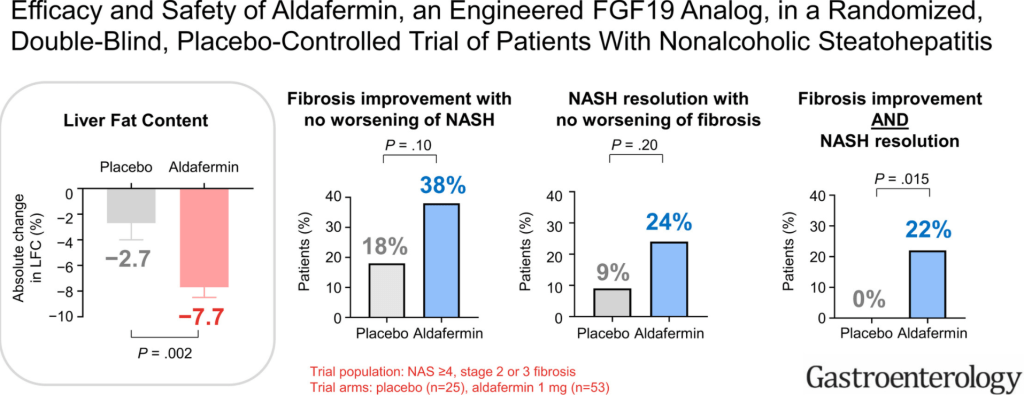
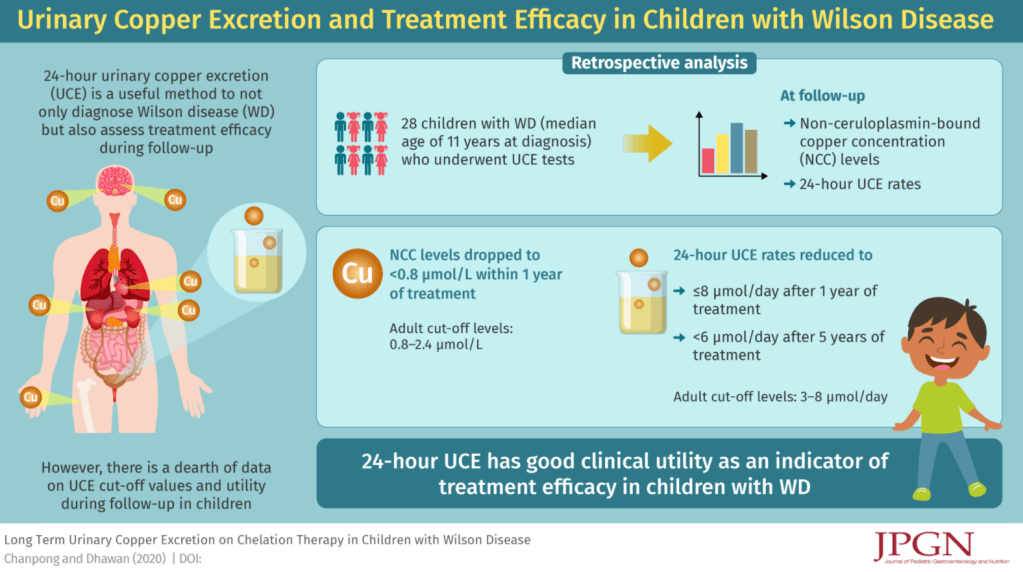
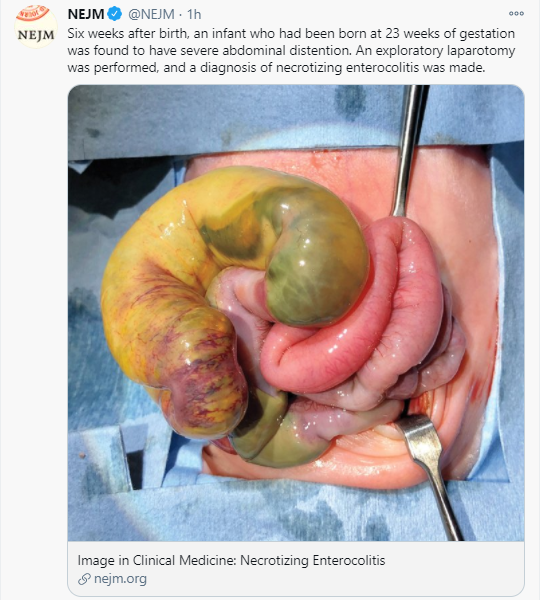
Aguilera-Lizarraga, J., Florens, M.V., Viola, M.F. et al. Local immune response to food antigens drives meal-induced abdominal pain. Nature (2021). https://doi.org/10.1038/s41586-020-03118-2 (Thanks to Ben Gold’s twitter feed for this reference)
Background: “Up to 20% of people worldwide develop gastrointestinal symptoms following a meal, leading to decreased quality of life, substantial morbidity and high medical costs”
“Here we show that a bacterial infection and bacterial toxins can trigger an immune response that leads to the production of dietary-antigen-specific IgE antibodies in mice, which are limited to the intestine. Following subsequent oral ingestion of the respective dietary antigen, an IgE- and mast-cell-dependent mechanism induced increased visceral pain. This aberrant pain signaling resulted from histamine receptor H1-mediated sensitization of visceral afferents. Moreover, injection of food antigens (gluten, wheat, soy and milk) into the rectosigmoid mucosa of patients with irritable bowel syndrome induced local oedema and mast cell activation.”
My take: This study shows how innocuous food can trigger pain after an intestinal infection.
Related blog posts:
- Is There a Way to Prove Which Dietary Factors Trigger IBS?
- Clinically Useful Biomarkers for Irritable Bowel Syndrome? | gutsandgrowth
- IBS Subtypes in Pediatrics | gutsandgrowth
- Mechanisms of irritable bowel syndrome
- Ondansetron for Irritable Bowel with Diarrhea? | gutsandgrowth