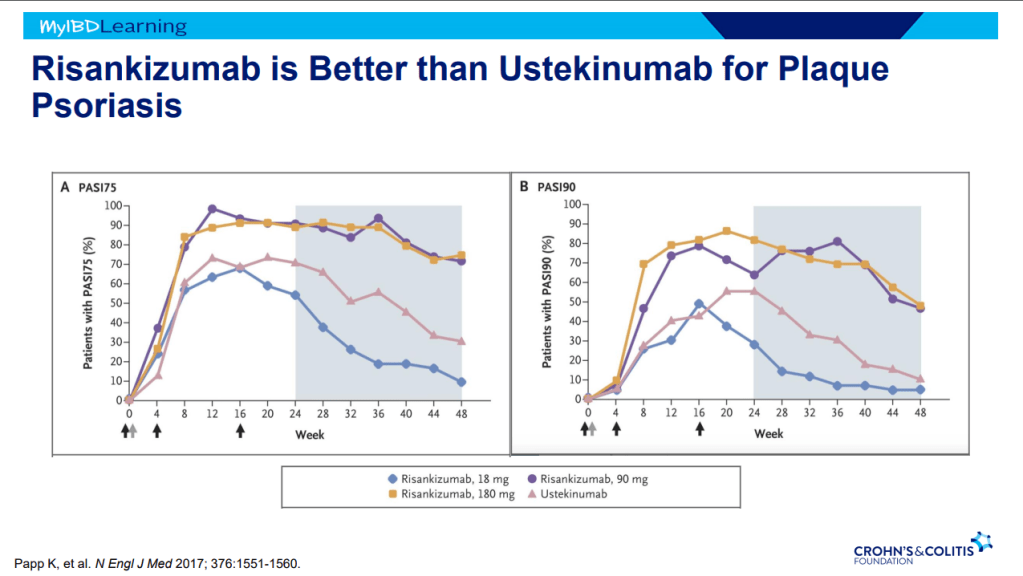
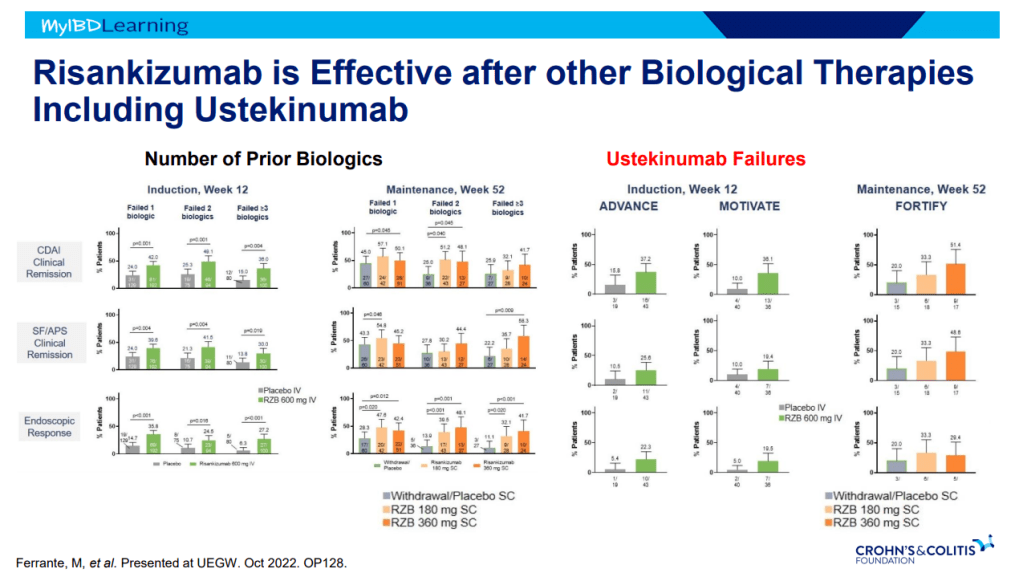
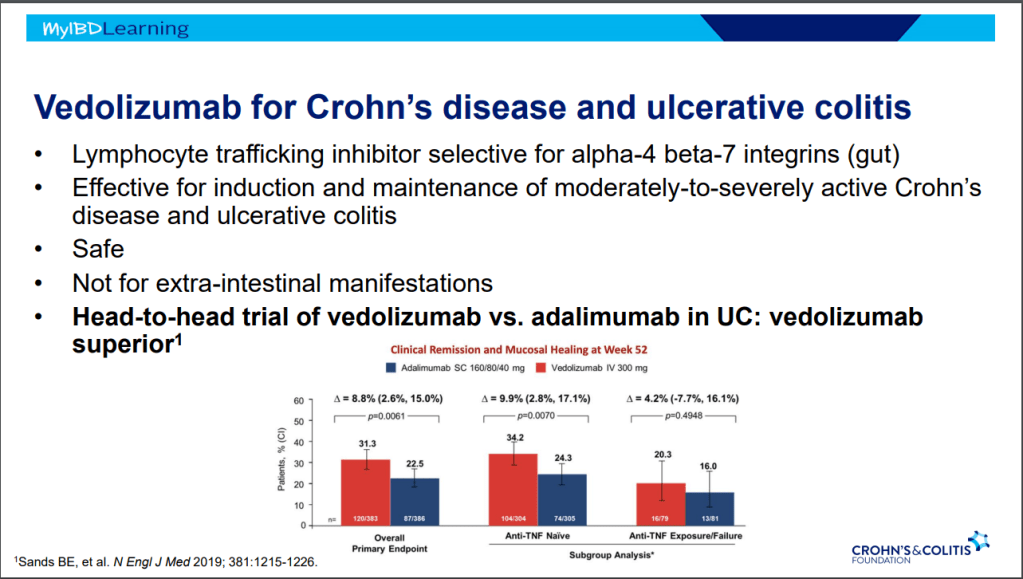
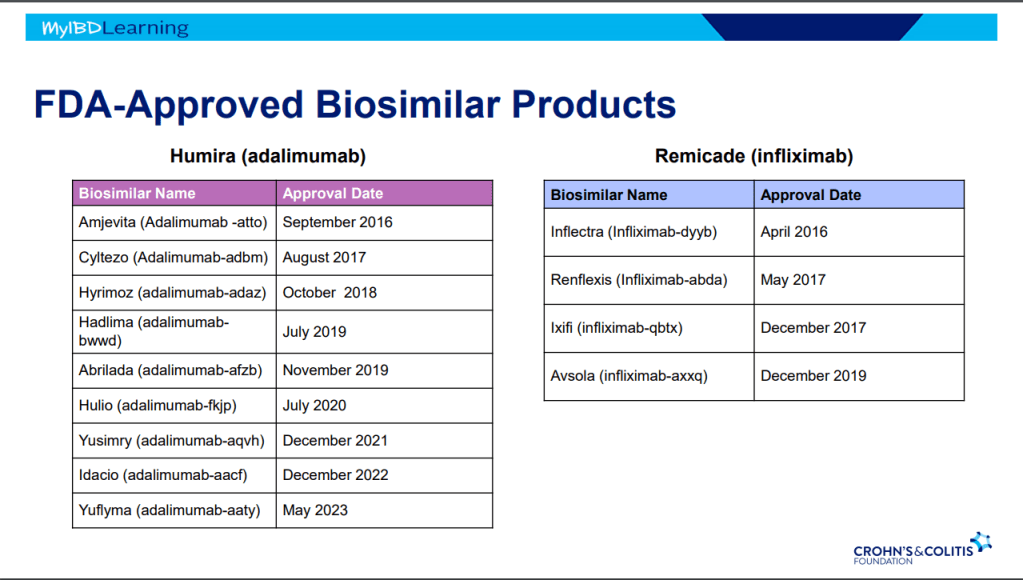
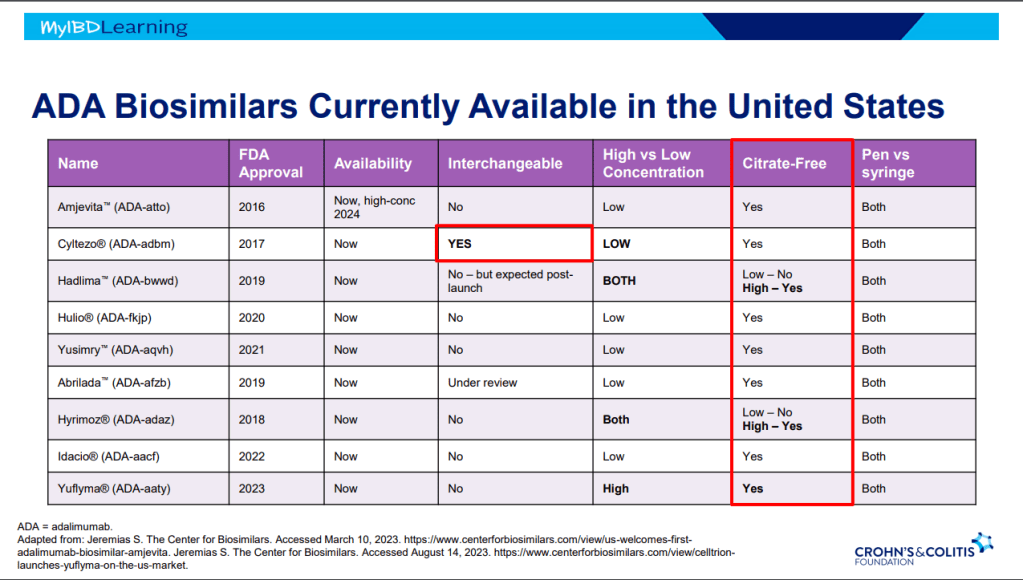
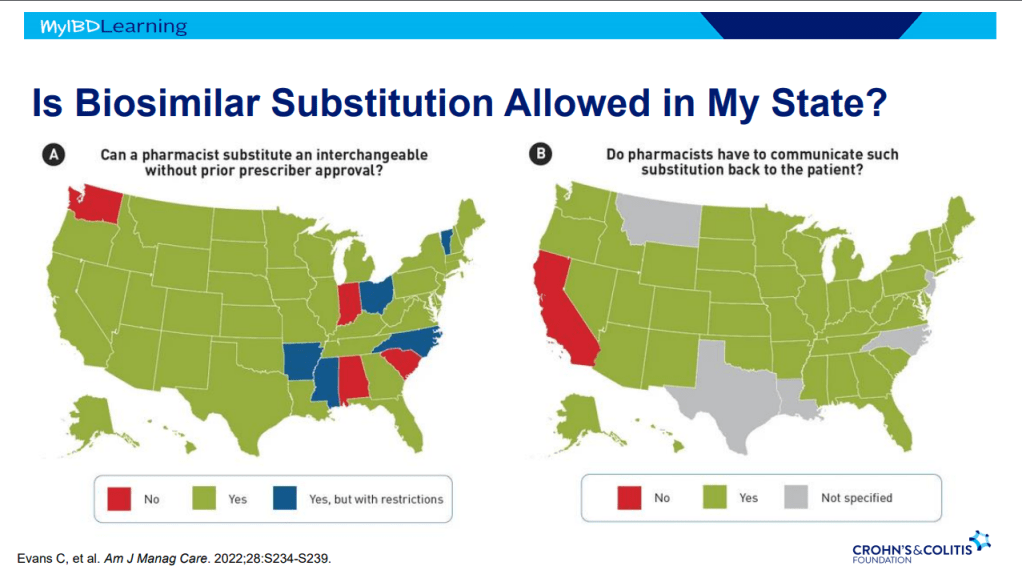
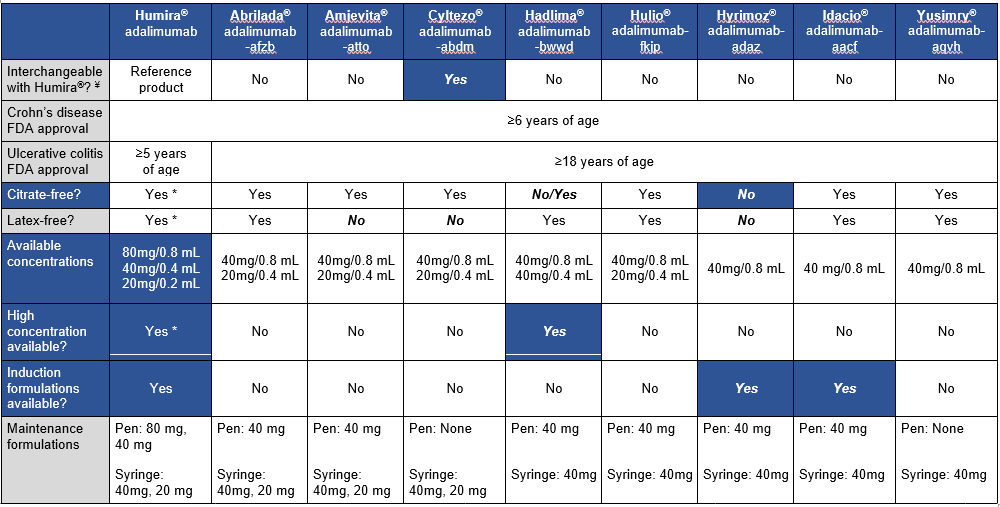
This is third day summarizing some of the talks at the regional CCFA conference. Erin Forster presented on Treatment with Oral Advanced Therapy. Below are my notes and some of the slides; my notes may have errors of omission or transcription. Can get access to full slide set: (n=22) here: Treatment with Oral Advanced Therapy
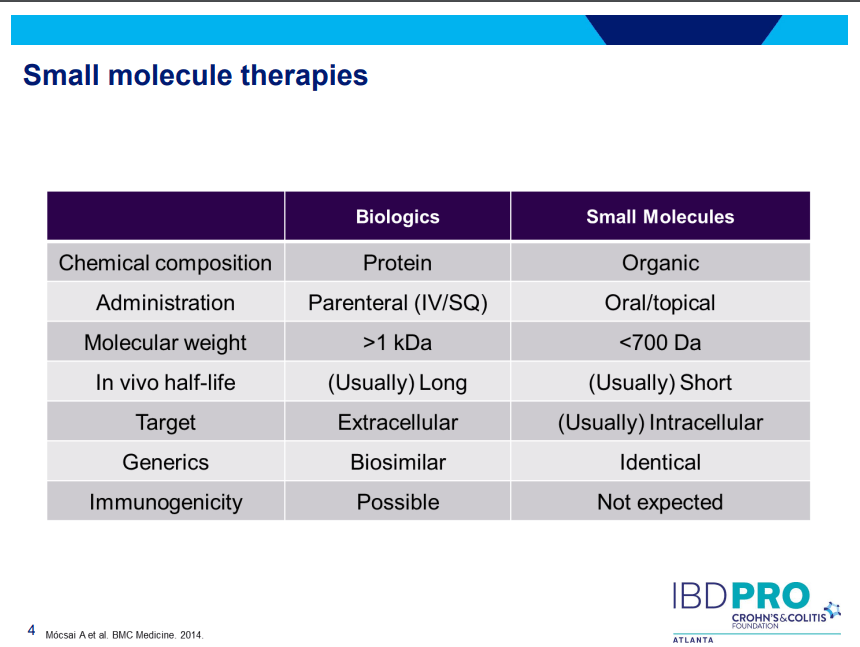
- JAK inhibitors (Tofacitinib, Upadacitinib) have rapid onset of action and are taken orally
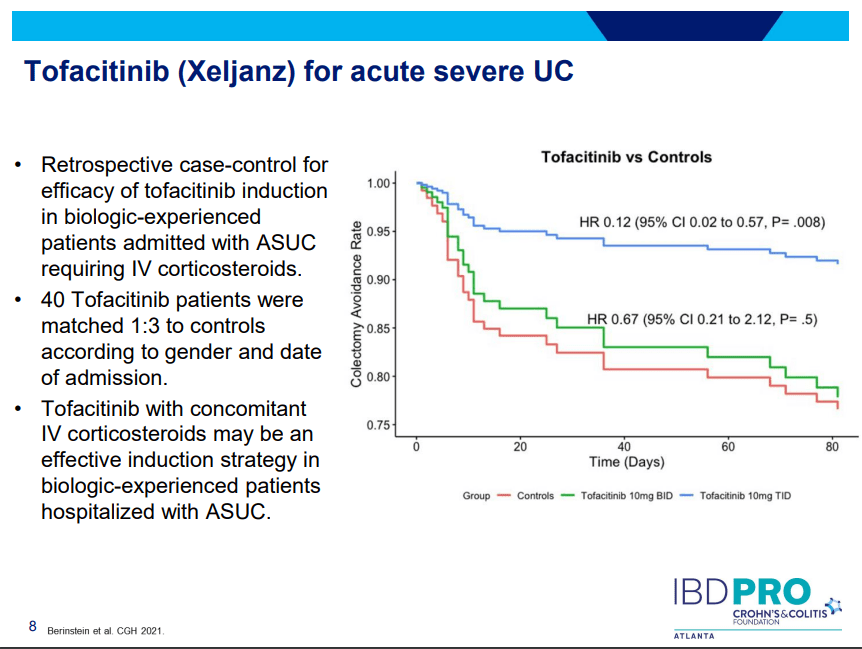
- Tofacitinib (Xeljanz) -concern about cardiovascular events was derived from elderly rheumatologic patients. Cardiovascular events are rare. Higher dose (TID) (in the hospital) associated with lower colectomy rates in acute severe ulcerative colitis.
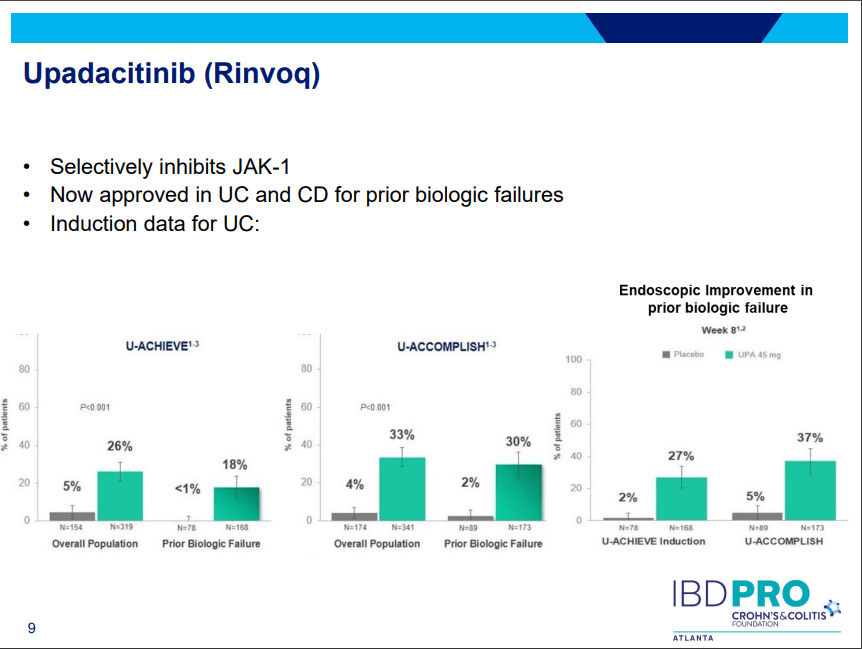
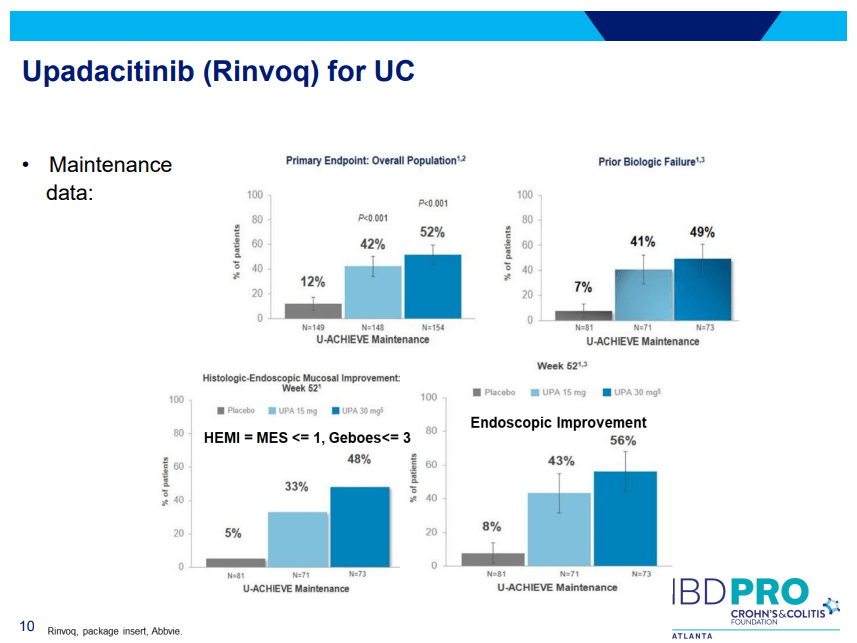
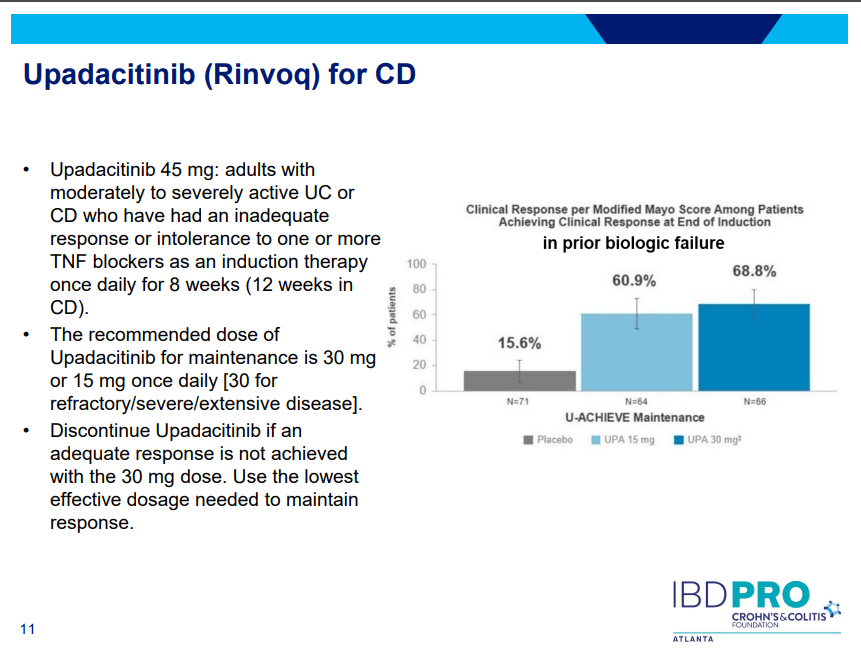
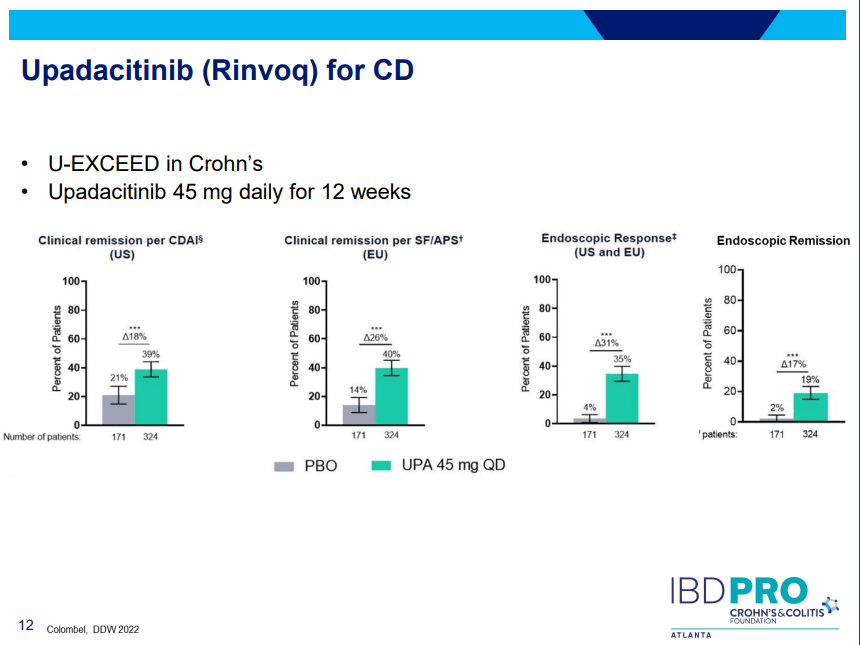
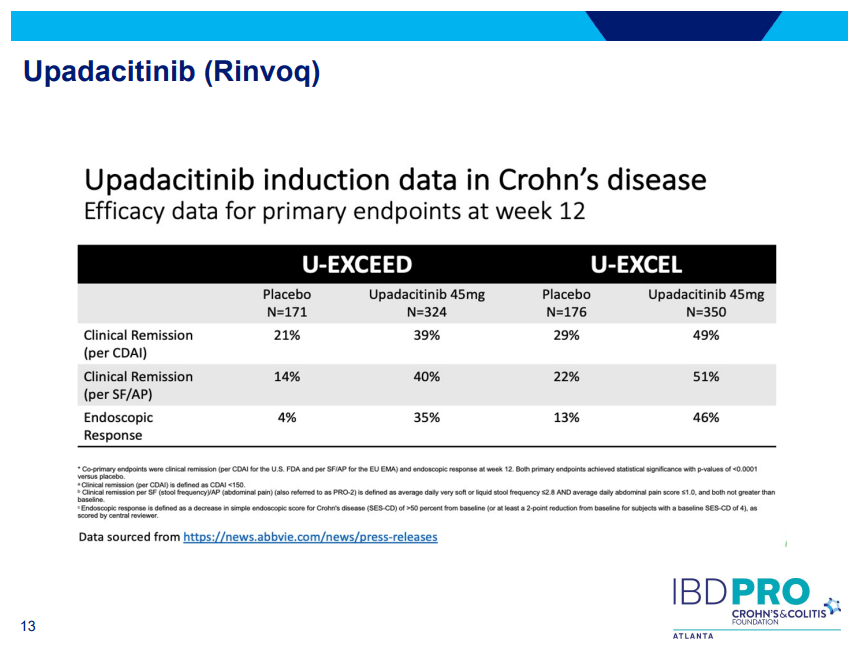
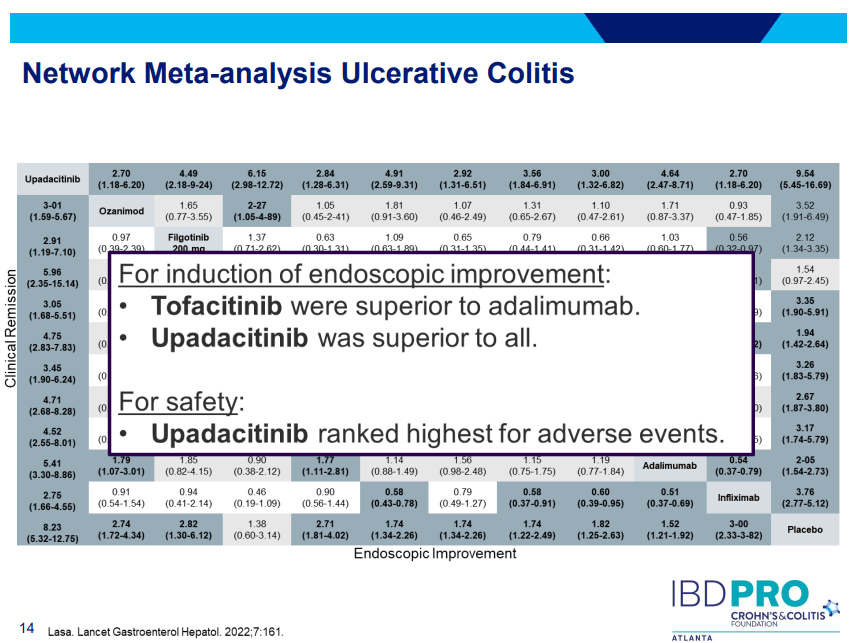
- Upadacitinib (Rinvoq) -now approved for CD and UC. Higher dosing could affect liver function (especially if underlying liver disease). Also, JAK inhibitors as a class have similar safety concerns: increased herpes zoster and concerns for cardiovascular concerns (esp if >50 years)..
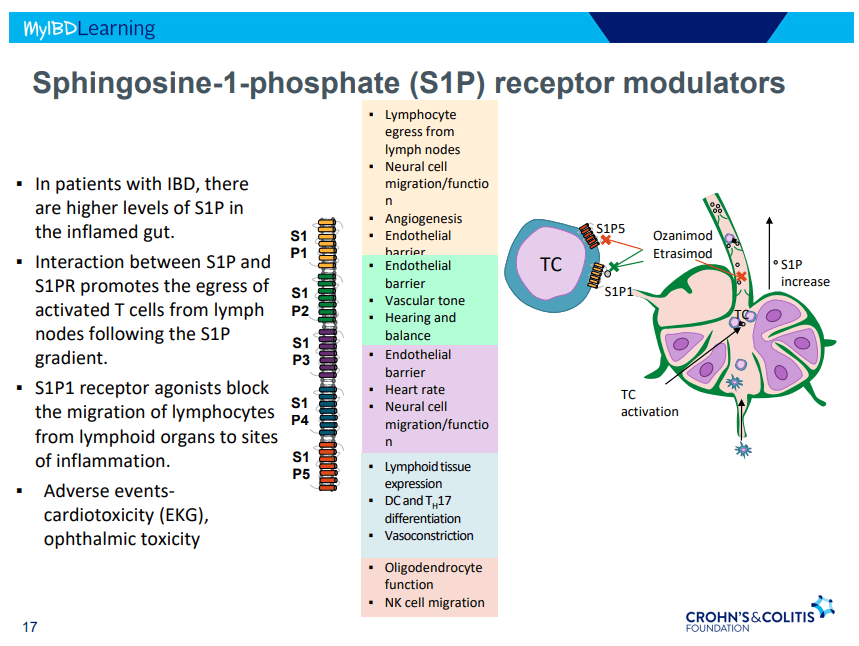
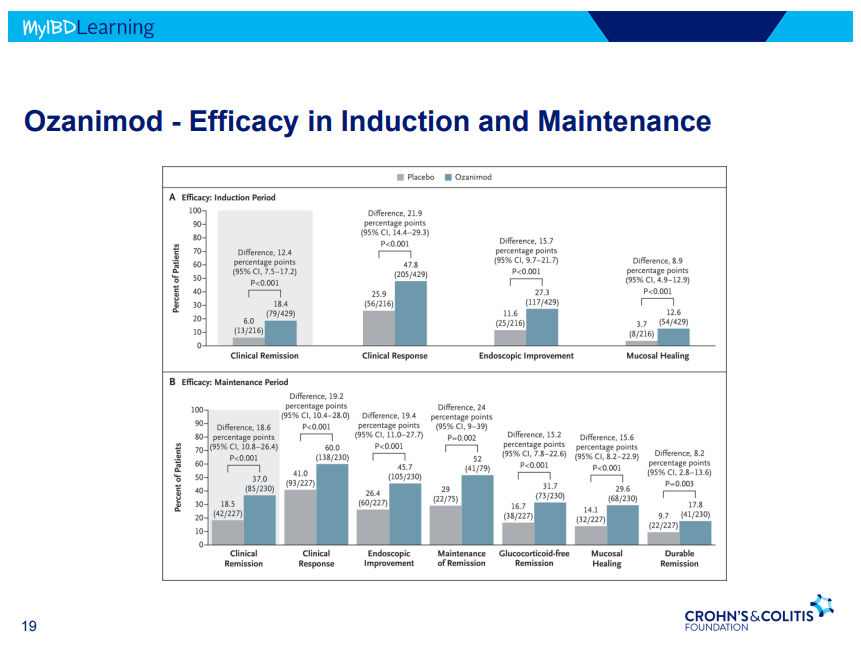
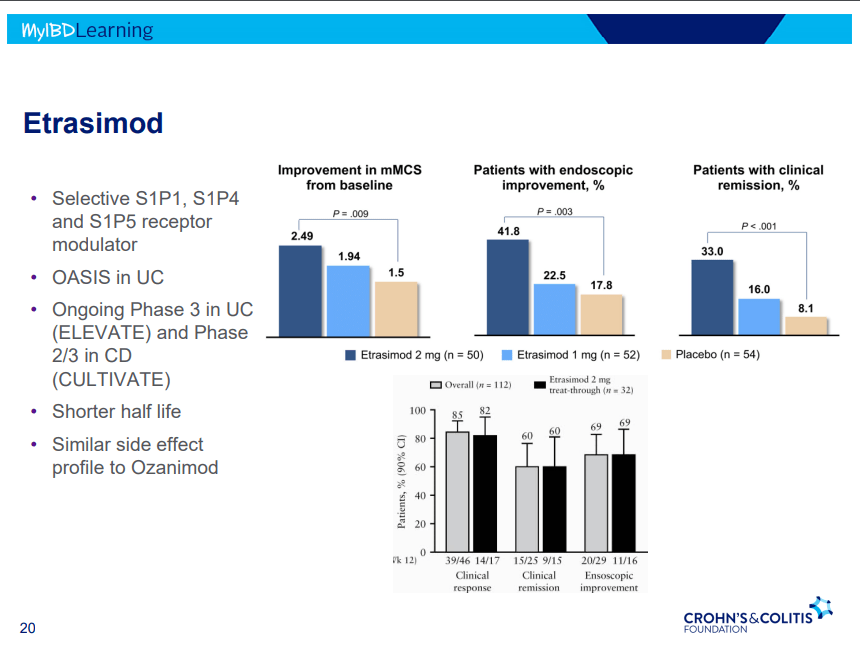
- S!P receptor modulators: Oznaimod, Etrasimod & Amiselimod. Can cause bradycardia -have to check EKG prior.
- None of the oral agents are safe in pregnancy