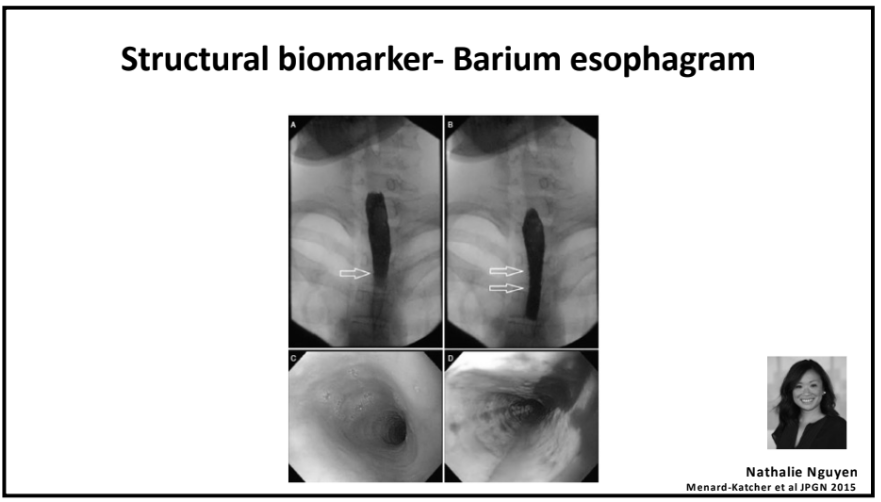
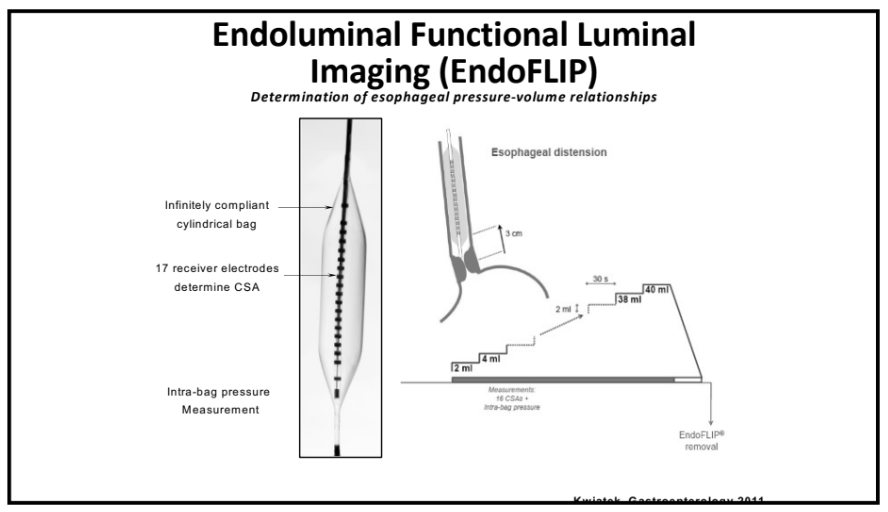
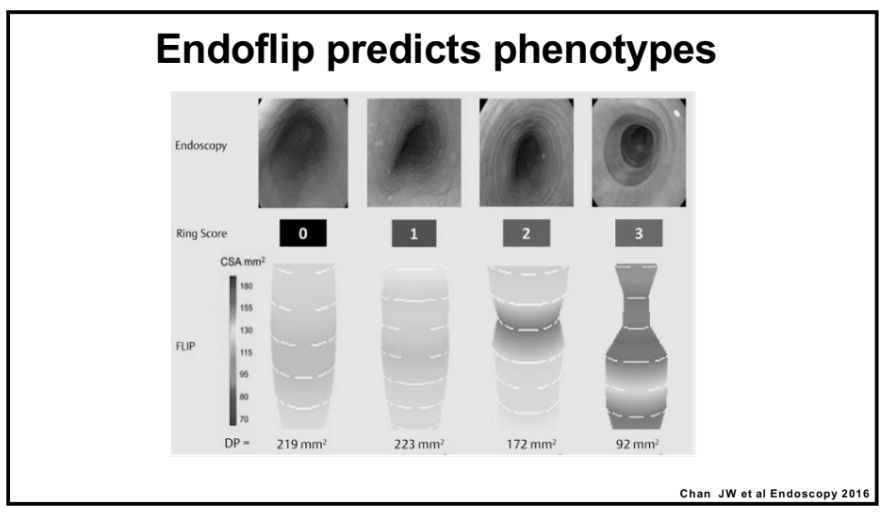
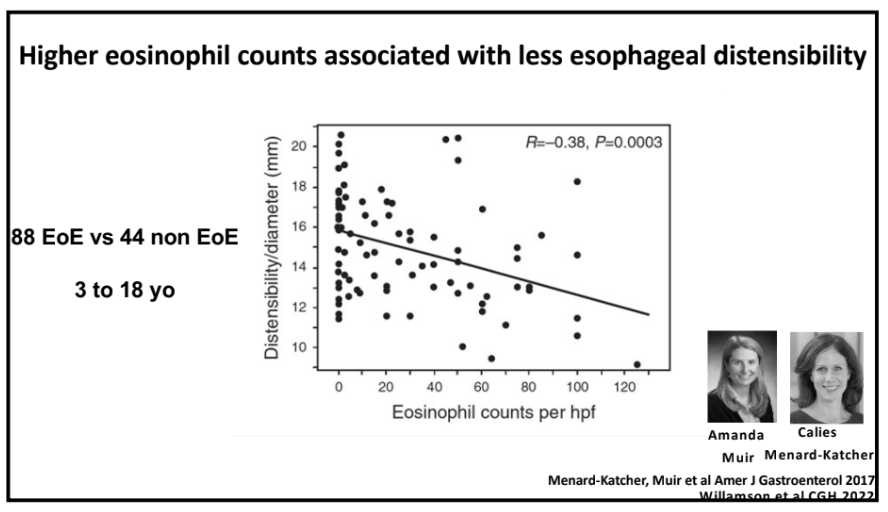
NY Times 2/19/24, Elizabeth Currid-Halkett: The Future of Medicine Is Unfolding Before Us. Are We Nurturing It?
This essay describes the terrific response of the author’s son with Duchenne Muscular Dystrophy to Elevidys. “At $3.2 million per patient, Elevidys is the second-most-expensive drug in the world.”
“D.M.D. prevents the production of dystrophin, a protein needed to protect and repair muscle cells. It is caused by a genetic mutation on the X chromosome, thus the disease almost exclusively affects boys (one in 3,300). Over time, children with D.M.D. lose muscle mass and thus the ability to do basic things like run and walk. Eventually they lose their ability to breathe, and they experience heart failure.”
She argues that “every child afflicted with a life-threatening disease deserves the chance Eliot has been given.” The article argues that the FDA should broaden the indications to cover older children with DMD in which the data are less convincing.
While this article discusses some ways to lower costs like reducing regulatory hurdles to get approval from FDA, it mainly discusses ways to force insurance companies to cover the cost. Disappointingly, the author (a public policy professor) does not address the reality that these costs are shared by everyone. For DMD alone, if the full cost is unchanged, that’s nearly $1000 for every person in the country. Yet, there are numerous other costly genetic therapies (for spinal muscular atrophy, hemophilia, Crigler-Najjar, others) in addition to extremely expensive treatments for cancer, inflammatory bowel disease, cystic fibrosis, rheumatoid arthritis, Alzheimer’s, obesity and many others.
My take: The ability to cure severe diseases like DMD is amazing. But, there is not a plan for how to deliver/afford these expensive therapies. If we cannot provide these treatments, it will be agonizing to watch patients deteriorate while a cure is just out of reach.
Meanwhile, we are not spending enough to address our biggest drivers of poor national health including poverty, education and poor diet.
Related blog posts:
- What is Wrong with the Glimmer of “Precision Medicine” | gutsandgrowth (2015) Key points: 1. “Precision medicine itself may ultimately make critical contributions to a narrow set of conditions, but the challenge we face…entails…willingness to address certain persistent social realities” 2.“Our public investments in broad, cross-sectional efforts to minimize…foundational drivers of poor health as poverty…are pitifully few in comparison with those of other countries.” 3. “We worry that an unstinting focus on precision medicine by trusted spokespeople for health is a mistake — and a distraction from the goal of producing a healthier population.”
- The Narrow Path of Personalized Cancer Medicine
- “Our Gene-Edited Future”
- NY Times: America can afford a world-class health system. Why don’t we have one?
- NY Times: “Our Food is Killing Too Many of Us”