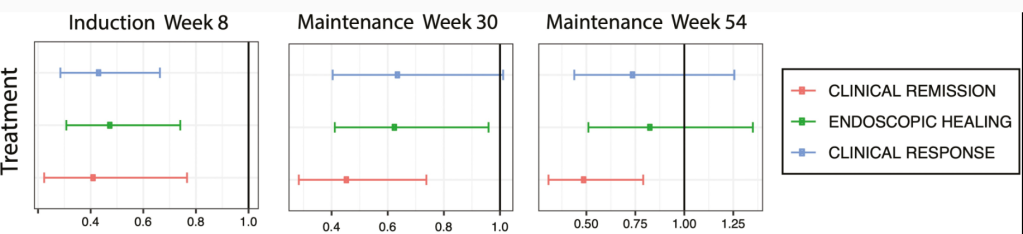
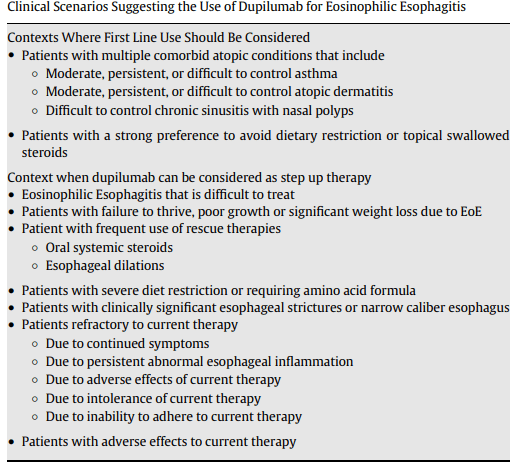
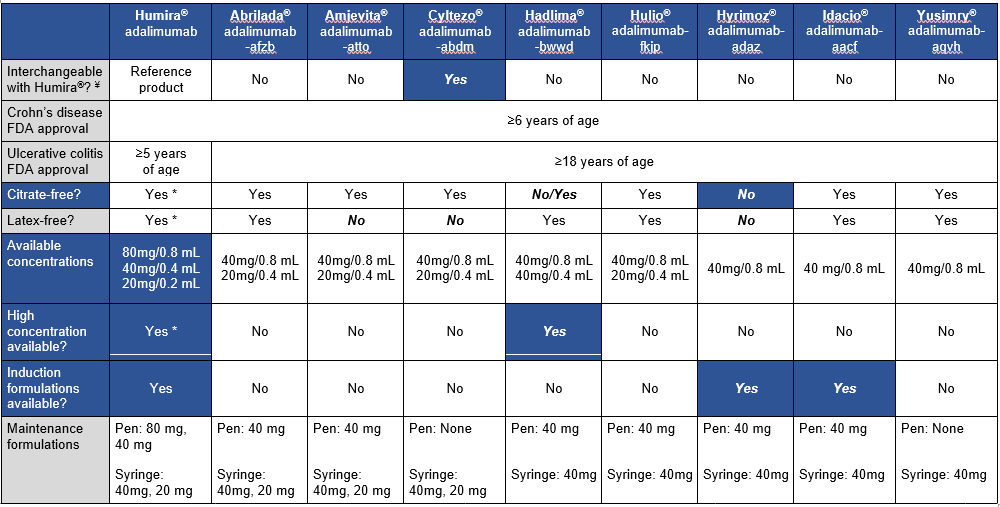
A recent blog post, Airway Impedance to Objectively Assess Airway Mucosal Integrity, reviewed a study by Rosen et al (R Rosen et al. J Pediatr 2023; 256: 5-10) which used a novel approach in assessing airway disease using airway impedance.
This month’s Journal of Pediatrics provides a terrific commentary by my partner, Jose Garza. Open Access: Airway Impedance: In Search of a New Tool (DOI:https://doi.org/10.1016/j.jpeds.2023.01.007).
The article first describes the history of identifying gastroesophageal reflux: “the first attempt to detect GER was in 1884 by Reichman, who lowered a sponge into the esophagus of a patient with heartburn and showed that it contained acid when retrieved. He found that fluid expressed from the sponge was acidic in persons with heartburn and alkaline in normal controls. The first in situ measurement of acid reflux in the esophagus is credited to Tuttle and Grossman 2 in 1958.”
With the development of multi-channel impedance, the concept of reflux has shifted in young children: “non-acid reflux events are more important to the development of symptoms than acid events.”
With regard to Rosen et al, the commentary reiterates that “measuring laryngeal mucosal integrity [using impedance] is safe and feasible in children with extraesophageal symptoms.” The study showed “no correlation between otolaryngology airway inflammation scores with airway impedance, further emphasizing the fact that we need to move away from these scores, as they have failed to show correlation with GERD and with laryngeal mucosal integrity.”
“The authors did find a decrease in airway impedance in patients with history of aspiration; those who aspirated more textures had lower median airway impedance. Therefore, not all airway inflammation is the result of reflux and oropharyngeal dysphagia–associated aspiration can cause GER-like symptoms and airway inflammation. Aspiration can be silent, and if we do not look for it, we are not going to find it.”
“Another group of patients with low airway impedance comprised patients receiving PPIs, which makes it clear that acid is not what is affecting the airway. Otherwise, PPIs would improve airway impedance.”
My take (borrowed from author): “It is not yet clear whether measuring airway impedance will eventually become a widespread test with clinical applicability. However, the concepts advanced in this report are clearly a step in the right direction.”