To follow Charlie Lees:
- Can sign up for his substack:Atomic IBD
- Can follow him on twitter: @charlie_Lees
- Twitter link to ‘master thread’ with links to numerous recent essays




To follow Charlie Lees:



M Agrawal et al. Gastroenterol 2022; 163: 1547-1554. Open Access! The Rising Burden of Inflammatory Bowel Disease in Denmark Over Two Decades: A Nationwide Cohort Study
Key findings:
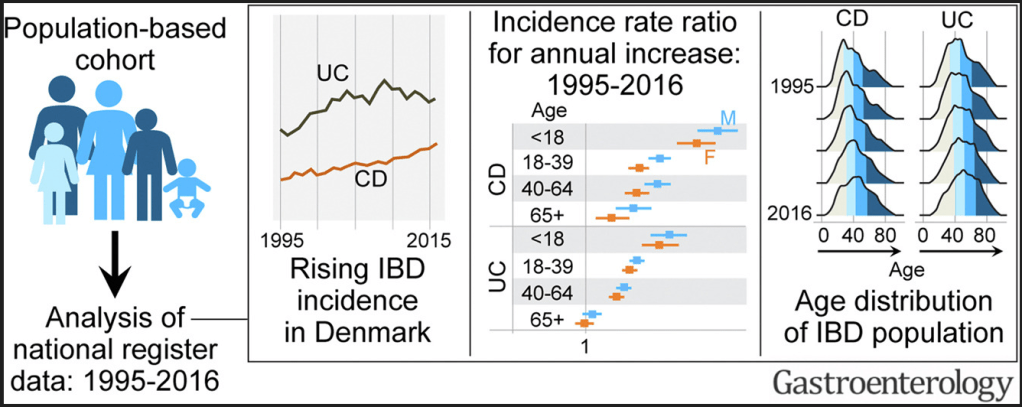
Y Pan et al. Inflamm Bowel Dis 2022; 28: 1865-1871. Utility of Therapeutic Drug Monitoring for Tumor Necrosis Factor Antagonists and Ustekinumab in Postoperative Crohn’s Disease
In this retrospective study (n=130), therapeutic drug levels in the postoperative period were associated with improved outcomes for anti-TNF agents (infliximab (IFX) or adalimumab (ADA) but NOT for ustekinumab (UST):
S Sasidharan et al. Inflamm Bowel Dis 2022; 28: 1833-1837. Fecal Calprotectin Is a Predictor of Need for Rescue Therapy in Hospitalized Severe Colitis
In this retrospective study (n=147), a fecal calprotectin >800 mcg/g independently predicted the need for inpatient medical rescue therapy (odds ratio, 2.61; 95% CI, 1.12-6.12). An admission calprotectin >800 mcg/g independently predicted surgery within 3 months (odds ratio, 2.88; 95% CI, 1.01-8.17). My take: This is the least surprising study I’ve read this past month —those with more severe colitis, based on calprotectin values, were more likely to need more intensive treatments.
R Dawson et al. Inflamm Bowel Dis 2022; 28: 1859-1864. Comparing Effectiveness of a Generic Oral Nutritional Supplement With Specialized Formula in the Treatment of Active Pediatric Crohn’s Disease
In this retrospective pediatric study (n=171), the authors found that a generic oral supplement (Fortsip) was as effective as a specialized formula (Modulen IBD) for enteral nutrition. “No difference was demonstrated in remission rate (Fortisip n = 67 of 106 [63%] vs Modulen IBD n = 41 of 64 [64%], P = .89), nonadherence rate (Fortisip n = 7 of 106 [7%] vs Modulen IBD 3 of 64 [5%], P = .57) or method of administration.” The main difference in outcome was a lower expense in the group receiving the generic formula. My take: This study is in agreement with previous studies.
Related blog posts:
F De Voogd, et al. Gastroenterol 2022; 163: 1569-1581. Intestinal Ultrasound Is Accurate to Determine Endoscopic Response and Remission in Patients With Moderate to Severe Ulcerative Colitis: A Longitudinal Prospective Cohort Study
27 patients with moderate to severe ulcerative colitis (UC) completed followup in this single-center, prospective, longitudinal cohort study. Key findings:

The associated editorial (C Palmela, C Maaster. Gastroenterol 2022; 163: 1485-1487. Open Access! The Use of Intestinal Ultrasound in Ulcerative Colitis-More Than a Mucosal Disease?) details other studies showing the utility of intestinal ultrasound, including the TRUST%UC study which enrolled 253 patients with UC. “. At baseline, 88.5% of patients had increased bowel wall thickness (BWT). Response to therapy could be detected as early as 2 weeks after initiation of therapy, as shown by reduction of abnormal BWT.” In anothre study with severe UC, “BWT reduction of >20% being an excellent predictor of response to intravenous steroids at 48 hours, as shown recently by Ivemark et al.10“
The editorial notes that intestinal ultrasound “is often thought as being operator dependent. Nonetheless, several studies have shown an excellent inter-observer agreement in IUS, especially for the assessment of BWT,7,12 as was also found in this [De Voogd] study.” An additional finding in the De Voogd study was that the “the submucosa was the most thickened layer, and after 8 weeks of therapy it was also the most responsive layer;” thus, UC is not simply a mucosal disease.
My take: This study shows that with more widespread adoption, many UC patients could be followed non-invasively with intestinal ultrasound (and calprotectin).
Related blog post:
D Tarabar et al. Inflamm Bowel Dis 2022; 28: 1549-1554. A Prospective Trial with Long Term Follow-up of Patients With Severe, Steroid-Resistant Ulcerative Colitis Who Received Induction Therapy With Cyclosporine and Were Maintained With Vedolizumab
As noted previously, in my view, “bad” inflammatory bowel disease (IBD) occurs when treatments are not working; though, many would argue that any IBD is bad IBD. Today’s post concludes several reviewed articles that focus on the problem of IBD that is not responding well to treatment.
Methods: Seventeen steroid-resistant adult UC patients were treated with cyclosporine in combination with vedolizumab, with a follow up of 52 weeks. Only 2 patients in this chort had failed infliximab therapy. The authors administered IV cyclosporine at a dose of “2 to 4 mg/kg/d IV for 7 days, titrated to a goal trough level of 300 to 400 ng/mL.” In those with a response, patients were started on oral therapy along with IV vedolizumab. During oral therapy (for 8 weeks), goal trough levels were 150 to 250 ng/mL (measured weekly).
Key findings:
My take: Cyclosporine is a fast-acting medication and thus appropriate as a salvage therapy in those with severe disease. Concerns for adverse effects have led most pediatric GIs to favor infliximab for refractory severe UC. However, in selected patients, it could be a useful “bridge” to slower-acting long-term treatments. It is possible (likely) that insurance issues would be less with cyclosporine than tofacitinib as a bridge therapy.
**An alternative agent to cyclosporine is tacrolimus. Hamel B, Wu M, Hamel EO, Bass DM, Park KT. Outcome of tacrolimus and vedolizumab after corticosteroid and anti-TNF failure in paediatric severe colitis. BMJ Open Gastroenterol. 2018;5(1):e000195 (“Positioning Biologic Therapies in the Management of Pediatric Inflammatory Bowel Disease” & 14% of U.S. Infected with COVID-19)
Related blog posts:
Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.

Island Ford National Recreational Area, Sandy Springs GA
As noted yesterday, in my view, “bad” inflammatory bowel disease (IBD) occurs when treatments are not working; though, many would argue that any IBD is bad IBD. Over the next few days, reviewed articles will focus on the problem of IBD that is not responding well to treatment. This article reports on the use of tofacitinib to avoid colectomy in children with severe ulcerative colitis.
BD Constant et al. JPGN 2022; 75: 724-730. Tofacitinib Salvage Therapy for Children Hospitalized for Corticosteroid- and Biologic-Refractory Ulcerative Colitis
This small (n=11) retrospective single-center cohort study of consecutive hospitalized pediatric patients initiating tofacitinib for refractory ulcerative colitis from 2018 to 2021. All patients demonstrated nonresponse to both intravenous corticosteroids and anti-TNF therapy prior to tofacitinib initiation.
Key findings:
My take: Given the small numbers, this is clearly an area where cooperation (& ImproveCareNow) could be helpful in determining the safety and effectiveness of tofacitinib for pediatric ASUC. Also, if tofacitinib is used as a ‘bridge’ this is likely to present insurance coverage issues.
Related article:
Hoisnard L, Pina Vegas L, Dray-Spira R, et al. Annals of the Rheumatic Diseases Published Online First: 05 October 2022. doi: 10.1136/ard-2022-222824. Risk of major adverse cardiovascular and venous thromboembolism events in patients with rheumatoid arthritis exposed to JAK inhibitors versus adalimumab: a nationwide cohort study Methods: This was a nationwide population-based cohort study (n=15,835) of the French national health data system, the exposed group initiating a JAKi and non-exposed group initiating adalimumab Key findings: Risk of major adverse cardiovascular events (MACEs) for the exposed versus non-exposed group was not significant: HRw 1.0 (95% CI 0.7 to 1.5) (p=0.99), nor was risk of VTEs significant: HRw 1.1 (0.7 to 1.6) (p=0.63). This study provides reassuring data regarding the risks of MACEs and VTEs in patients initiating a JAKi versus adalimumab, including patients at high risk of cardiovascular diseases.
Related blog posts:
From Crohn’s and Colitis Foundation, Georgia Chapter, December Newsletter: Donate to Cohen-Saripkin Fund


K Hawa et al. JPGN Reports 2022; doi: 10.1097/PG9.0000000000000255. Open Access! Postcolectomy Enteritis in a Pediatric Patient With Ulcerative Colitis
In this case report, the authors describe a 16 yo male with ulcerative colitis who on postoperative day 4 after colectomy developed an early onset of non-infectious enteritis. Treatment included corticosteroids “without significant improvement over 2 weeks. As his corticosteroid dose was tapered by 5 mg/day each week, ostomy output decreased, and abdominal pain and distension improved.” He continued to improve without further interventions. “6 weeks postoperatively, repeat upper endoscopy and ileoscopy demonstrated resolution of his duodenitis and ileitis grossly.”
“This is the first published case of a pediatric patient with PCE [postcolectomy enteritis], an entity previously only described in adults. PCE may be difficult to diagnose; in patients initially diagnosed with UC who develop small bowel inflammation following colectomy, the concern is often misdiagnosed Crohn’s disease.” The authors note that the “presentation is differentiated from Crohn’s disease based on timing [days to months after surgery], histology and diffuse pattern of mucosal involvement (3).”
My take: Rare cases PCE (a self-limited enteritis) occur and can be difficult to distinguish from Crohn’s disease. With PCE, if findings improve, this would suggest PCE whereas if symptoms persist, then this would suggest Crohn’s disease.
This case reminds me of the swimming test for a witch. Sinking to the bottom indicated that the accused was innocent while floating indicated a guilty verdict. Which is to say that we don’t have a great test to tell if someone has PCE at presentation.
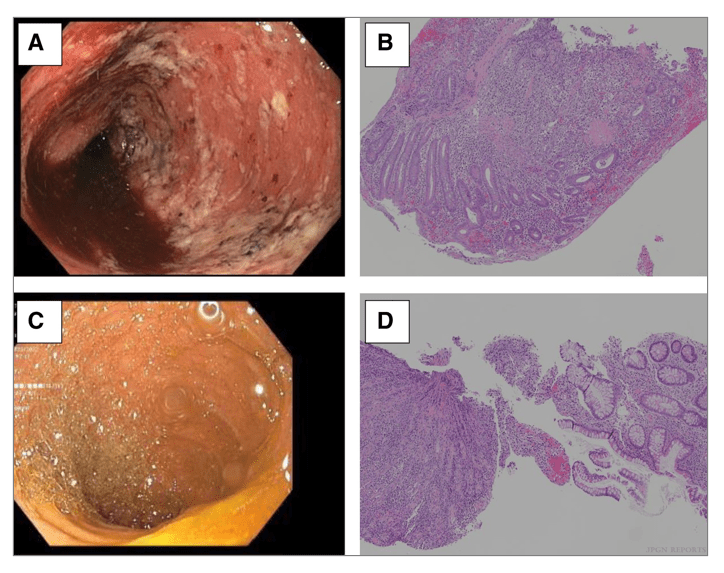
(A) Initial ileoscopy image—diffuse inflammation characterized by erythema, exudate, and friability. (B) Initial ileal histopathology—severe active ileitis, erosion, and focal crypt irregularity (magnification 100×).
Repeat ileoscopy image– (C) normal mucosa. (D) Repeat ileal histopathology—nonspecific changes including patchy lamina propria lymphoplasma cell infiltrate, eosinophilia, and spotty glandular and intraepithelial lymphocytosis (magnification 100×).
D Ley et al. Clin Gastroenterol Hepatol 2022; 20: 2588-2597. Open Access! New Therapeutic Strategies Have Changed the Natural History of Pediatric Crohn’s Disease: A Two-Decade Population-Based Study
This retrospective study dating back to 1988 examined 1007 patients diagnosed with CD who were followed up for a median duration of 8.8 years.
Key findings:
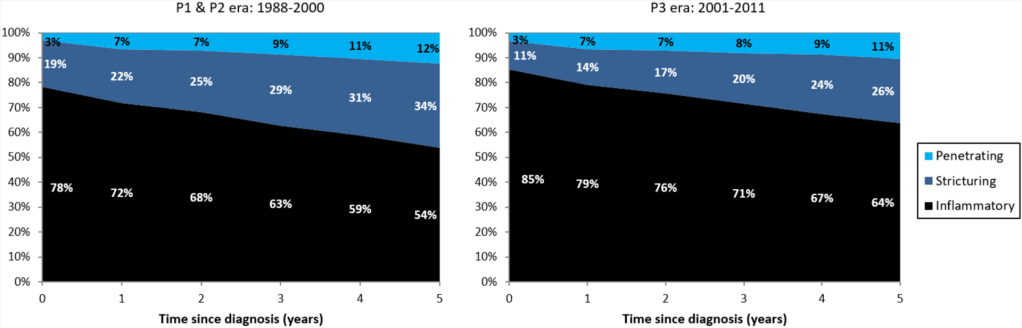
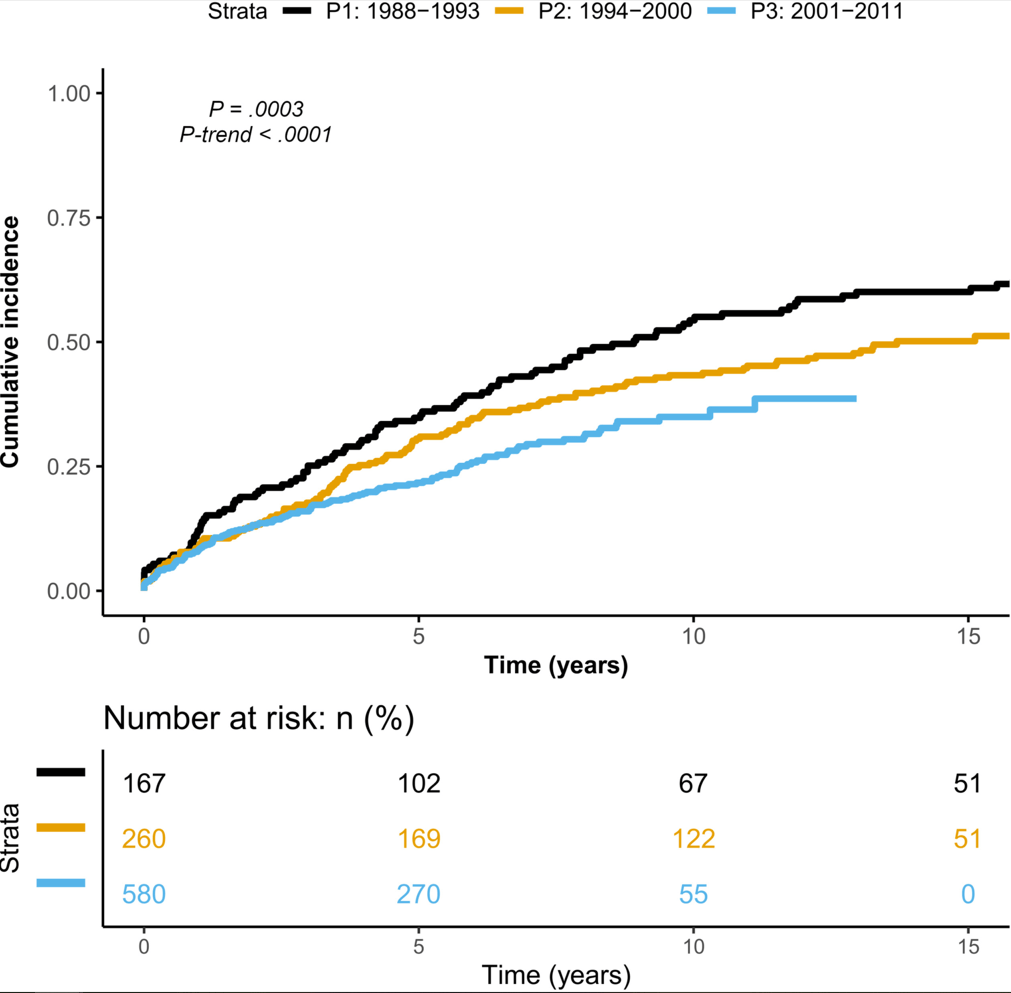
LE Targownik et al. Clin Gastroenterol Hepatol 2022; 20: 2607-2618. Earlier Anti-TNF Initiation Leads to Long-term Lower Health Care Utilization in Crohn’s Disease but Not in Ulcerative Colitis
Methods: The authors “used health administrative data from Manitoba, Canada to identify all persons with a new diagnosis of inflammatory bowel disease (IBD) between 2001 and 2018 who received tumor necrosis factor antagonists (anti-TNF) therapy and had at least 1 year of post anti-TNF initiation follow-up.”
Key findings:
My take: These two studies show that use of biologic therapy is associated with better outcomes in Crohn’s disease including fewer intestinal resections and fewer hospitalizations. It appears that earlier use may alter the natural history in part by reducing the likelihood of stricturing disease. Interestingly, the RISK study showed a reduction in penetrating disease with early use of biologics but not a reduction in stricturing disease (Related blog post: CCFA: Updates in Inflammatory Bowel Disease 2017 (part 3))
BN Limketkai et al. Inflamm Bowel Dis 2022; 28: 1627-1636. Open Access! Dietary Patterns and Their Association With Symptoms Activity in Inflammatory Bowel Diseases. This retrospective study with dietary surveys of 691 participants found the following:
Diet PB1 (“Plant-based Diet 1”) was characterized by much higher intake of fruits, vegetables, plant-based proteins, and cooked grains than most other dietary clusters. There was low water intake in favor of juices and other beverages. There was otherwise average intake of added fats and oils, sugars, seafood, and dairy products, and modest intake of meats, eggs, mixed grains, and breads.
Diet PB2 (“Plant-based Diet 2”) was characterized by high intake of fruits, vegetables, plant proteins, and cooked grains and low intake of animal proteins (especially red and cured meats), added fats, sweetened beverages, sweet bakery products, other desserts, eggs, and breads. There was also a reduction of other beverages in favor of water. There was otherwise an average intake of seafood and dairy products.
M Chaparro et al. Inflamm Bowel Dis 2022; 28: 1725-1736. Open Access! Long-Term Real-World Effectiveness and Safety of Ustekinumab in Crohn’s Disease Patients: The SUSTAIN Study In this retrospective study, 97% of the 463 patients had received prior biological therapy.
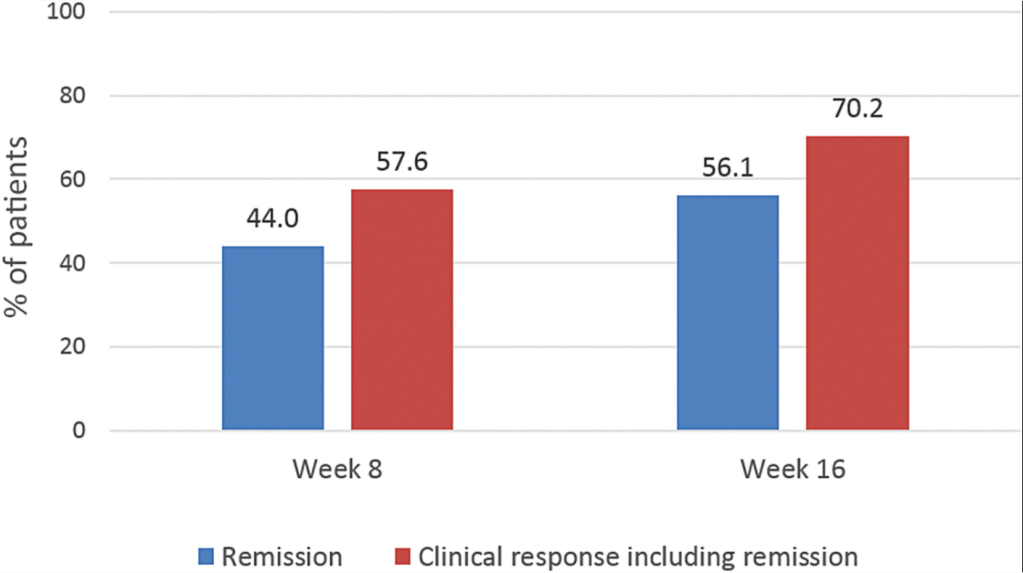
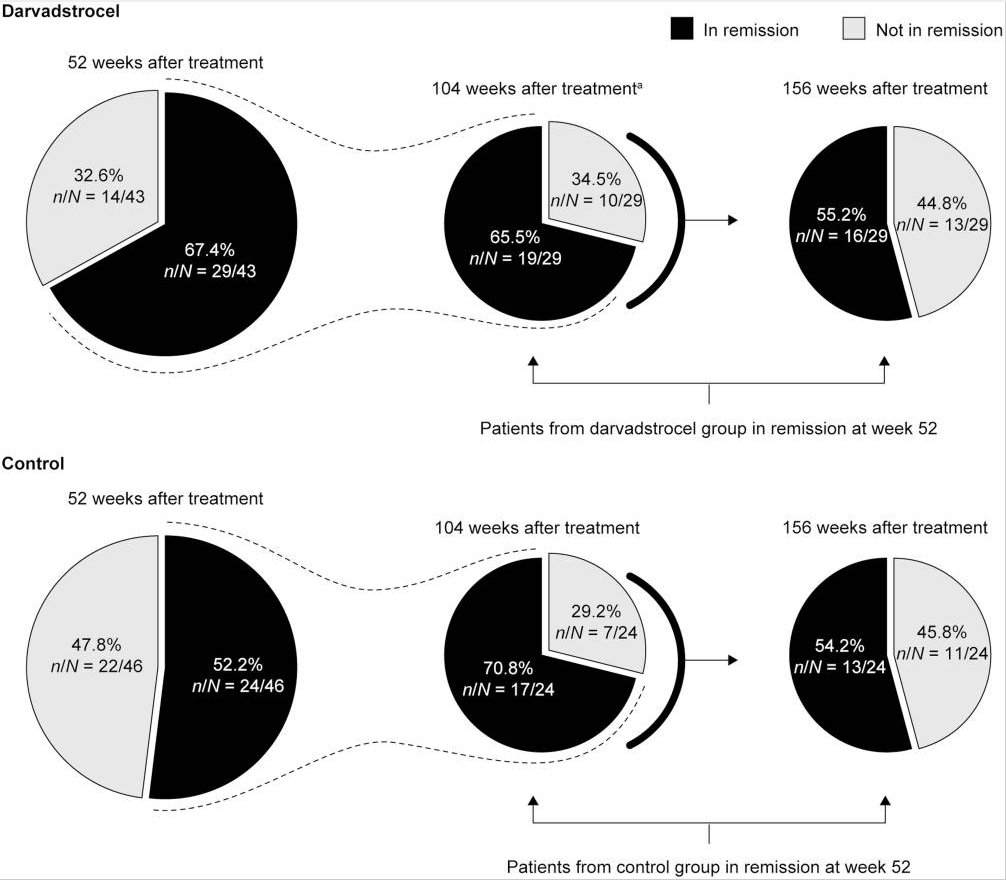
J Panes et al. Inflamm Bowel Dis 2022; 28: 1737-1745. Open Access! INSPECT: A Retrospective Study to Evaluate Long-term Effectiveness and Safety of Darvadstrocel in Patients With Perianal Fistulizing Crohn’s Disease Treated in the ADMIRE-CD Trial
Background: The current chart review study evaluated the longer-term effectiveness and safety of darvadstrocel (expanded allogeneic adipose-derived mesenchymal stem cells).; n=43 treated patient and n=46 controls.
Key findings:
Q Buck et al. JPGN 2022; 75: 462-465. Routine Histology-Based Diagnosis of CMV Colitis Was Rare in Pediatric Patients
Key findings from this retrospective review (2011-2019):
The study finding that half of the cases of CMV in the IBD population were identified prior to treatment indicates that the underlying IBD may be a more important susceptibility factor than the immunosuppressive medications.
My take: This study indicates that CMV colitis remains important in the post-transplant population but is rarely consequential in the pediatric IBD population.
Related blog posts:

Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.
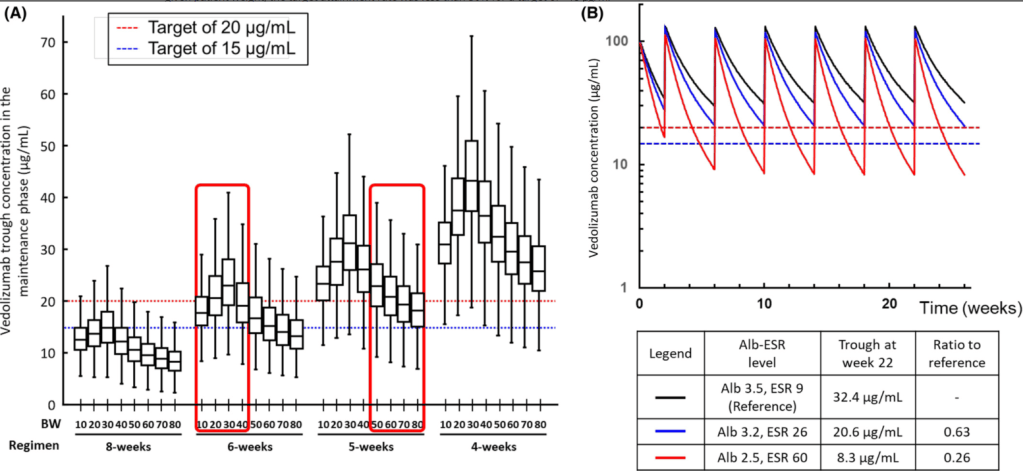
RJ Colman et al. AP&T 2022; https://doi.org/10.1111/apt.17277. Open access! Real world population pharmacokinetic study in children and young adults with inflammatory bowel disease discovers novel blood and stool microbial predictors of vedolizumab clearance
“The study included data from 463 observed vedolizumab concentrations (59 peaks and 404 troughs) from 74 patients with IBD (52 with Crohn’s disease and 22 with ulcerative colitis or unclassified IBD, median age 16 years)…This study was part of the multicentre REFINE study, which aimed to investigate paediatric PK factors among different biological therapies. Both induction and maintenance doses were between 6 and 10 mg/kg for patients less than 30 kg and 300 mg for patients above 30 kg.”
Key findings:
My take: This study shows that therapeutic drug monitoring (TDM) is likely to be beneficial in improving outcomes in pediatric patients receiving vedolizumab. Low albumin in particular is associated with increased drug clearance. From this study, it looks like most pediatric patients will need dosing every 4 to 6 weeks to achieve good levels. The authors in their discussion reinforce the utility of TDM to “guide anti-TNF dose optimisations has been shown to improve durability and reduce both immunogenicity and loss of response.”
References:
13 Dubinsky MC, Mendiolaza ML, Phan BL, Moran HR, Tse SS, Mould DR. Dashboard-driven accelerated infliximab induction dosing increases infliximab durability and reduces immunogenicity. Inflamm Bowel Dis. 2022; 28: 1375– 85.
51 Strik AS, Löwenberg M, Mould DR, Berends SE, Ponsioen CI, van den Brande JMH, et al. Efficacy of dashboard driven dosing of infliximab in inflammatory bowel disease patients: a randomized controlled trial. Scand J Gastroenterol 2021; 56: 145– 154.
Related blog posts:

Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.