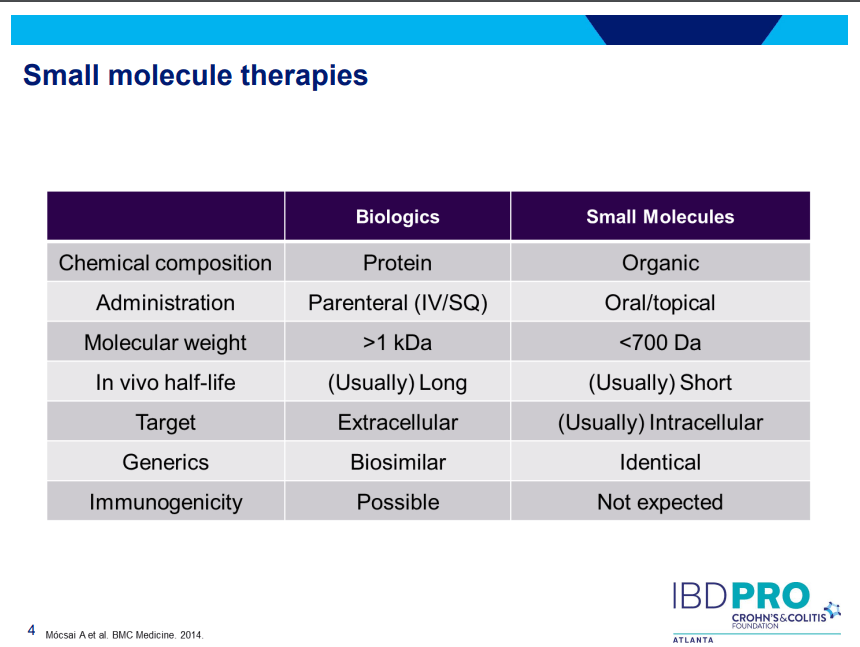
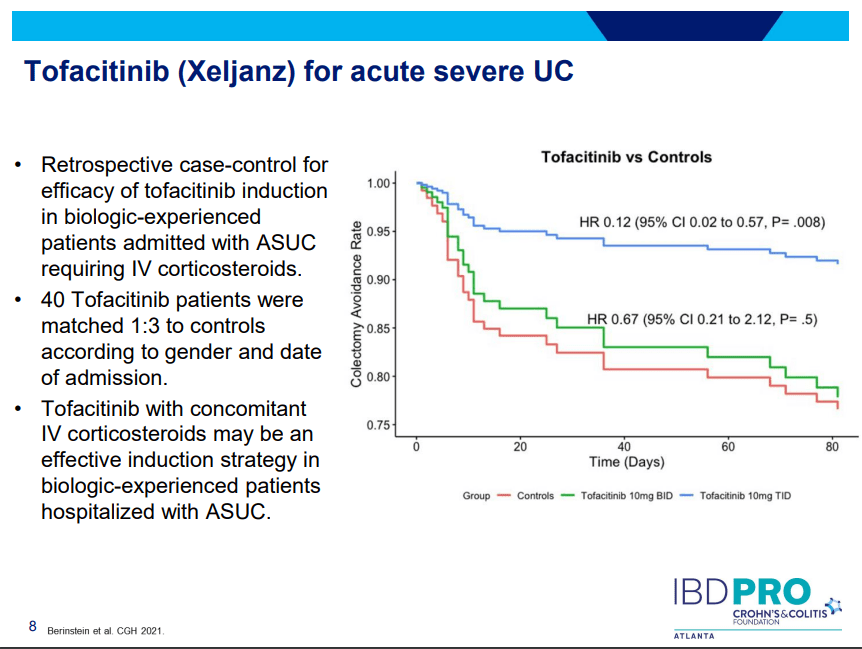
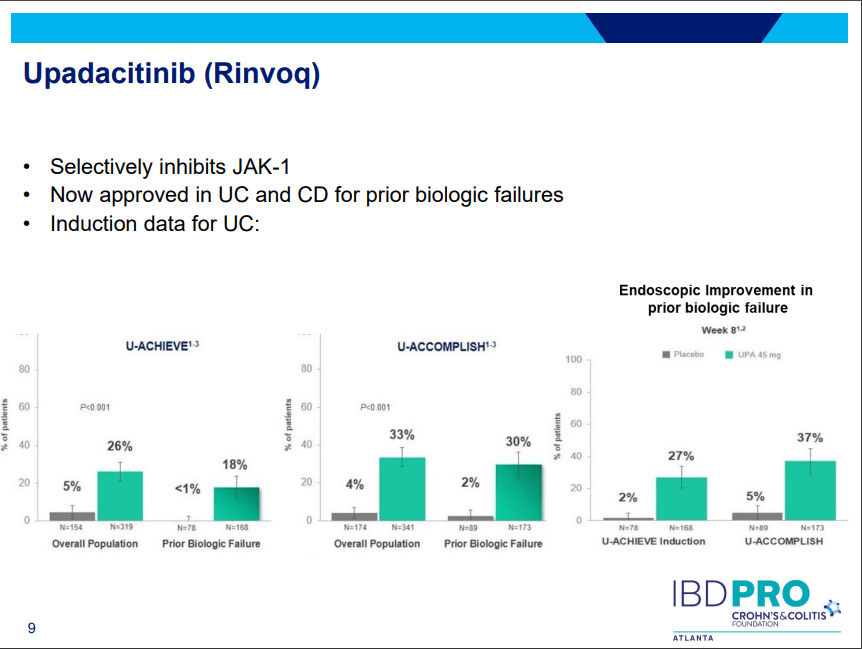
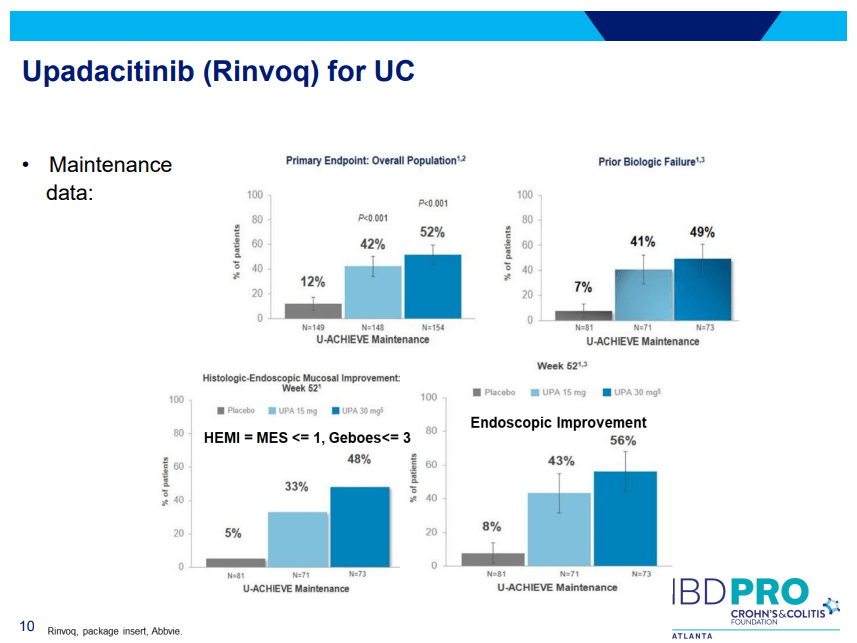
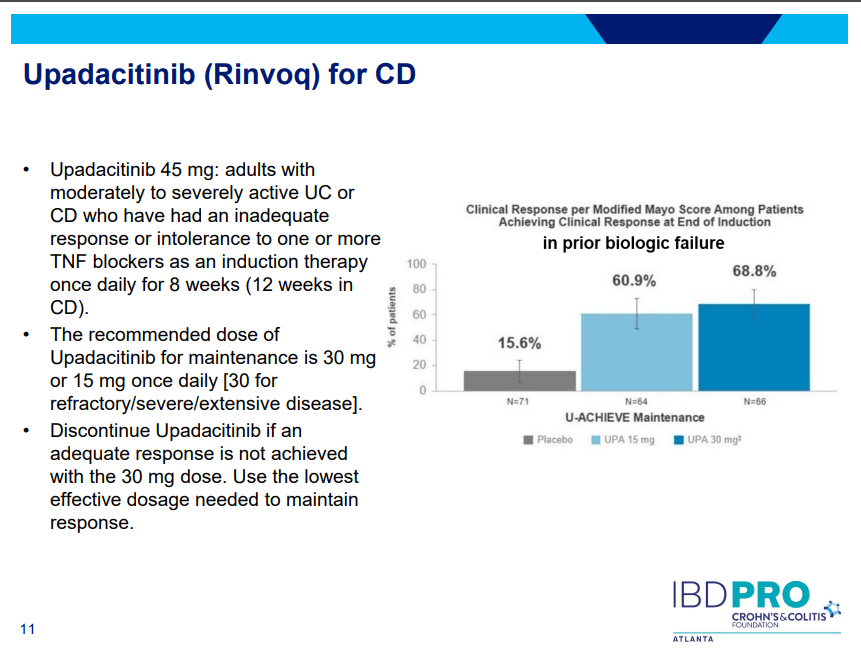
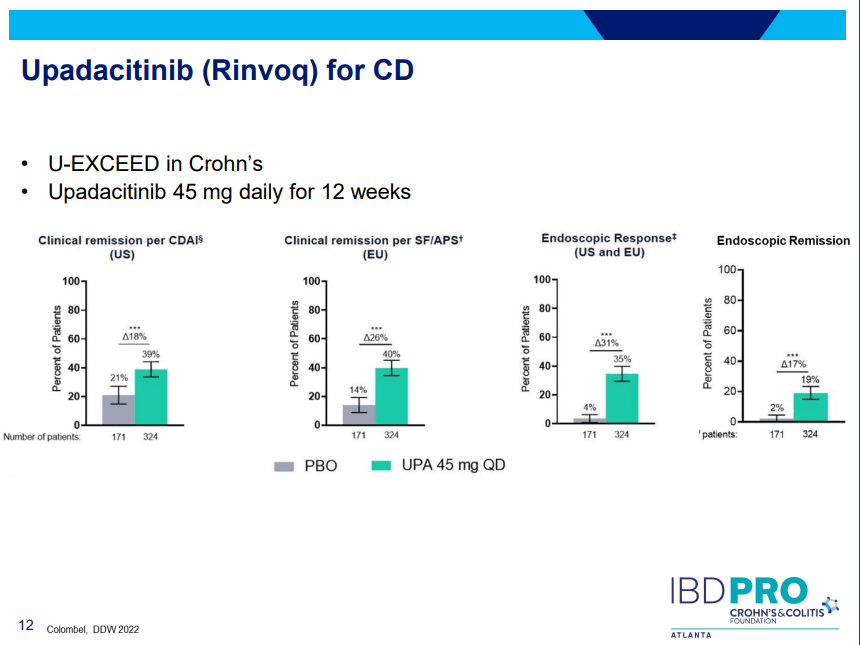
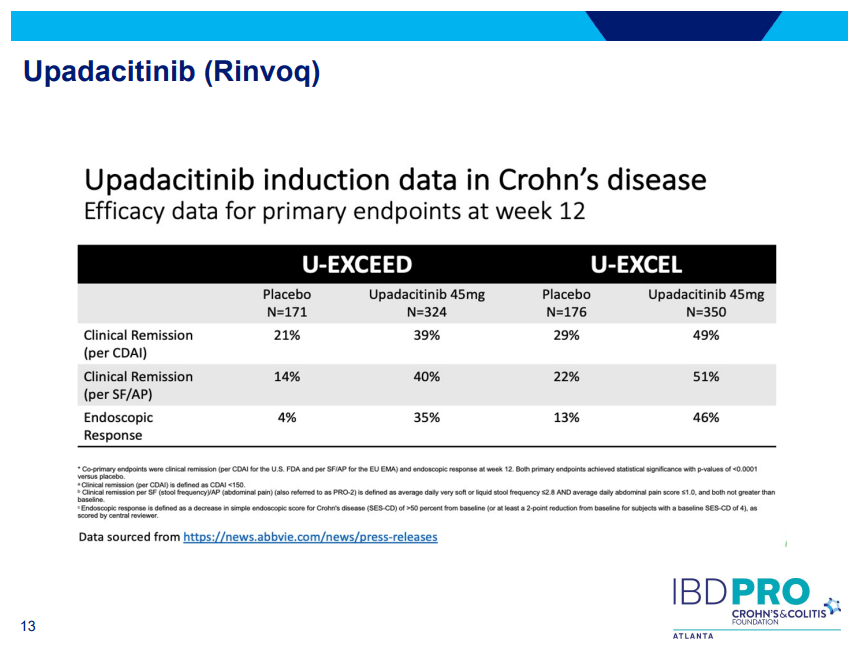
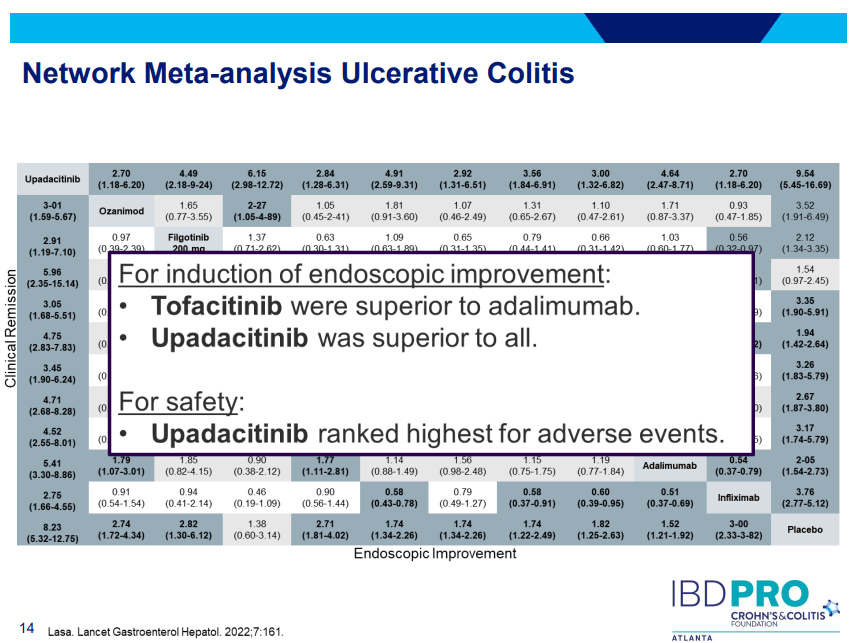
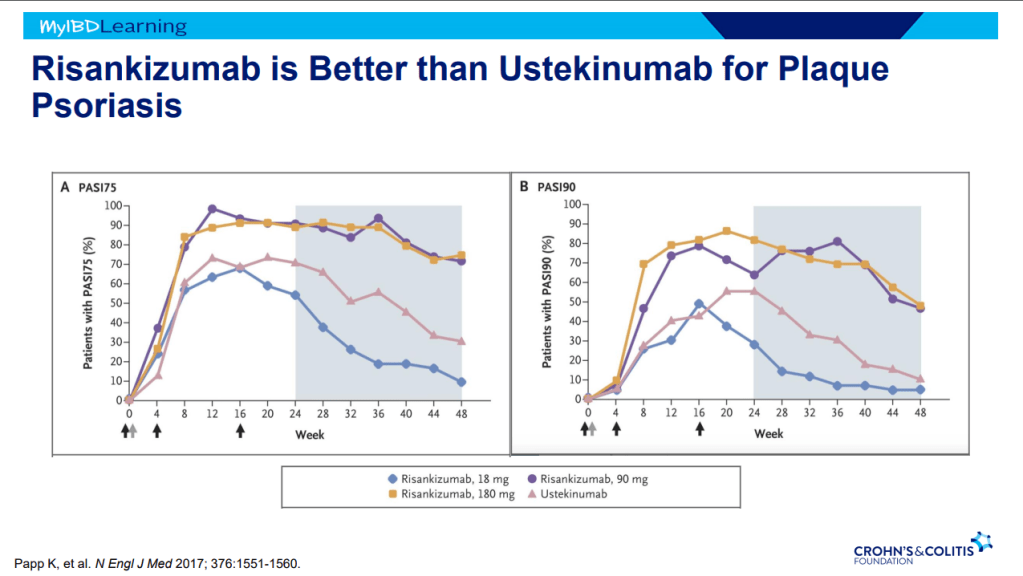
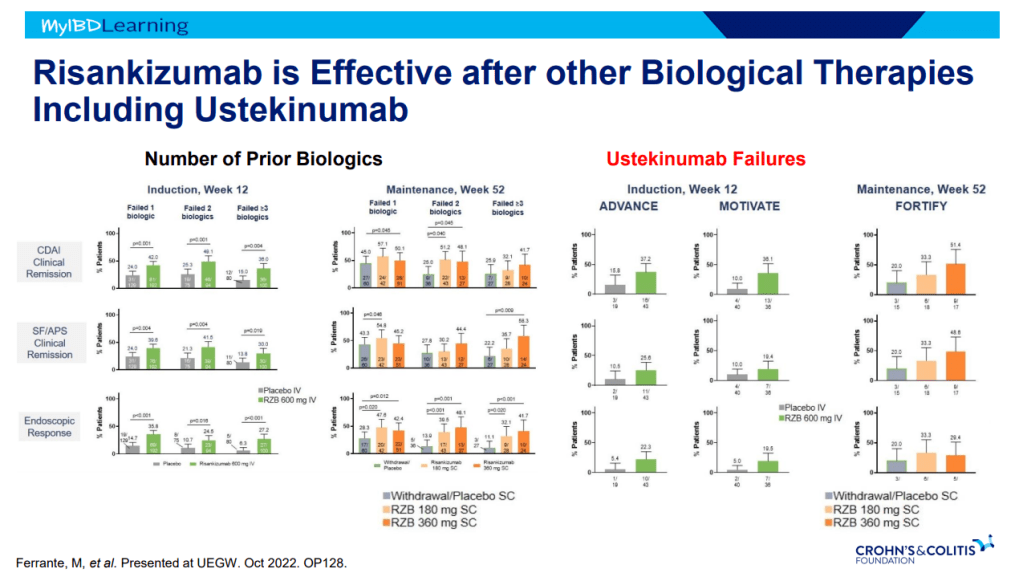
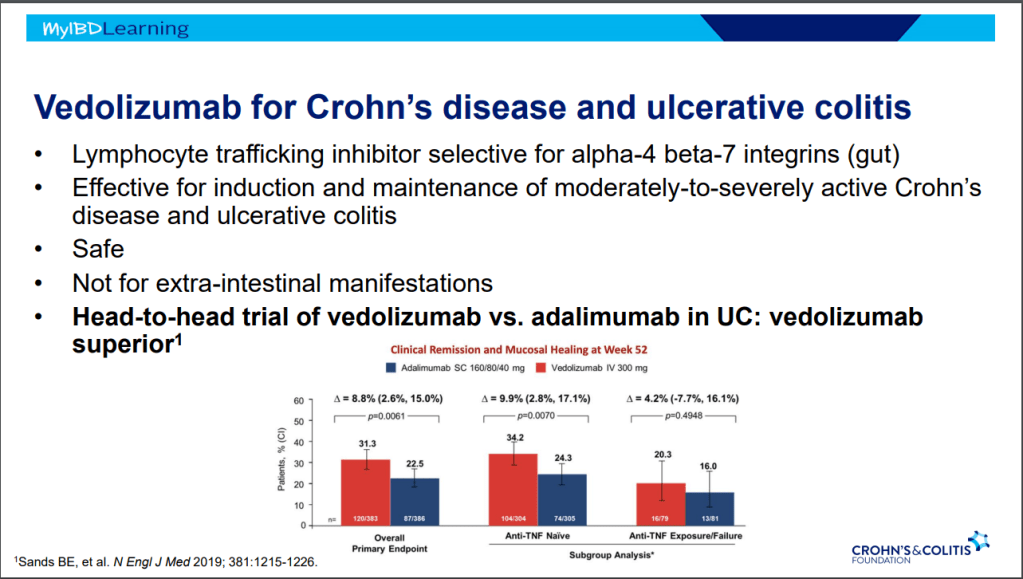
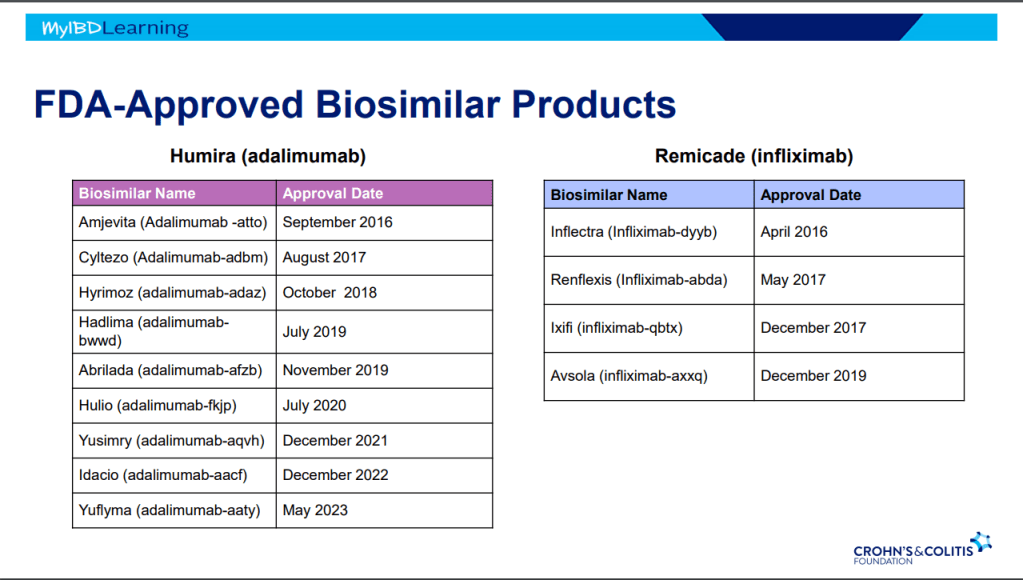
Another very good review by David Schwartz on The Daunting Duo: Management of Stictures and Fistula. Below are some of my notes and some slides; my notes may contain errors in transcription or omission. Can get access to all 49 slides here: IBD Pro CCFA 2023 Atlanta
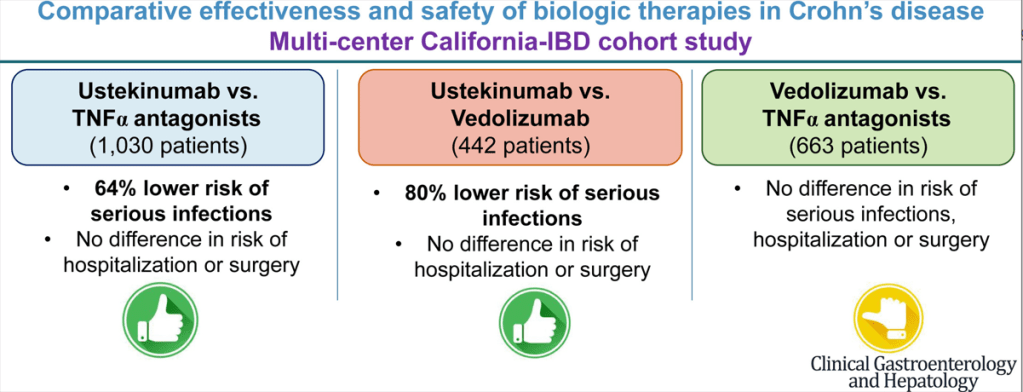
- Fistula healing rates are poor
- 78% have complex fistulas
- Long-term healing: 67% with simple fistulas but only 37% with complex fistulas
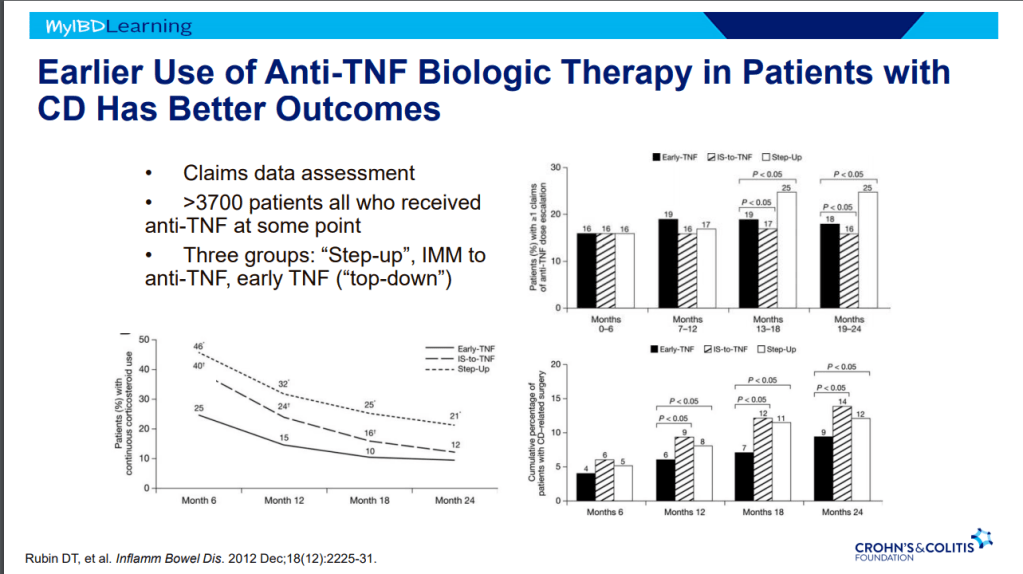
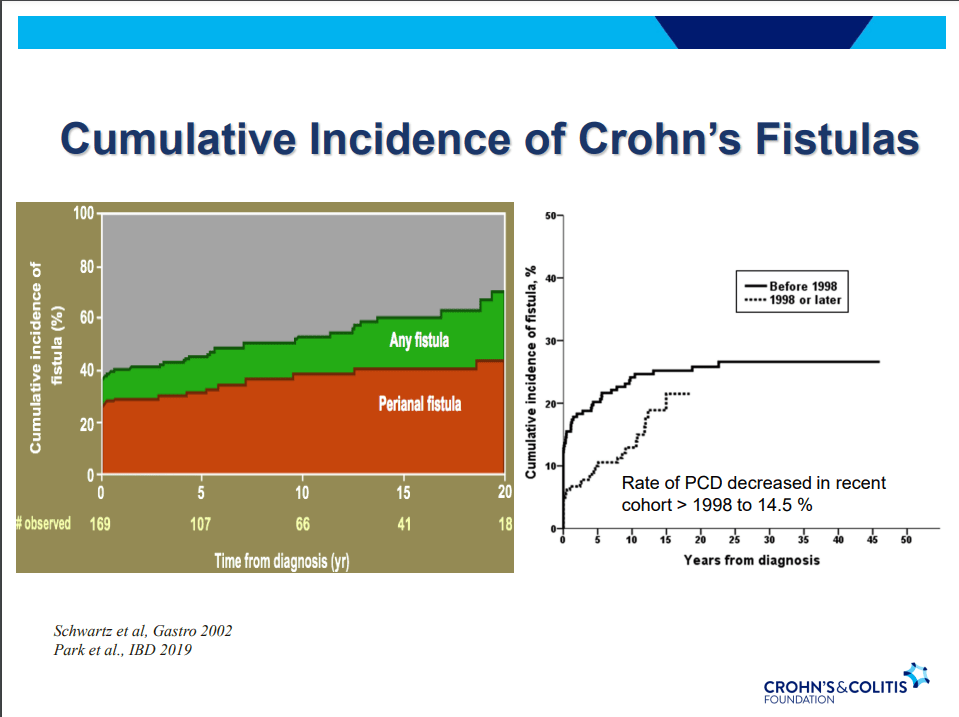
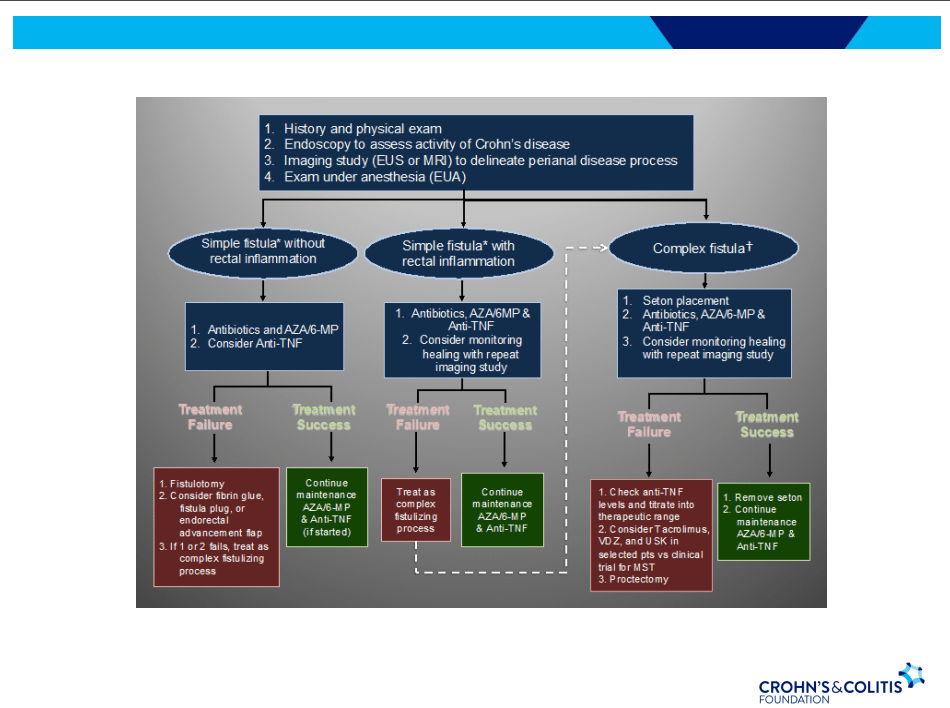
- Seton placement prior to biologic therapy increases likelihood of better outcomes
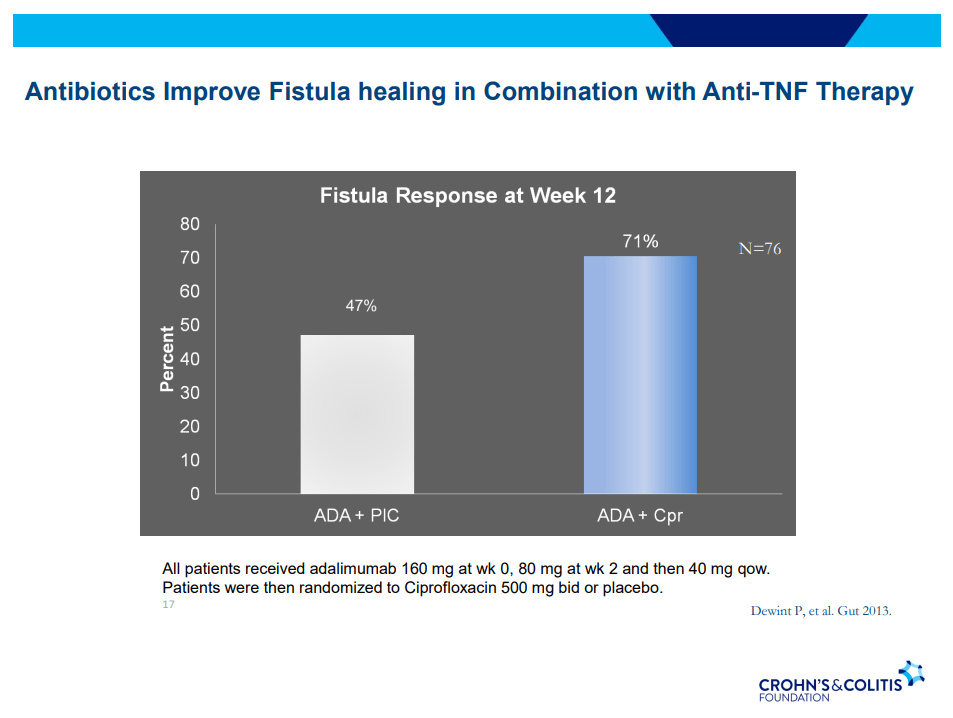
- Antibiotic therapy recommended until Seton removed (not short-term treatment) –improved healing rates along with medical therapy
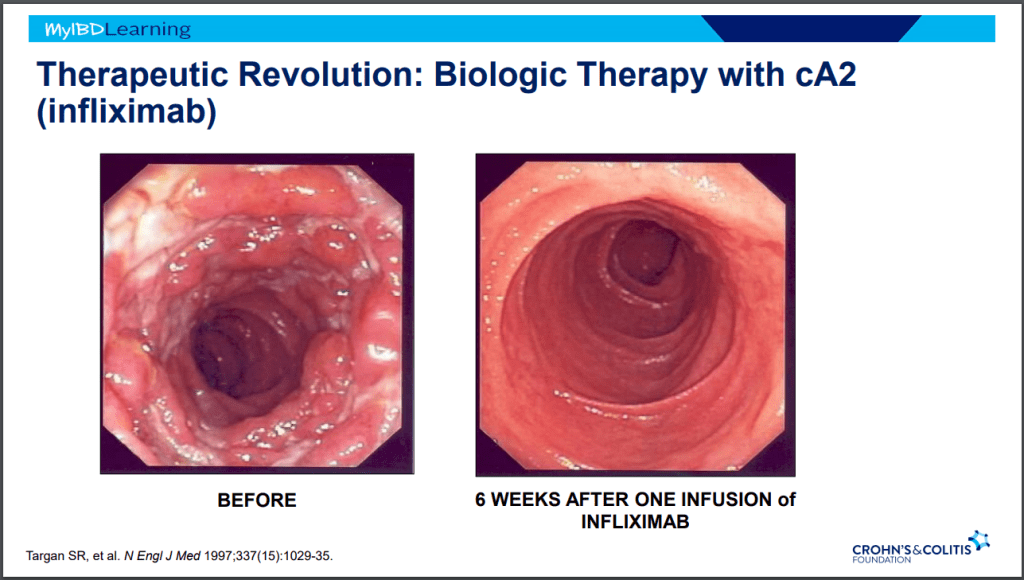
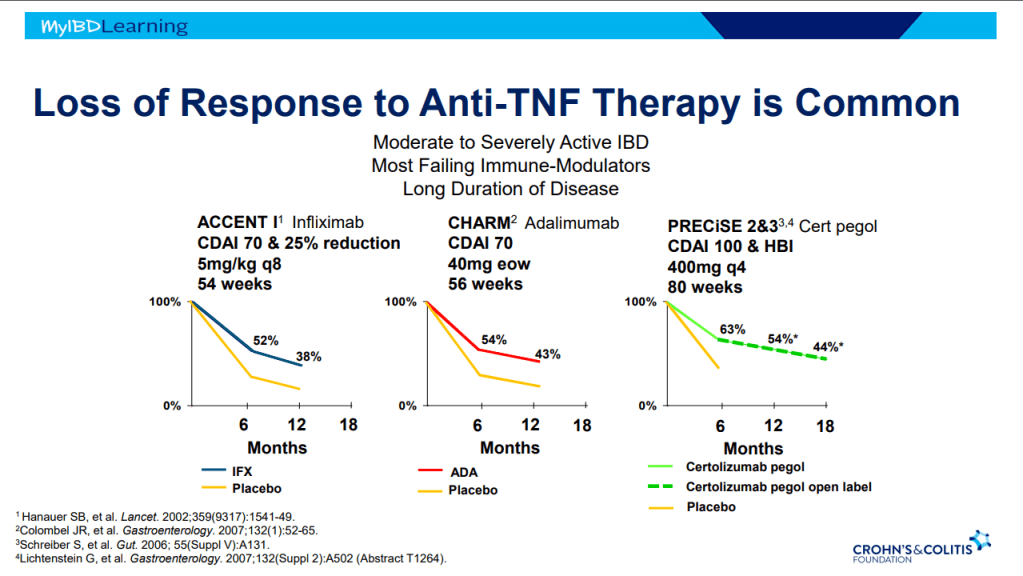
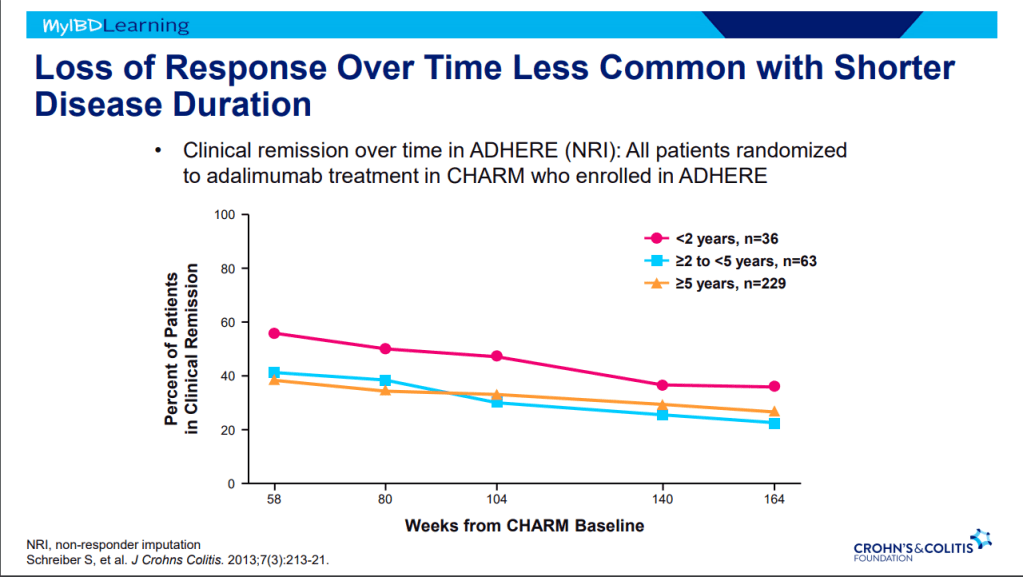
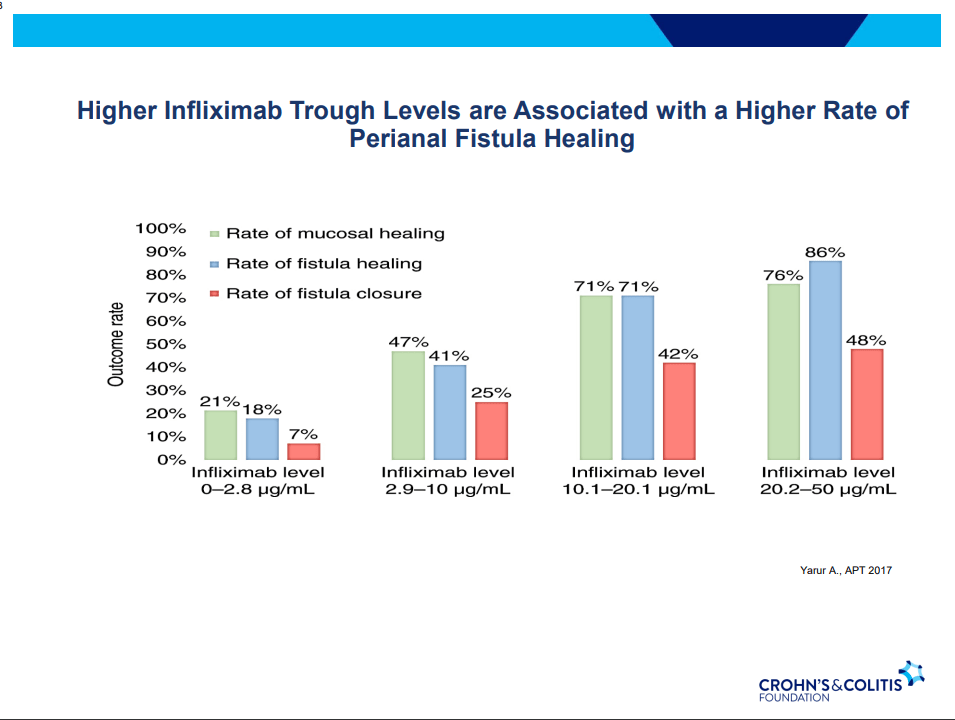
- Anti-TNF therapy effective in ~40% long-term; higher rates of healing with higher anti-TNF levels
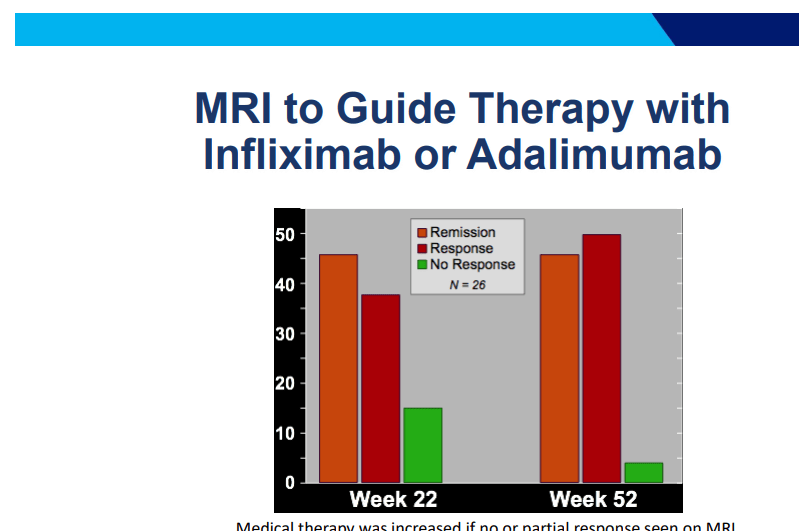
- MRI and/or EUS helpful in improving fistula healing rates
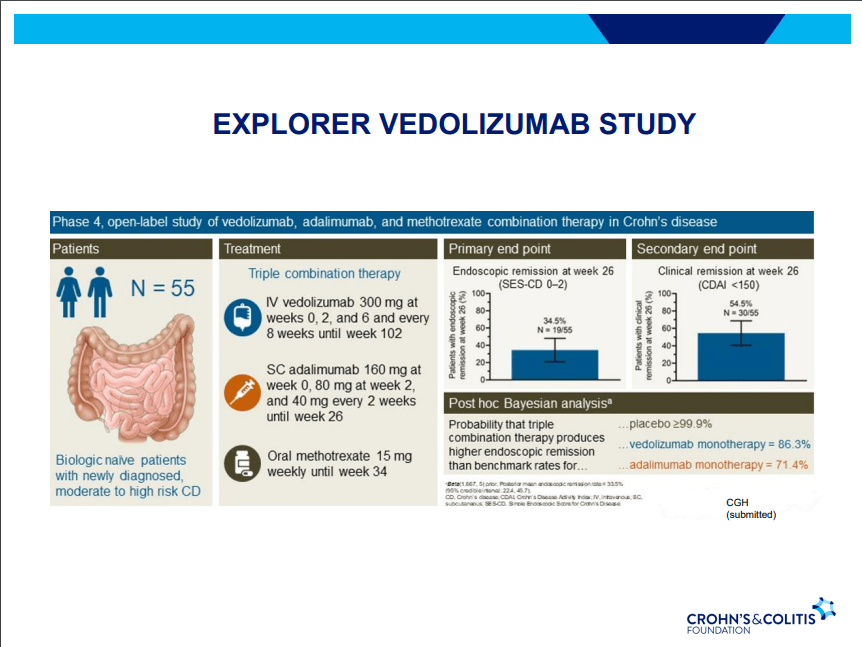
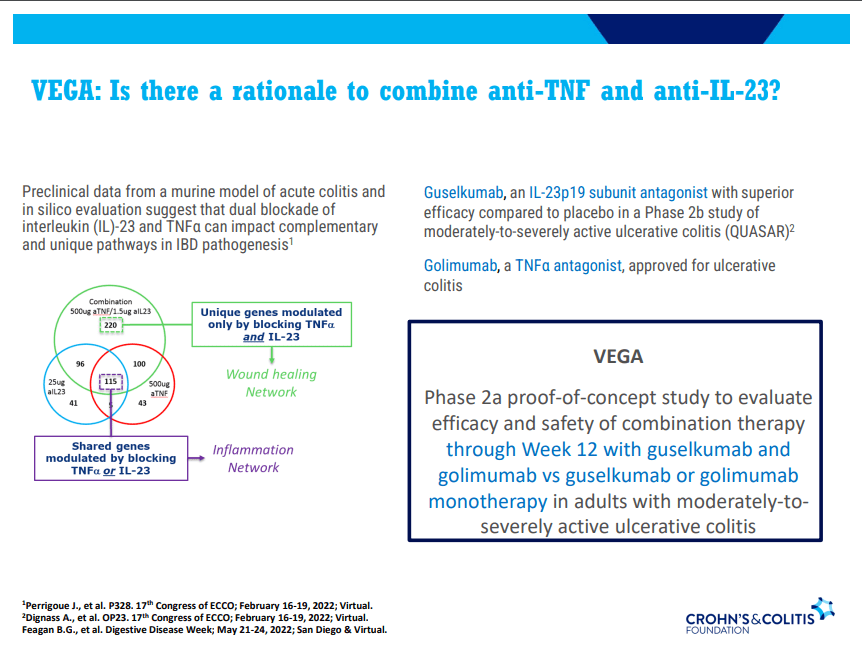
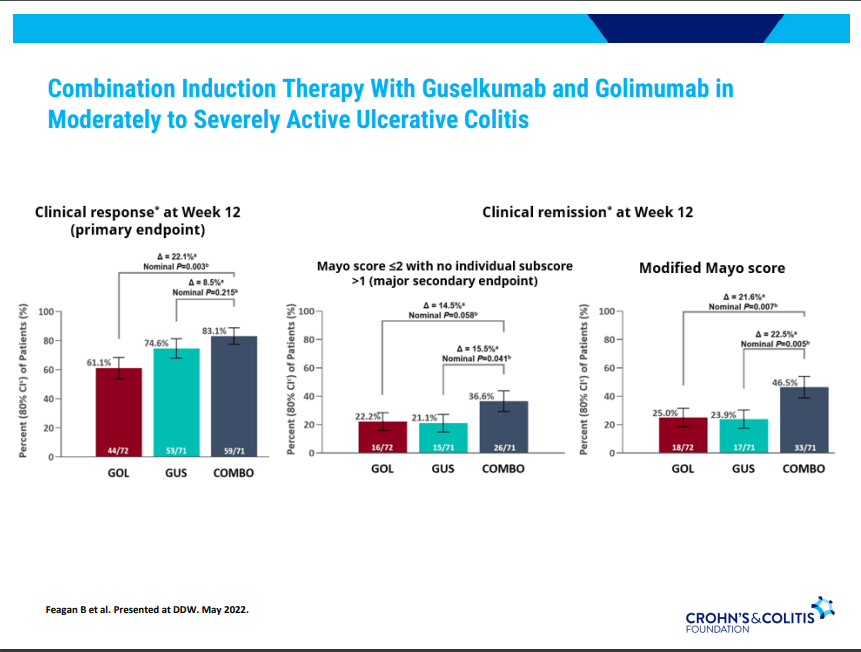
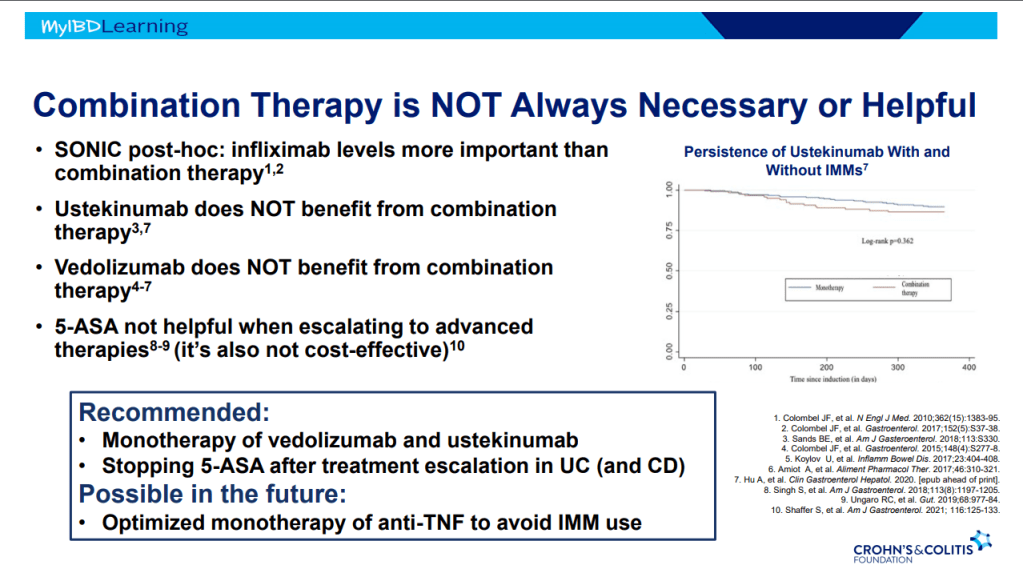
- Ustekinumab and Vedolizumab both had fistula healing rates ~40%
- Adipose derived stem cells with ~50% healing rates (study with high placebo healing rate too ~37%); changes local cytokine profile, cells gone in about 2 weeks but goal for changing trajectory



Strictures
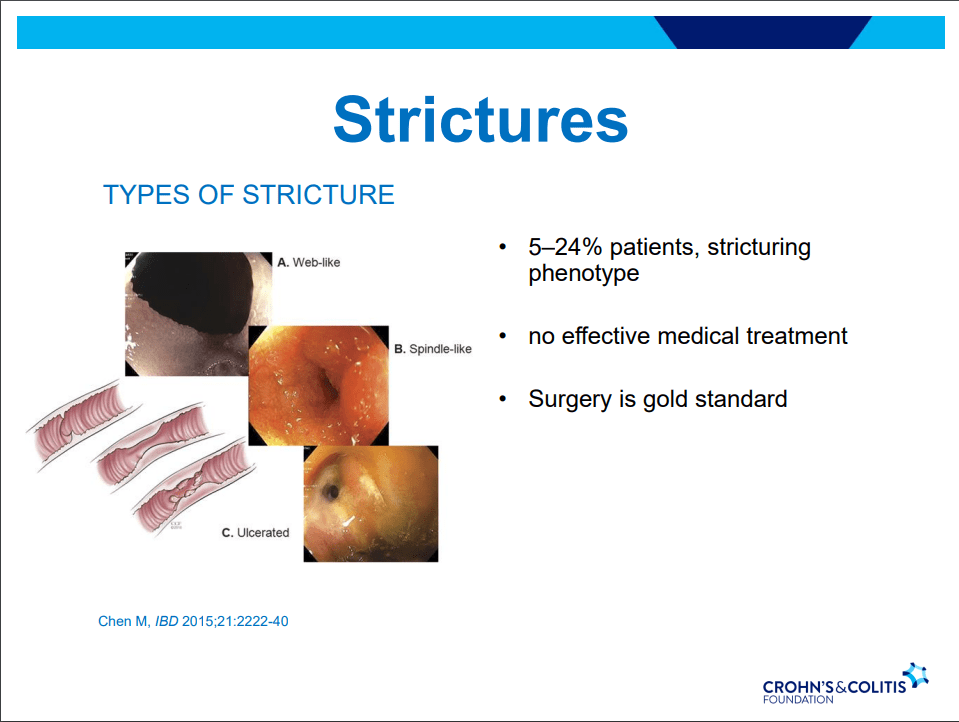
- 5-24% with stricturing phenotype
- No effective medical treatments
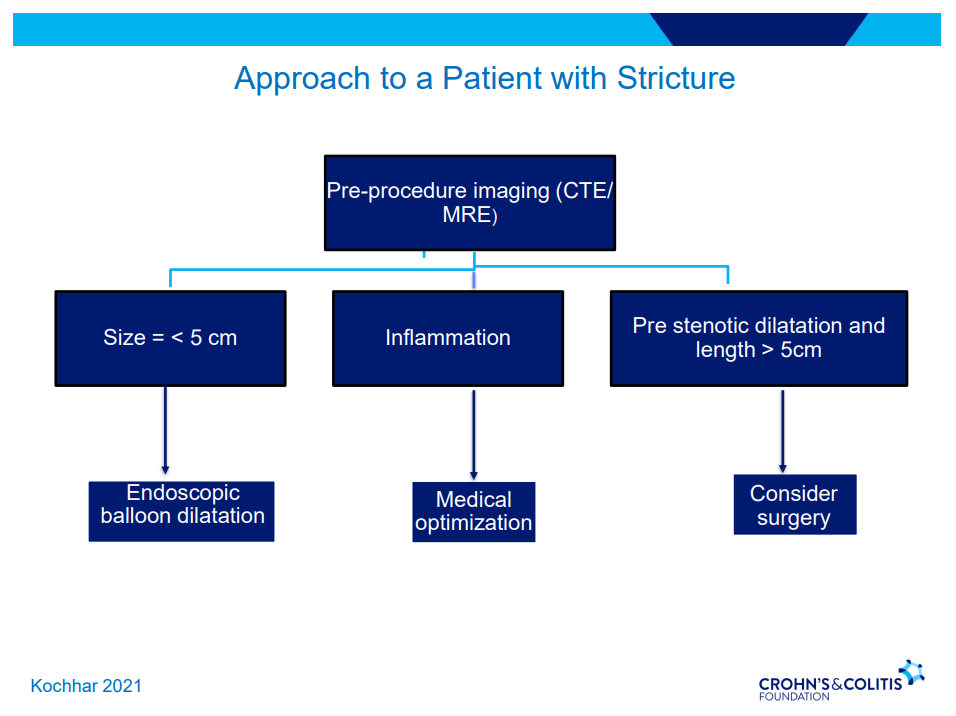
- Endoscopic balloon dilatation in shorter strictures (<5 cm) without associated abscess or fistula (needs imaging prior)
- One-third of balloon dilated strictures will still wind up needing surgery despite dilatation. 5-10% risk of complication. Goal is 15 mm or more (Dr. Schwartz typically does not increase by more than 3 sizes from baseline in one session)




Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.