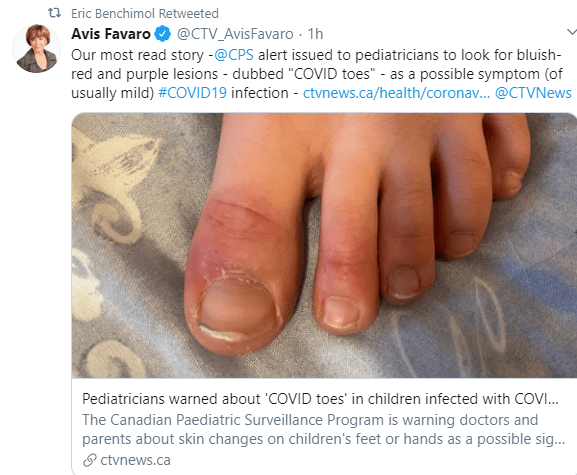
NEJM: Children with Covid-19 in Pediatric Emergency Departments in Italy
Key points:
- Children younger than 18 years of age who had Covid-19 composed only 1% of the total number of patients; 11% of these children were hospitalized, and none died
- The Coronavirus Infection in Pediatric Emergency Departments (CONFIDENCE) study involved a cohort of 100 Italian children younger than 18 years of age with Covid-19 (median age 3.3 years)
- .Common symptoms were cough (in 44% of the patients) and no feeding or difficulty feeding (in 23%) (especially if <2 years)
- Fever, cough, or shortness of breath occurred in 28 of 54 of febrile patients (52%)
- Of the 9 patients who received respiratory support, 6 had coexisting conditions
My take: This study provides additional data indicating that severe outcomes are rare in children with Covid-19.
Related article from NY Times: How Coronavirus Mutates and Spreads
An excerpt:
Researchers have found that the coronavirus is mutating relatively slowly compared to some other RNA viruses, in part because virus proteins acting as proofreaders are able to fix some mistakes. Each month, a lineage of coronaviruses might acquire only two single-letter mutations.
In the future, the coronavirus may pick up some mutations that help it evade our immune systems. But the slow mutation rate of the coronavirus means that these changes will emerge over the course of years.
That bodes well for vaccines currently in development for Covid-19. If people get vaccinated in 2021 against the new coronavirus, they may well enjoy a protection that lasts for years.
Related blog posts:
- NY Times: How Will We Know When to Reopen the Country? & Timely Tweets
- Quietly testing famotidine for COVID-19
- COVID-19 Projections -IHME Data IHME Link: IHME Website for COVID-19 This post details the projected needs (ventilators, ICU beds) and projected mortality. Currently, peak of this pandemic in U.S. is anticipated to be April 16th.
- How to Do a Colonoscopic Polypectomy and U.S. COVID-19 Tracker NPR: Map: Tracking The Spread Of The Coronavirus In The U.S This tracker details the pandemic in every state.
- How to Protect Healthcare Workers from COVID-19 -Lessons from Hong Kong and Singapore Atul Gawande has a very pertinent article in the New Yorker: Keeping the Coronavirus from Infecting Health-Care Workers
- What is the Current Standard of Care for PPE and Endoscopy Cases? Link to manuscript: COVID-19 in Endoscopy: Time to do more?
- Bill Gates: What We Need to Do Now for COVID-19, False-negative testing & Article Describing 3 Stages of Infection From NY Times: If You Have Coronavirus Symptoms, Assume You Have the Illness, Even if You Test Negative Bill Gates: Here’s how to make up for lost time on covid-19 & article describing 3 stages of infection: COVD-19 Illness in Native and Immunosuppressed States: A Clinical-Therapeutic Staging Proposal
- Allocating Scarce Resources During COVID-19 Pandemic Links CDC Link: Testing for COVID-19 Full Link NEJM 2020 (Ezekial J Emmanuel et al): Fair Allocation of Scarce Medical Resources in the Time of Covid-19. This post also displays some data on sensitivity/specificity of testing
- Financial Times: Coronavirus tracked: the latest figures as the pandemic spreads | Free to read & from Johns Hopkins: COVID19 Caseload & Outcomes Worldwide
- COVID-19: Veneto vs. Lombardy and Georgia’s Part of this Pandemic Link: Harvard Business Review: Lessons from Italy’s Response to Coronavirus & Georgia DPH: COVID-19 Daily Status Report
- Ethical Dilemmas and Digestive Symptoms –Common with COVID-19 Full link: NEJM: Facing Covid-19 in Italy — Ethics, Logistics, and Therapeutics on the Epidemic’s Front Line & digestive symptoms ACG: Full Link: ACG Media Statement
- More Advice on Coronavirus for Pediatric GIs: NASPGHAN and CCFA CCFA Guidance for Pediatric Caregivers and Patients —Updates on COVID-19 and IBD