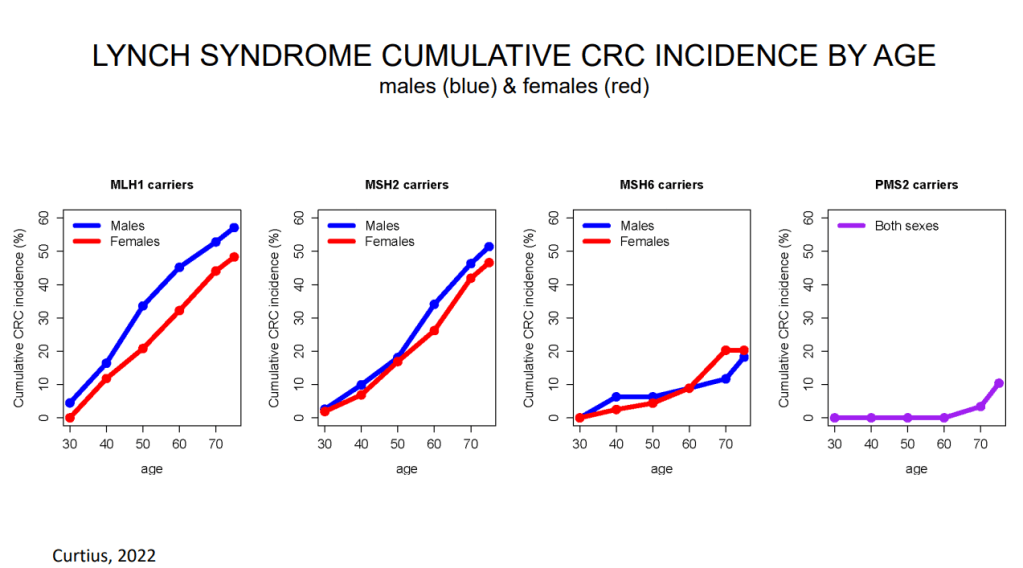
K Van de Houte et al. Gastroenterol 2024; 167: 333-342. Open Access! Efficacy and Findings of a Blinded Randomized Reintroduction Phase for the Low FODMAP Diet in Irritable Bowel Syndrome
Methods: Responders (n=94 of 117) to a 6-week low FODMAP diet, defined by a drop in IBS symptom severity score (IBS-SSS) compared with baseline, entered a 9-week blinded randomized reintroduction phase with 6 FODMAP powders (fructans, fructose, galacto-oligosaccharides, lactose, mannitol, sorbitol) or control (glucose). A rise in IBS-SSS (≥50 points) defined a FODMAP trigger. Patients were challenged with 6 FODMAPs or glucose as a control (3/day x 7 days) while continuing with the low FODMAP diet. At the end of the seventh day, patients entered 2 days of washout before starting with the next blinded FODMAP or control powder.
Key findings:
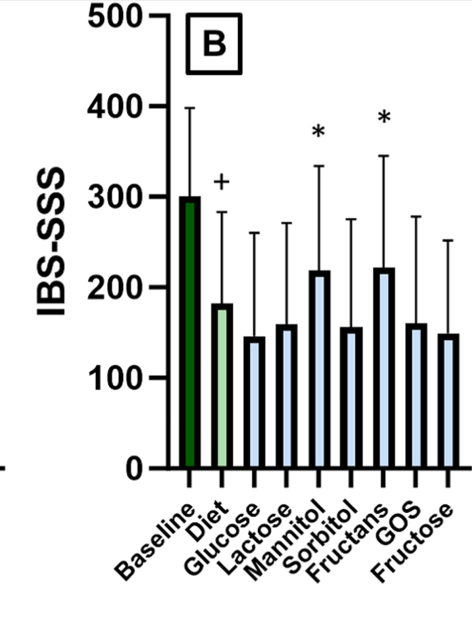
- IBS-SSS improved significantly after the elimination period compared with baseline (150 vs. 301, P < .0001, 80% responders)
- Symptom recurrence was triggered in 85% of the FODMAP powders, by an average of 2.5 FODMAPs/patient
- The most prevalent triggers were fructans (56%) and mannitol (54%), followed by galacto-oligosaccharides, lactose, fructose, sorbitol, and glucose (respectively 35%, 28%, 27%, 23%, and 26%) with a significant increase in abdominal pain at day 1 for sorbitol/mannitol, day 2 for fructans/galacto-oligosaccharides, and day 3 for lactose.
One limitation of the study was selecting the dose for the challenge/reintroduction. “In comparison to clinical practice, our selected dose was higher, intended to maximize the potential of inducing symptoms. On the other hand, if that information was available, we aimed to stay below a dose for an individual FODMAP that was shown to elicit symptoms in healthy controls.”
My take:
- Fructans and Mannitol had the highest prevalence rate as trigger foods upon reintroduction. However, the other groups all had at least a 23% chance of being a food trigger as well.
- Having available powders of the FODMAP grouping could potential expedite and standardize reintroduction in clinical practice. If a patient did well with the specific FODMAP powder, there is a good likelihood that the related foods would be tolerated as well.
Related blog posts:
- Which Diet is Best for Irritable Bowel Syndrome? A Randomized Trial
- Fructans and FODMAPs in Children with Irritable Bowel Syndrome
- Another Study: Low FODMAPs Diet for Irritable Bowel Syndrome
- Low FODMAP –Real World Experience
- An Unexpected Twist for “Gluten Sensitivity”
- Fructans, not Gluten, Inducing Symptoms In Patients with Reported Non-Celiac Gluten Sensitivity
Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.