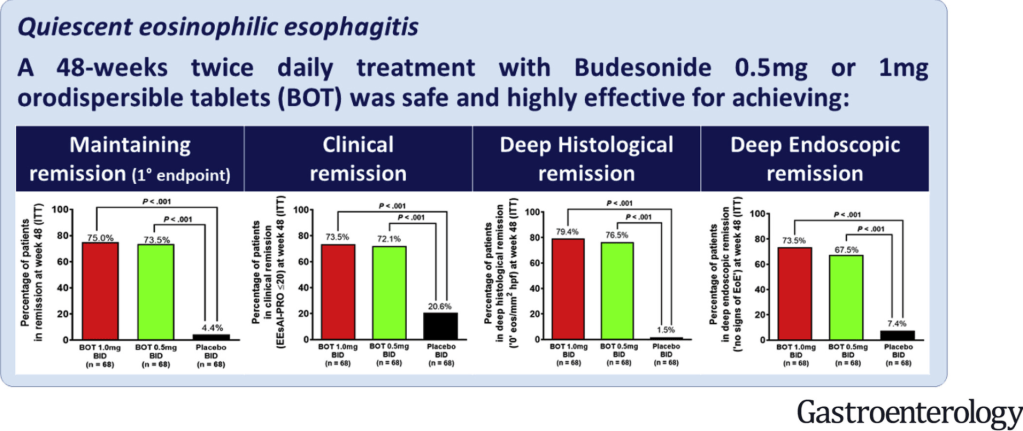
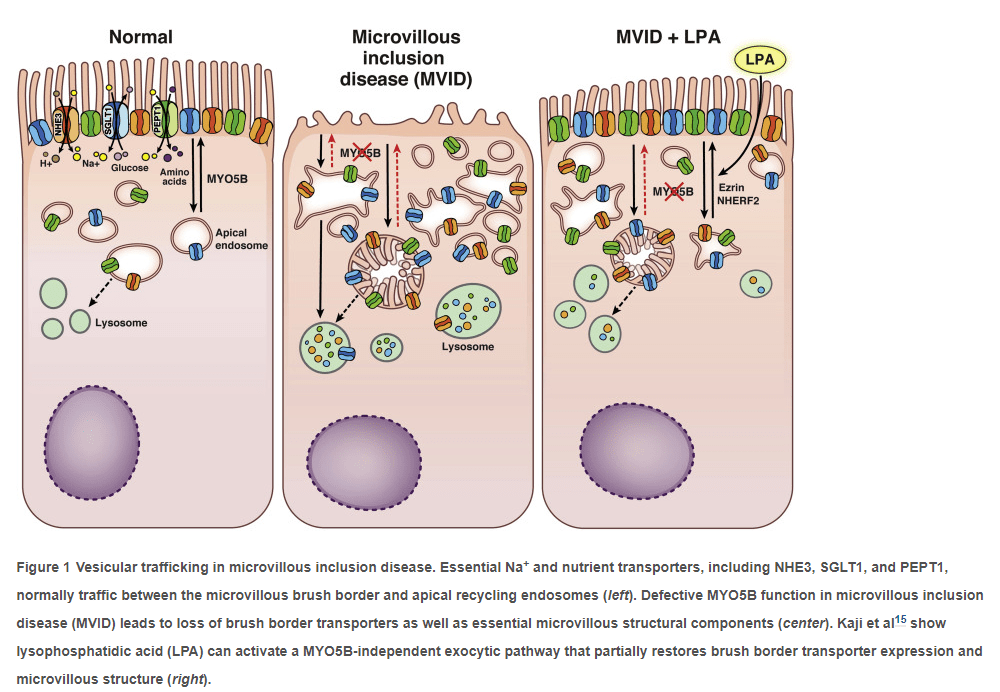
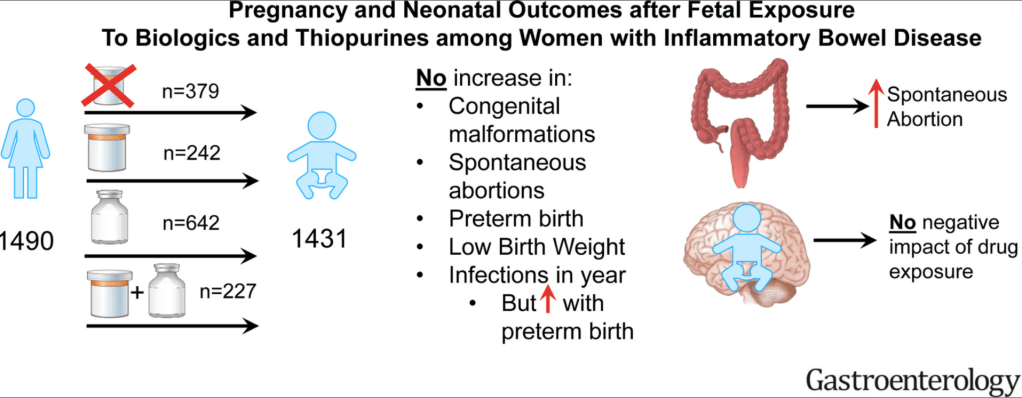
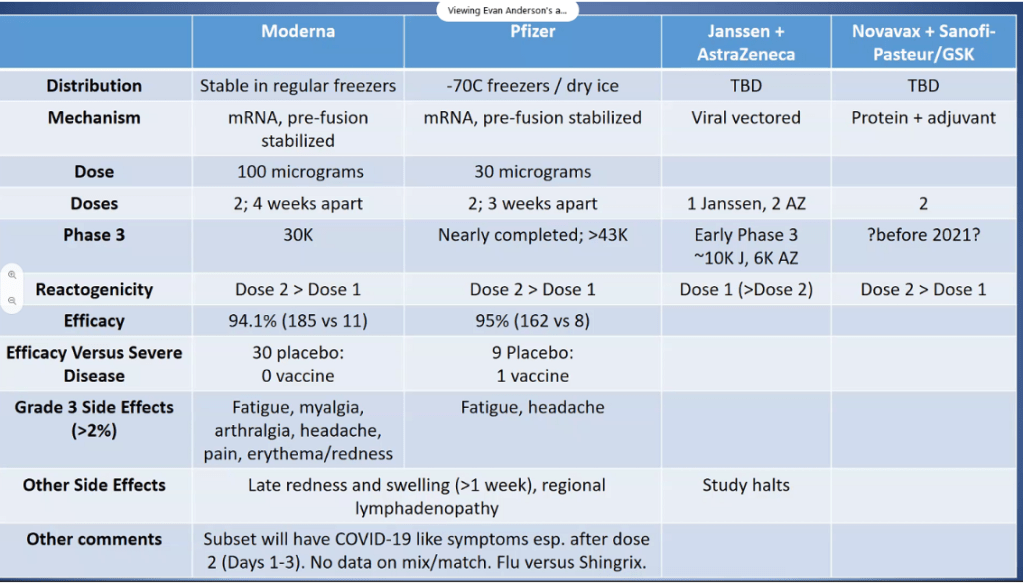
CR Kelly et al. Gastroenterol 2020; doi.org/10.1053/j.gastro.2020.09.038 (in press). Fecal Microbiota Transplantation Is Highly Effective in Real-World Practice: Initial Results From the FMT National Registry
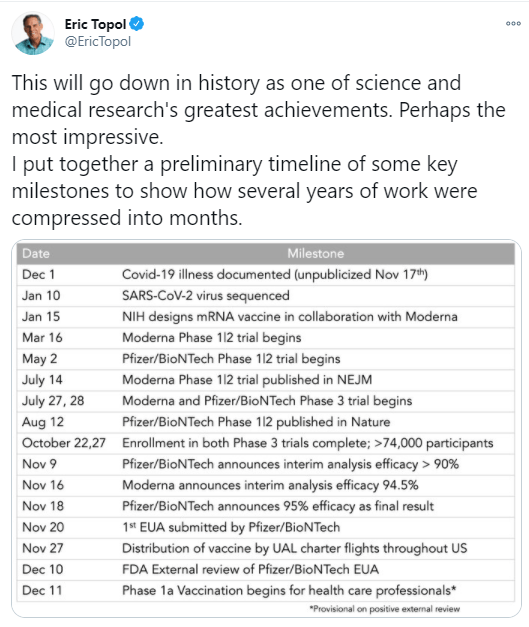
Background: “The FMT National Registry was designed to assess FMT methods and both safety and effectiveness outcomes from North American FMT providers.” n=259 with 222 who completed short-term follow-up.
Key findings:
- All FMTs were done for CDI and 249 (96%) used an unknown donor (eg, stool bank).
- 90% (n=200) were considered cured at one month. Of these, 197 (98%) received only 1 FMT.
- Among 112 patients with initial cure who were followed to 6 months, 4 (4%) had CDI recurrence.
- Safety: Severe symptoms reported within 1-month of FMT included diarrhea (n = 5 [2%]) and abdominal pain (n = 4 [2%]); 3 patients (1%) had hospitalizations possibly related to FMT. At 6 months, new diagnoses of irritable bowel syndrome were made in 2 patients (1%) and inflammatory bowel disease in 2 patients (1%). Milder adverse events were noted in 45% with symptoms including diarrhea, abdominal pain, bloating or constipation.
My take: Overall, the findings from this prospective registry confirm that FMT works fairly well for CDI. Long-term follow-up will provide more answers on the safety of FMT.
Related blog posts:
- Large Study Show FMT Efficacy/Safety in Children (n=335)
- Two Studies: 1. COVID-19 Transmissibility 2.Fecal Microbiota Transplantation in 372 Children
- FMT in the “Real World”
- Clostridium difficile/Fecal Microbiota Transplantation Video
- Consensus Guidelines on FMT | gutsandgrowth
- Something New with FMT
- Fecal Transplantation: “Frozen is as Good as Fresh” | gutsandgrowth
- Rejected! Most Stool is Not Good Enough for FMT | gutsandgrowth