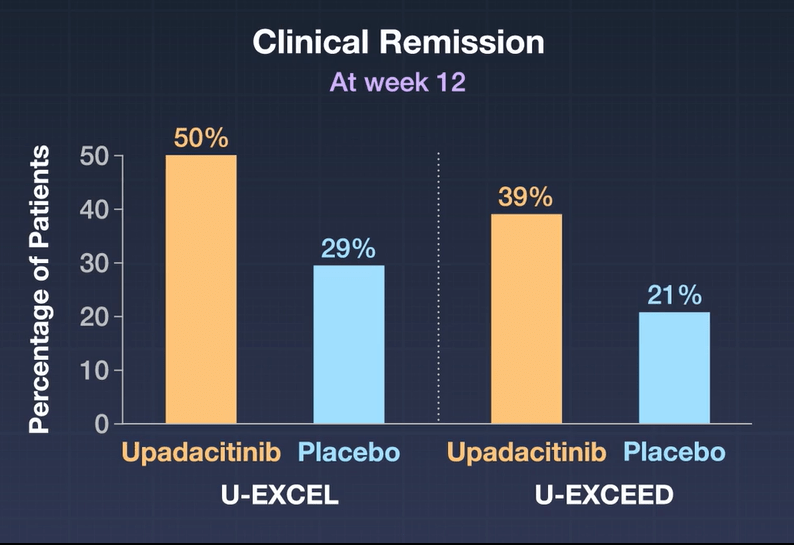
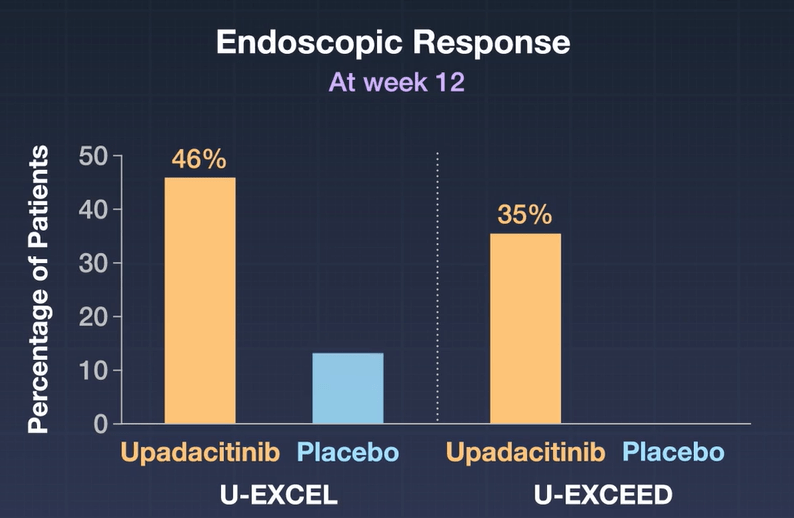
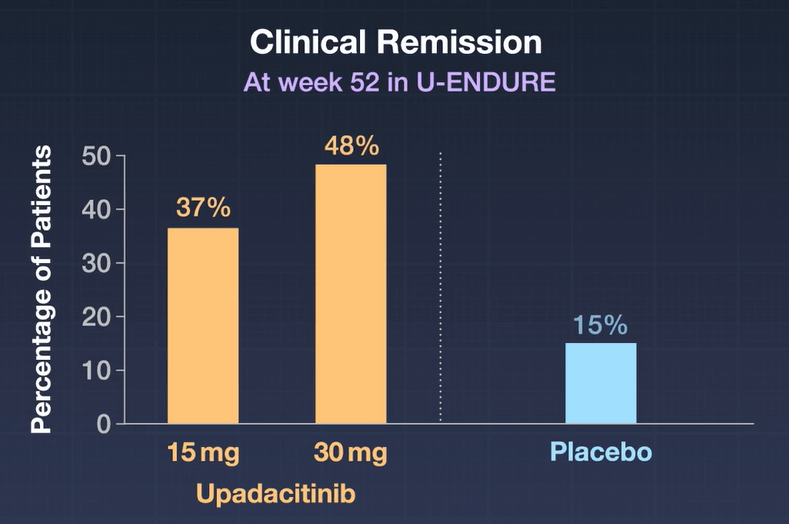
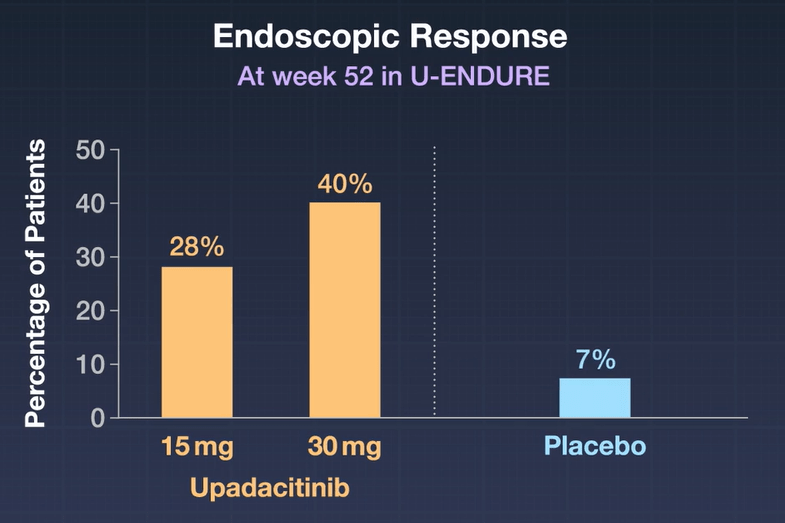
SR Gupta et al. JPGN 2023; 76: 776-781. Outcomes for Standardized Home and Hospital-Based Infusions of Infliximab for Children With Inflammatory Bowel Disease
In this retrospective study with 102 children, key findings:
- There were similar outcomes among carefully-selected children receiving home infusions (HI), “drug durability, AOs [adverse outcomes], and laboratory values were similar between HI and hospital-based infusions.” 30% of eligible patients received HI.
- Within 2 years, only 19% remained on 5 mg/kg every 8 week dosing and the remainder required increased dosing or decreased interval. (Further supporting data showing that 5 mg/kg every 8 week dosing is inadequate in ~80%)
The authors note that HI were arranged with a single home health company with pediatric PALS-trained nurses. In addition, there was “direct communication between the home health nurse and IBD nurse after each infusion.”
Prior studies of HI have shown increased AOs in patients receiving HI including stopping therapy, ER visits, and hospitalizations (Clin Gastroenterol Hepatol 2020; 18: 257-258, Am J Gastroenterol 2020; 115: 1698-1706, JAMA New Open 2021; 4: e2110268).
My take: If set up properly, home infusions could be a reasonable alternative to hospital-based or office-based infusions.
- The Downside of Home Infusion of Biologics
- How Do Home Infusions Stack Up?
- Pediatric Home and Office Biologic Infusions -What is Needed
- Another Study Justifying Higher Infliximab Dosing in Pediatrics
“Denials, Dilly-dallying and Despair” - Kids Are Different: Therapeutic Drug Monitoring
- Modeling Trough Levels to Predict Optimal Infliximab Dosing
- Improving Outcomes with Proactive Therapeutic Drug Monitoring (JAMA 2021 study)
- Can Therapeutic Drug Monitoring with Monotherapy Achieve Similar Results as Combination Therapy?

In this article, from May 31, 2023: Sick Workers Tied to 40% of Food Poisoning Outbreaks, C.D.C. Says
“Each year, 48 million people become sick from a food-borne illness, according to C.D.C. estimates. Of those, 128,000 are hospitalized and 3,000 die.”