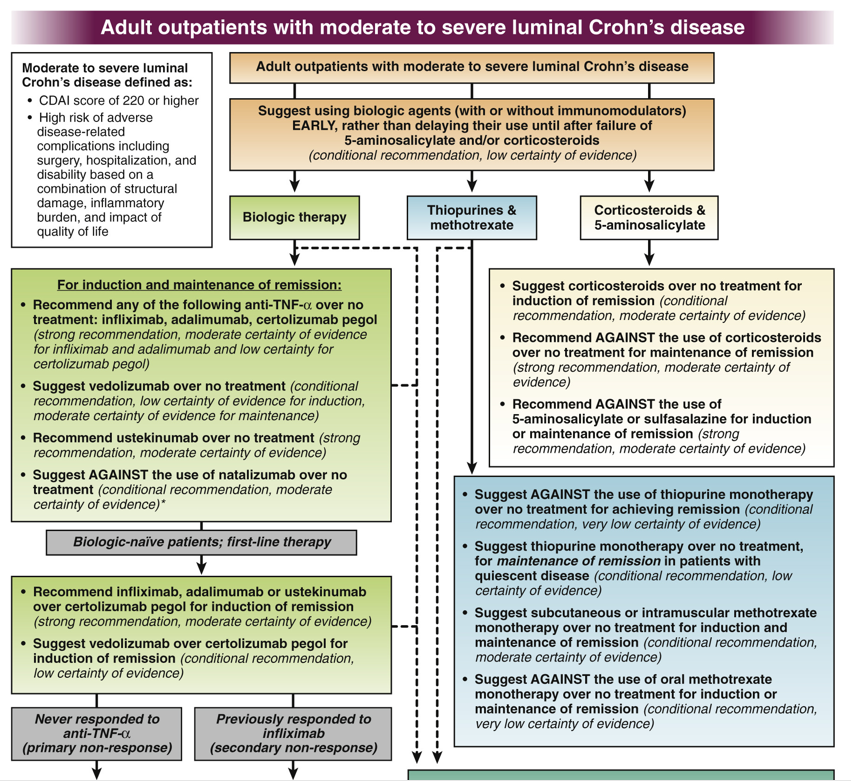
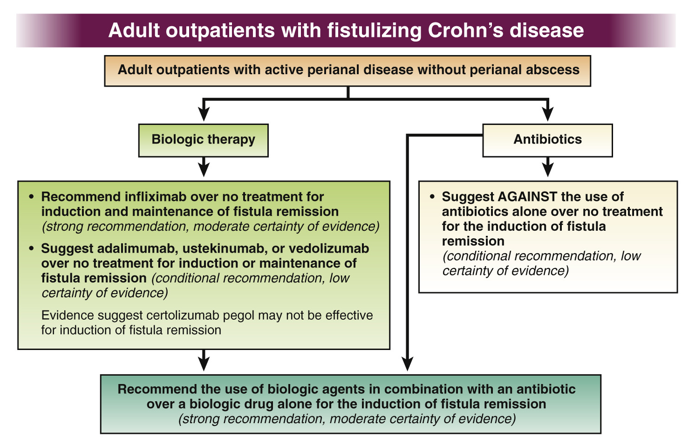
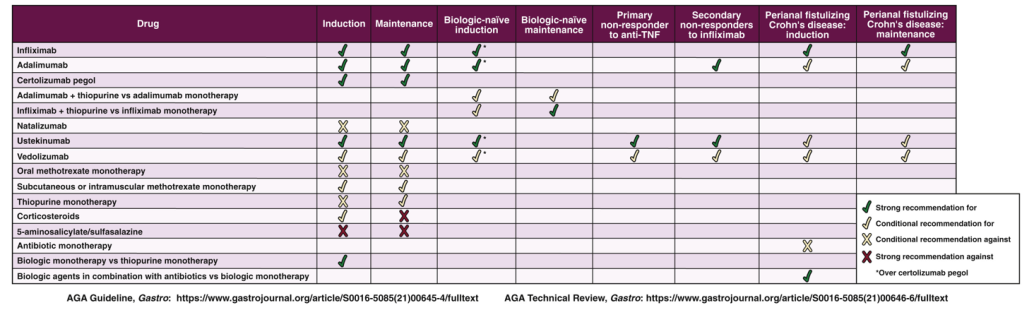
WJ Sandborn et al. Inflamm Bowel Dis 2021; 27: 994-1007. Full text: Safety of Ustekinumab in Inflammatory Bowel Disease: Pooled Safety Analysis of Results from Phase 2/3 Studies
Methods: Data from 6 ustekinumab phase 2/3 CD and UC studies were pooled, and safety was evaluated through 1 year; this included 2574 patients (1733 patient-years of follow-up)
Key Safety findings –Events per 100 patient years -placebo vs ustekinumab respectively:
- Adverse events: 165.99 [95% CI, 155.81–176.67] vs 118.32 [95% CI, 113.25–123.55])
- Serious AEs: 27.50 [95% CI, 23.45–32.04] vs 21.23 [95% CI, 19.12–23.51])
- Infections 80.31 [95% CI, 73.28–87.84] vs 64.32 [95% CI, 60.60–68.21])
- Serious infections: 5.53 [95% CI, 3.81–7.77] vs 5.02 [95% CI, 4.02–6.19])
- Malignancies excluding nonmelanoma skin cancer: 0.17 [95% CI, 0.00–0.93] vs 0.40 [95% CI, 0.16–0.83])
- Major cardiovascular events were rare with 2 in placebo group 0.34 and 2 in the ustekinumab group 0.12
More key findings:
- No cases of progressive multifocal leukoencephalopathy or reversible posterior leukoencephalopathy
- Antibodies to ustekinumab were identified in 3.6% of patients
My take: This study showed similar safety between ustekinumab and placebo, but is limited by short followup. The authors note that 5-year data from ustekinumab’s use with psoriasis has found no safety signals for malignancy.
Related blog posts:
- Stelara (Ustekinumab) Safety
- Ustekinumab for Refractory Pediatric UC
- AGA Guidelines: Moderate to Severe Ulcerative Colitis
- “Positioning Biologic Therapies in the Management of Pediatric Inflammatory Bowel Disease” & 14% of U.S. Infected with COVID-19 | gutsandgrowth
- Ustekinumab Over Vedolizumab as 2nd Line Agent for Crohn’s Disease | gutsandgrowth
Unrelated article: E Verduci et al. JPGN 2021; 72: 769-783: Full text: Role of Dietary Factors, Food Habits, and Lifestyle in Childhood Obesity Development: A Position Paper From the European Society for Paediatric Gastroenterology, Hepatology and Nutrition Committee on Nutrition