The Guardian: DeepMind uncovers structure of 200m proteins in scientific leap forward

From The Washington Post via Eric Topol’s Twitter Feed

Link: AlphaFold developed by DeepMind



The Guardian: DeepMind uncovers structure of 200m proteins in scientific leap forward

From The Washington Post via Eric Topol’s Twitter Feed

Link: AlphaFold developed by DeepMind


AH Everhov et al. Inflamm Bowel Dis 2022; 28: 1160-1168. Open Access! Probability of Stoma in Incident Patients With Crohn’s Disease in Sweden 2003-2019: A Population-based Study
In a nationwide Swedish cohort of 18,815 incident patients with a minimum 5 years of follow-up, 652 (3.5%) underwent formation of a stoma. The 5-year cumulative incidence of stoma formation was 2.5%, with no differences between calendar periods (2003–2006, 2007–2010, and 2011–2014).
RWM Pauweis et al. Clin Gastroentol Hepatol 2022; 20: 1671-1686. Open Access! Prediction of Relapse After Anti-Tumor Necrosis Factor Cessation in Crohn’s Disease: Individual Participant Data Meta-analysis of 1317 Patients From 14 Studies
C Ma. Clin Gastroentol Hepatol 2022; 20: 1668-1670. Associated editorial. Open Access! To Stop or Not to Stop? Predicting Relapse After Anti-TNF Cessation in Patients With Crohn’s Disease
This study captured data from 1317 patients (including 927 patients stopping infliximab and 390 patients stopping adalimumab) to develop risk prediction models. “The authors confirm many of the high risk, albeit rather intuitive, factors that are associated with the risk of relapse, including younger age, younger age at diagnosis, smoking, upper gastrointestinal tract involvement, longer disease duration, absence of concomitant immunosuppressant use, previous anti-TNF failure, and absence of clinical remission.”
The editorial notes that even in the lowest risk group, more than 20% had risk of relapse within 1 year; in addition, stopping therapy increases risk of not recapturing remission with restart of treatment. “Stopping anti-TNF therapy is a highly personalized treatment decision and is one that carries considerable risks…therapeutic discontinuation of TNF antagonists should be reserved for the very small minority of patients who are in deep remission, have a strong desire to stop treatment, have no (or very few) characteristics of high-risk CD, can tolerate a substantial disease flare, and are fully informed of the risks of therapeutic withdrawal.”
Related blog posts:
WJ Sandborn et al. Clin Gastroenterol Hepatol 2022; 20: 1821-1830. Open Access! Efficacy and Safety of Extended Induction With Tofacitinib for the Treatment of Ulcerative Colitis
Graphical abstract below shows that 52.2% of patients who did not achieve clinical response to 8 weeks’ treatment with tofacitinib 10 mg BID in the induction studies achieved a clinical response following extended induction (delayed responders). At Month 12 of OCTAVE Open, 70.3%, 56.8%, and 44.6% of delayed responders maintained clinical response and achieved endoscopic improvement and remission, respectively. Corresponding values at Month 36 were 56.1%, 52.0%, and 44.6%.
My take: By extending the treatment induction to 16 weeks to determine response (rather than 8 weeks), the authors showed that 75% of patients with ulcerative colitis in the initial cohort respond to tofacitinib.
Related blog posts:

JB Wechsler et al. Clin Gastroenterol Hepatol 2022; 20: 1748-1756. A Single-Food Milk Elimination Diet Is Effective for Treatment of Eosinophilic Esophagitis in Children
Design: A prospective observational single-center study in 41 children with EoE treated with the 1-food elimination diet (1FED). Upper endoscopy with biopsies was performed after 8 to 12 weeks of treatment. The primary end point was histologic remission, defined as fewer than 15 eosinophils per high-power field.
Key findings:
My take: This study should prompt more widespread use of dairy elimination as a first line treatment prior to consideration of medications for long-term treatment. This study also reinforces the concept that symptom improvement remains an inadequate indicator of response. Perhaps, Chick-Fil-A marketing needs to be used for our EoE patients to shun cows (cow’s milk in this case).
Related blog posts:

P Martin et al. Clin Gastroenterol Hepatol 2022; 20: 1766-1775. Open access! Treatment Algorithm for Managing Chronic Hepatitis B Virus Infection in the United States: 2021 Update
This article provides an updated treatment algorithm with many changes since 2015.
Key points:
New terminology. The authors provide updated terminology. The term ‘immune tolerant’ is losing favor. “There is also evidence that during the immune tolerant phase virologic events occur, such as integration of viral DNA into the host genome, which may help set the stage for the subsequent development of HCC.” As such, the new terminology focuses on “describing the 2 main characteristics of chronicity, infection alone and infection with evidence of ongoing hepatic inflammation (ie, hepatitis).” (See below)
TAF preferred over TDF. “The 4 first-line therapies available for managing CHB infection in the United States are peginterferon alfa-2a, entecavir (ETV), tenofovir disoproxil fumarate (TDF), and tenofovir alafenamide (TAF)….It is the opinion of most—but not all—members of the panel that when antiviral therapy is contemplated, TAF is preferred over TDF because of the lower risks of renal or bone side effects and higher likelihood of ALT normalization through 48 weeks.” For decompensated cirrhosis, TAF and peginterferon are NOT recommended.
Long term treatment is the rule (not the exception). “HBeAg-positive patients with evidence of less extensive fibrosis (<F3) should be treated long-term, even after HBeAg seroconversion and virologic suppression because of the risks of virologic relapse55 and ALT flares, except when treatment is initiated solely for the purpose of prevention of vertical transmission…For HBeAg-negative patients without HBsAg seroconversion, the panel does not recommend stopping treatment.”
Monitoring. “Primary nonresponse to ETV, TDF, or TAF is rare; therefore, any patients who are not responsive to these agents after 12 to 24 weeks should be evaluated for compliance.”
Table 1 (see below) summarizes treatment recommendations. In almost all patients with HBV, if there is elevated ALT, treatment is recommended.
Terminology:

Table for Treatment Recommendations:
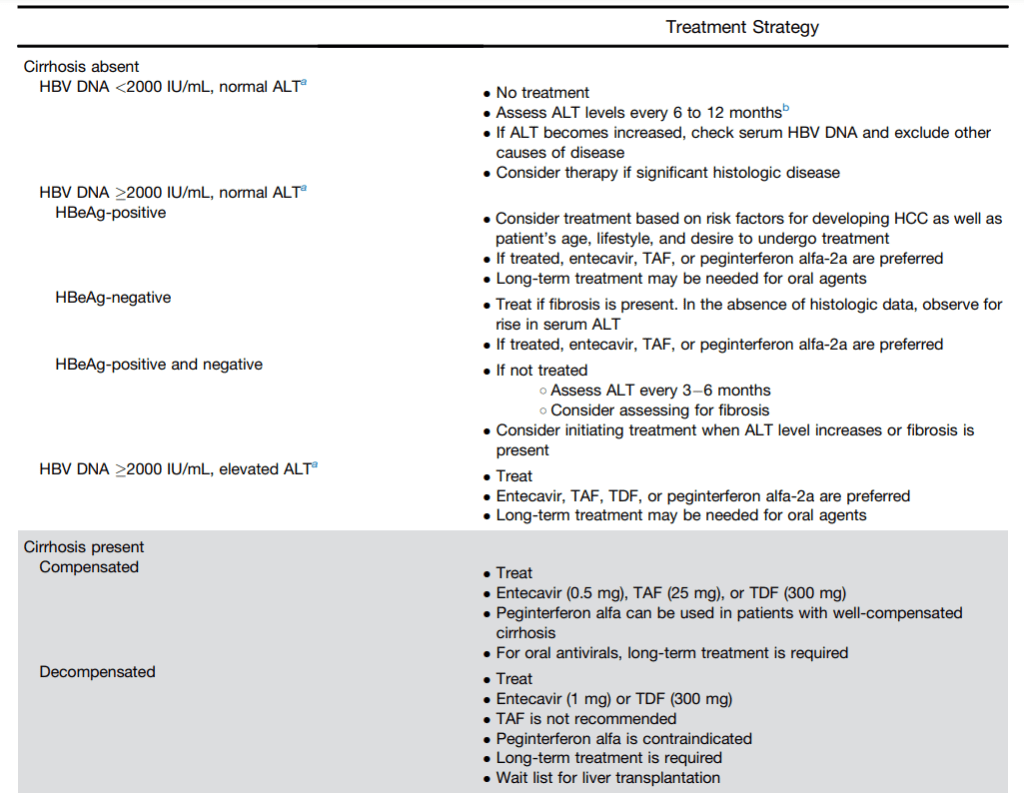
My take: Forget about “immunotolerant” HBV. Until better treatments emerge, patients with HBV and elevated liver enzymes are likely to need long-treatment if using TAF, TDF or entecavir.
Related blog posts:

Thanks to Ben Gold for this reference
My take: This is a step in the right direction.
Related posts:

Fun story form Avi Yemini on Twitter (98 second video): “You can never get tired of hearing this story about Queen Elizabeth II”
——————————————————-
HI Allen et al. Clin Exp Allergy 2022; 52: 848-858. https://doi.org/10.1111/cea.14179. Open Access: Detection and management of milk allergy: Delphi consensus study
This study reviewed the topic of milk allergy (both IgE-mediated and non-IgE mediated milk allergy) and provides consensus recommendations from 28 non-conflicted multidisciplinary international experts.
Key points:
My take: Milk allergy is overdiagnosed in infancy. This guideline supports a more supportive treatment approach. Because breastmilk is hypoallergenic, maternal dietary restriction may be more harmful than helpful in those with mild symptoms (especially if not having trouble with growth or with hypoalbuminemia). Table 1 below lists some of the most important of the 38 consensus recommendations.
Related blog posts:


If you have not seen this video from 2014 (42 seconds), I recommend it for a good laugh. I’ve seen it many times and I think it is funny every time.
————————–
S Haflidadottir et al. JPGN 2022; 75: 138-144. Mycophenolate Mofetil Use Is Associated With Reduced Incidence of Food Allergy in Liver Transplanted Children. N=107. Key finding: Children treated with MMF in addition to tacrolimus 1 year after transplantation reported less food allergy (12.5% vs 37.8%, P = 0.003) and sensitization to food allergens one year after transplantation (8.9% vs 17.8%, P = 0.02) than those not receiving MMF. The effect of MMF was not due to reduced trough levels of tacrolimus.
MB Vos et al. Hepatology 2022; 76: 429-444. Open access: Randomized placebo-controlled trial of losartan for pediatric NAFLD Key finding: Losartan did not significantly reduce ALT in children (n=83) with NAFLD when compared with placebo in this multicenter, double-masked, placebo-controlled, randomized clinical trial
G Indoli et al. Hepatology 2022; 76: 445-455. Sofosbuvir-velpatasvir-voxilaprevir in adolescents 12 to 17 years old with HCV infection All patients (n = 21) were naïve to HCV DAAs, and none had cirrhosis. Key finding: 100% of patients (21 of 21) reached SVR12 (8 week treatment course)

CR Meijer et al. Gastroenterol 2022; 163: 426-436. Open access: Prediction Models for Celiac Disease Development in Children From High-Risk Families: Data From the PreventCD Cohort
B Lebwohl, L Greco. Gastroenterol 2022; 163: 368-369 (editorial). Open access: Can We Predict the Onset of Celiac Disease?
Design: “In this study, the investigators analyze long-term follow-up data from the PreventCD trial, a randomized trial of infants [n=944] with a first-degree relative with CD that was designed to test the strategy of low-dose gluten introduction at age 4 months. The trial did not show that this strategy reduced the risk of CD development,7 but the abundant data collected during this trial have allowed these investigators to study risk factors for the development of CD among the trial participants.” The median f/u was 8.3 yrs.
Key points from study and editorial:
HLA testing in this setting has historically been performed primarily due to its excellent negative predictive value. Because HLA DQ2 and DQ8 are present in nearly 100% of people with CD, the primary value of its use has been in ruling out CD when an individual is found to have neither haplotype. This study shows some usefulness in predicting the likelihood of CD.
My take: This study showed 14% of high-risk children developed celiac disease and the number is likely to escalate with more time. In first-degree relatives, checking HLA-DQ2/8–typing may help determine frequency of screening in asymptomatic individuals –though simply choosing to screen every 1-2 years would be a reasonable alternative.
It should be noted that current expert guidelines provide divergent advice; “NASPGHAN recommends that asymptomatic children in high-risk groups (including first-degree relatives) be screened, 4 but the United States Preventive Services Task Force concluded that the evidence is insufficient to warrant recommending for or against screening asymptomatic individuals.”
Related blog posts:

Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.
Briefly noted –all of these articles are open access:
A Volkers et al. AP&T 2022; https://doi.org/10.1111/apt.17153 Open access: Real-world experience of switching from intravenous to subcutaneous vedolizumab maintenance treatment for inflammatory bowel disease. In this prospective cohort study, patients (n=135) with IBD who had ≥4 months IV vedolizumab were switched to SC vedolizumab.
Key findings:
Related blog posts:
HC Kaplan et al. Am J Gastroenterol 2022 Jun 1;117(6):902-917. Open access: Personalized Research on Diet in Ulcerative Colitis and Crohn’s Disease: A Series of N-of-1 Diet Trials. In this study, 21 patients (completed trial) were randomized to 1 of 2 sequences of 4 alternating 8-week SCD (specific carbohydrate diet) and MSCD (modified specific carbohydrate diet) periods.
Key findings: “SCD and MSCD did not consistently improve symptoms or inflammation.” “Some individuals had improvement in symptoms and fecal calprotectin compared with their UD, whereas others did not.” The authors note that it took 18 months to recruit 54 patients for this study across 19 research sites.
Related blog posts:
EL Barnes et al. Inflamm Bowel Dis 2022; 28: 983-987. Open access: Racial and Ethnic Distribution of Inflammatory Bowel Disease in the United States The authors electronic health records from 337 centers from January 2013 to December 2018 with nearly 40 million patients in U.S.
Key findings:
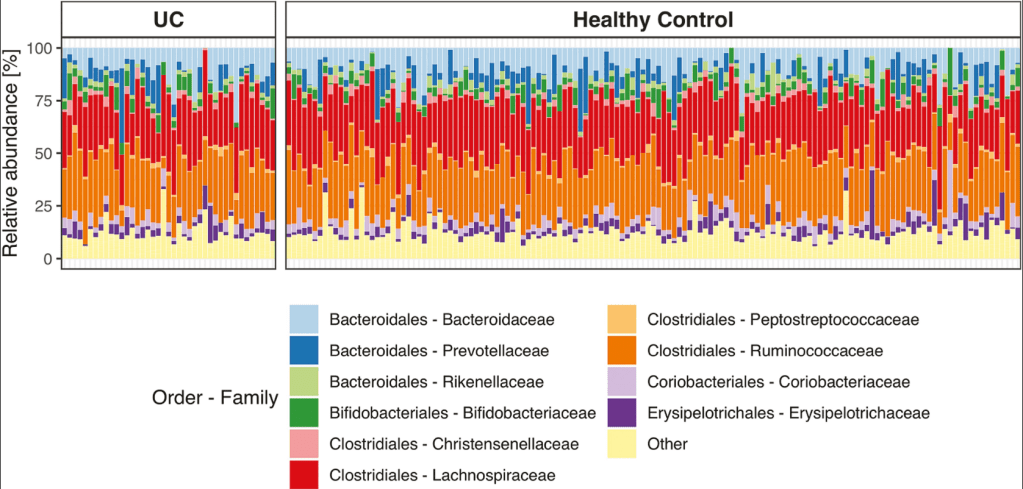
M Frioirksmork et al. Inflamm Bowel Dis 2022; 28: 1081-1089. Open access: Similar Gut Bacterial Composition Between Patients With Ulcerative Colitis and Healthy Controls in a High Incidence Population: A Cross-sectional Study of the Faroe Islands IBD Cohort. This cross-sectional study from the Faroe Islands (which has very high incidence of IBD) consisted of 41 patients with established ulcerative colitis and 144 age- and sex-matched healthy controls.
Key findings: There was a similarity in bacterial community composition and absence of the beneficial Akkermansia genus in both groups.