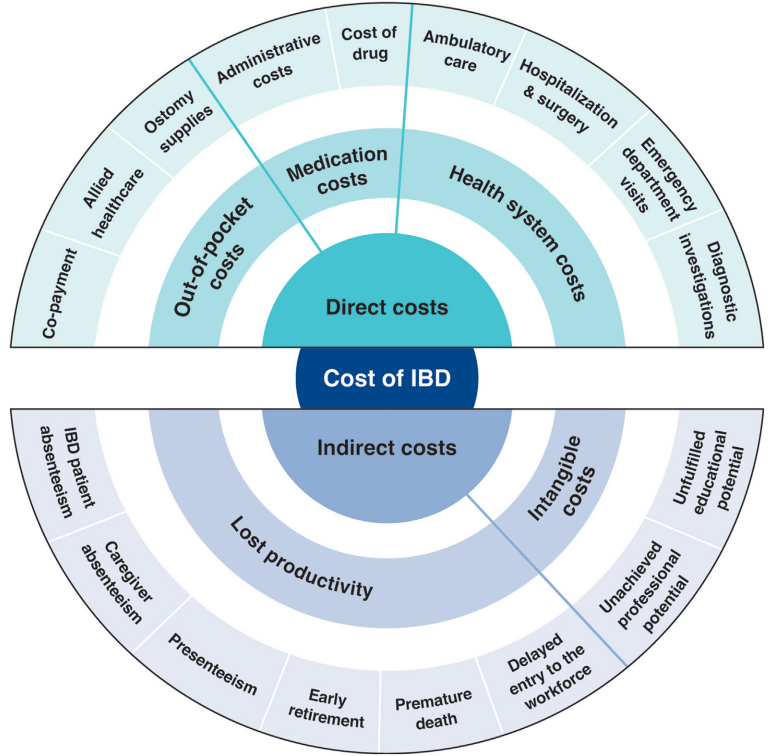
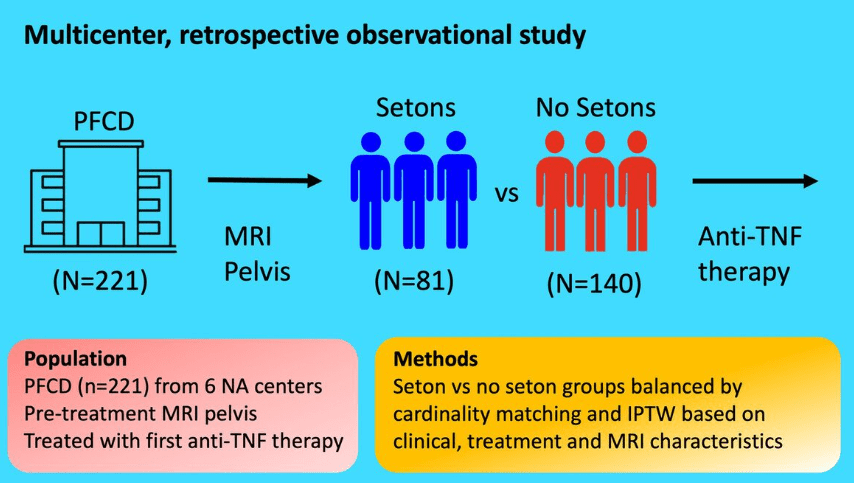
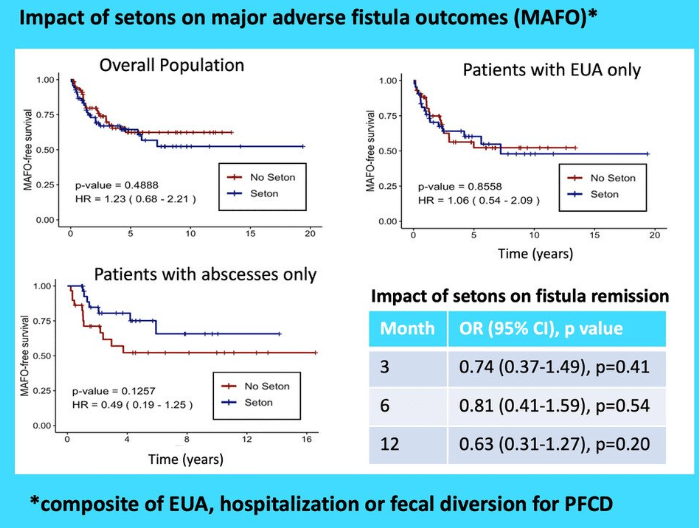
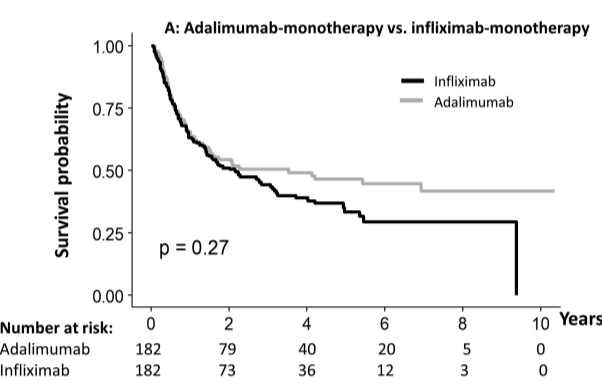
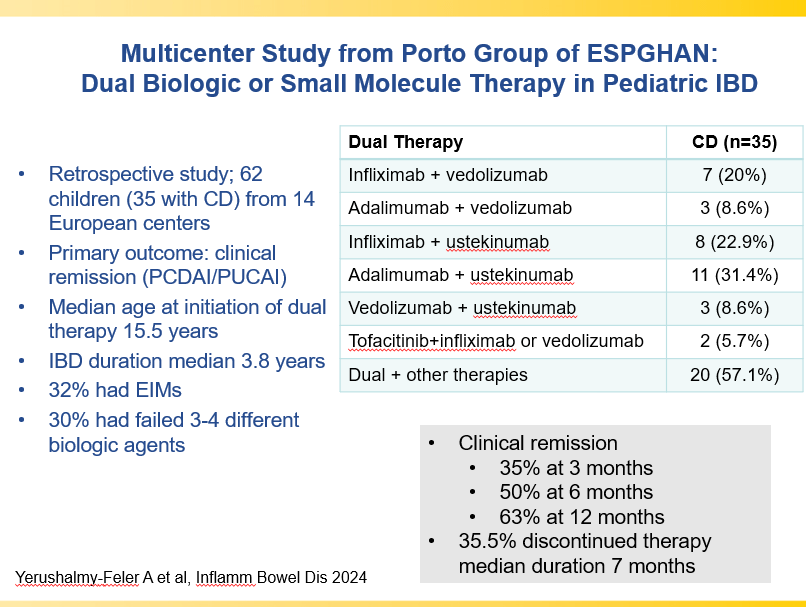
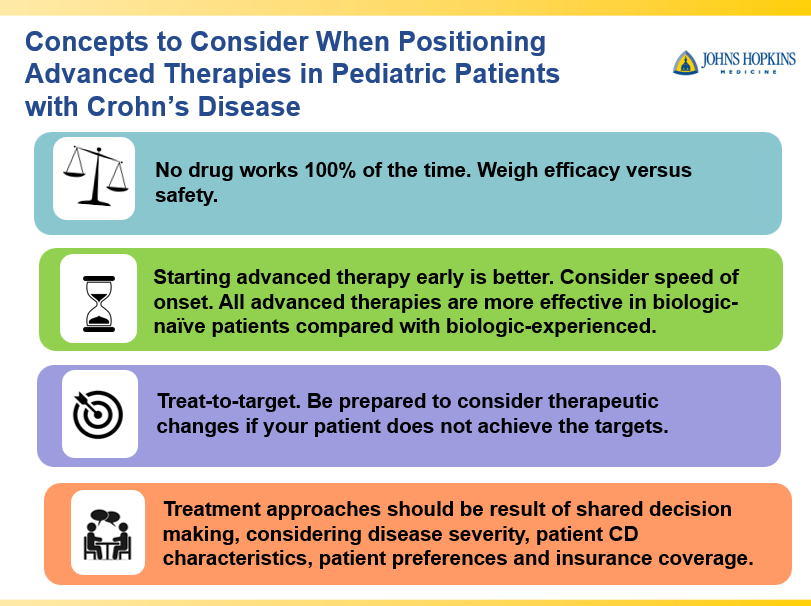
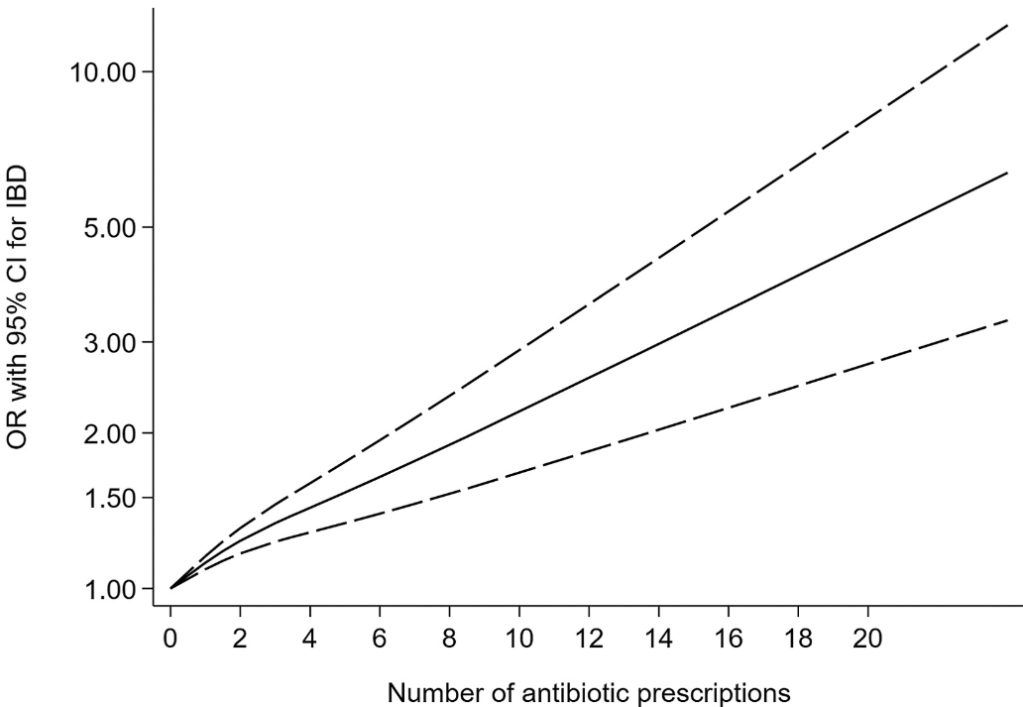
S Cenni et al. J Pediatr Gastroenterol Nutr. 2025;80:799–806. The prevalence of constipation in children with new diagnosis of inflammatory bowel disease: A retrospective study
This was a cross-sectional observational study in pediatric IBD-patients (n=238) with 104 (43.6%) with Crohn disease (CD), 130 (54.6%) with ulcerative colitis (UC) and 4 (1.6%). Only patients who filled out the Rome IV questionnaire for FC, through dedicated symptom recall at the next clinic appointment or telephone recall, were finally enrolled in the study for subsequent analysis.
Key findings:
- Forty-seven out of 238 (19.7%) had a functional constipation history before the IBD diagnosis. In the CD children the prevalence of constipation before the IBD diagnosis was 19/104 (18.2%) and in the UC patients was 28/130 (21.5%).
- The difference in terms of endoscopic localization was statistically significant in UC patients presenting FC (p = 0.026) with a prevalence of proctitis and left side colitis (30% and 15%, respectively)
- There was a delay in the diagnosis of patients with preceding constipation
Discussion Points:
- The main limitations of the present study are certainly related to the retrospective nature and, therefore, the possibility of recall biases must be taken into account.
- Rectal bleeding that persists despite stool softener therapy should be investigated
My take: While this study shows that constipation is fairly common prior to a diagnosis of IBD, many times a parent is told that their child is constipated on the basis of an xray or simply because the child complained of stomach pain. This likely increases the risk of recall bias. My guess is that a prospective study involving careful questioning at the time of the initial colonoscopy would yield a lower number of children who had constipation at the time of diagnosis.
Related blog posts:
- Diagnosis and Misdiagnosis of Constipation
- What’s Wrong with Ordering an AXR for Constipation in the ER?
- Radiographs and Constipation -Bad Practice and Good Study
- Quality Improvement: Fewer Xrays for Constipation
- Does It Make Sense to Look for Celiac Disease in Children with Functional Constipation?
- Refractory Constipation -Terrific Update | gutsandgrowth