Hepatology 83(4):p 970-973, April 2026. Open Access! Visual Brief Summary: Updated AASLD Guidelines for Chronic Hepatitis B Treatment






Related blog posts:

Hepatology 83(4):p 970-973, April 2026. Open Access! Visual Brief Summary: Updated AASLD Guidelines for Chronic Hepatitis B Treatment






Related blog posts:
Hoskins BJ, Ng K, RexDK. J Pediatr GastroenterolNutr. 2025;81:162‐166. Open Access! Is it time to revisit the need for pediatric polypectomy guidelines?
This commentary/review makes a number of useful points:

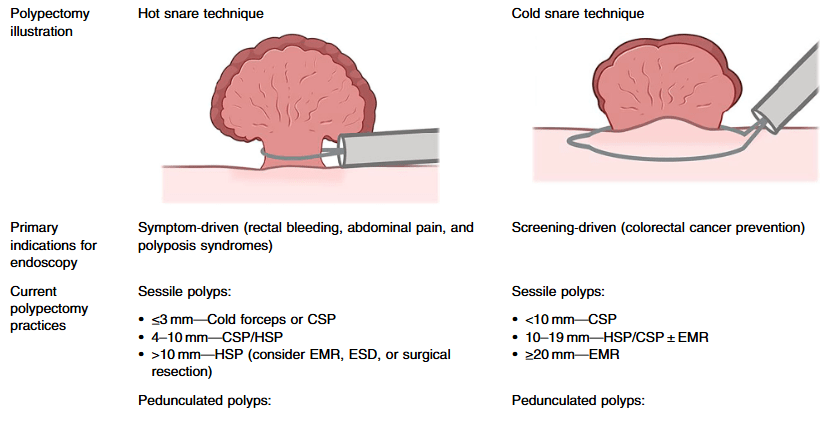
My take (borrowed from authors): “Polypectomy is central to pediatric endoscopy, yet evidence-based, pediatric-specific guidelines are lacking.”
Related article: Hoskins BJ, Grabau JM, Rex DK. J Pediatr Gastroenterol Nutr. 2025;81:1311-131. Pediatric endoscopic mucosal resection: a 10-year single-center experience. . https://doi.org/10.1002/jpn3.70194
Related blog posts:
Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition
We had a terrific lecture given to our group by Dr. Bonney Reed. She is a pediatric psychologist with a clinical and research focus on children with inflammatory bowel disease. Our group has worked closely with Dr. Reed for many years. Many of her slides are included below along with my notes; my notes may contain errors in transcription and in omission.

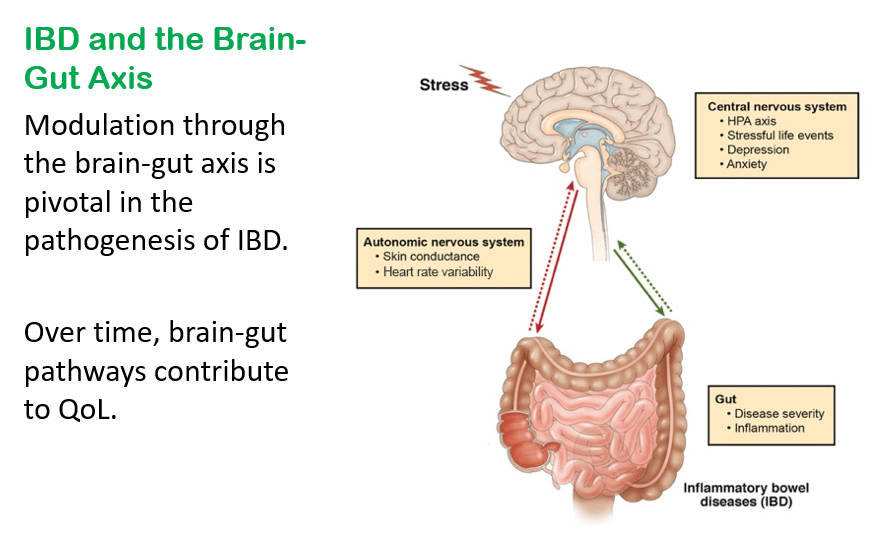
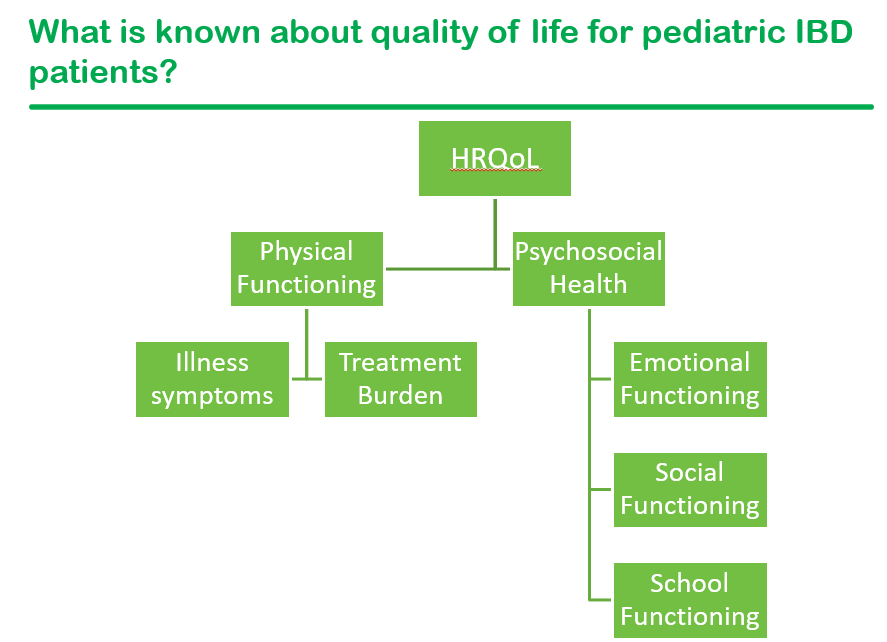

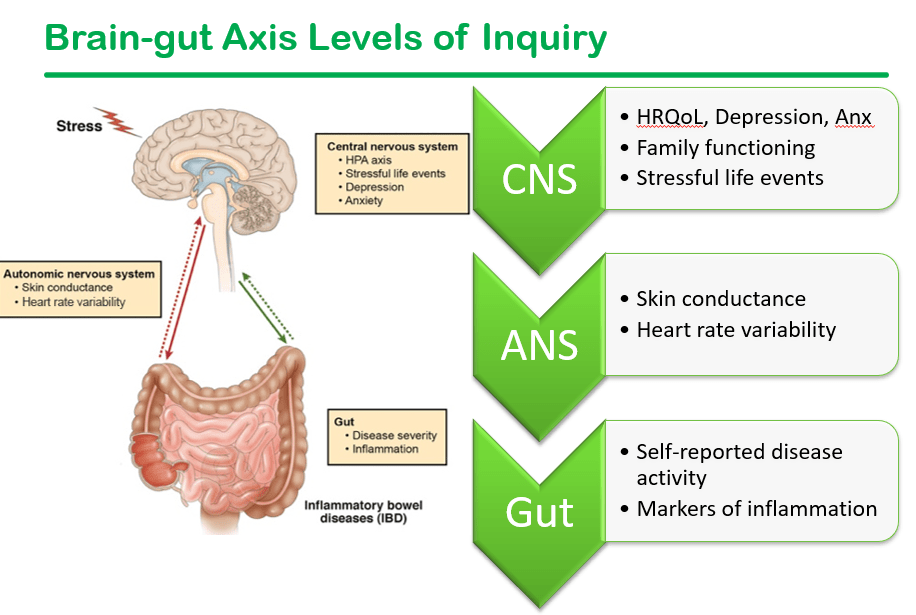


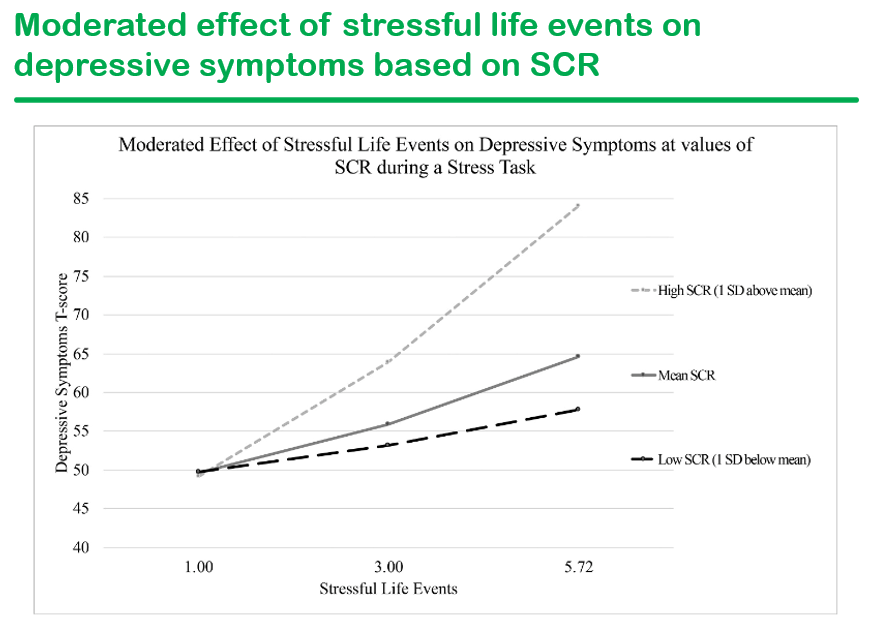
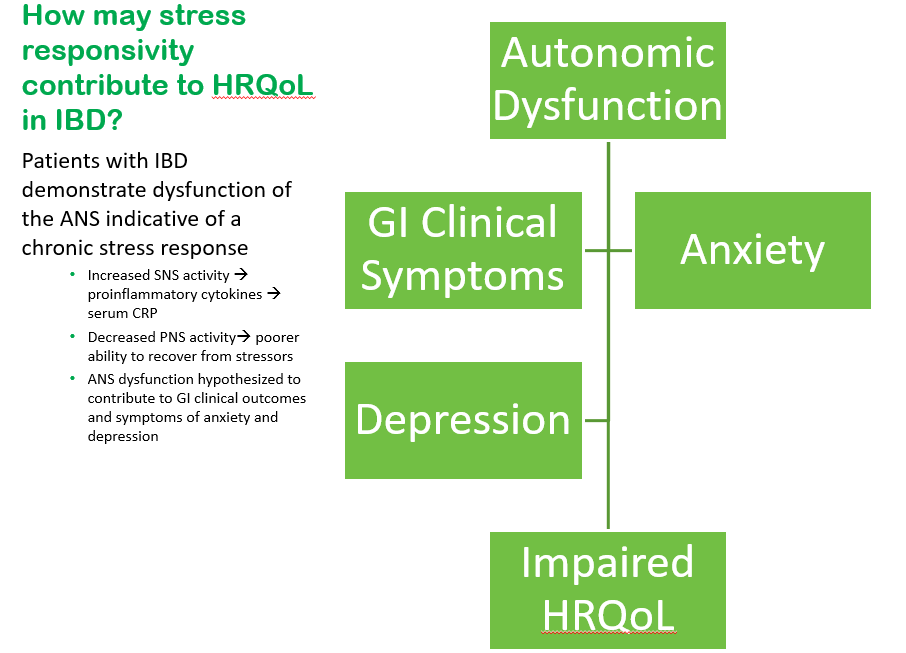
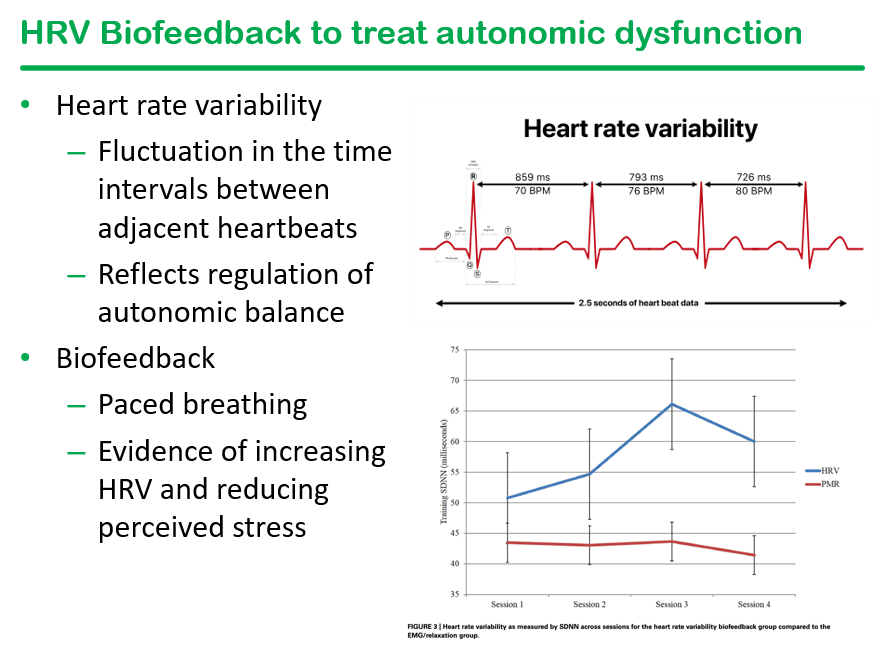

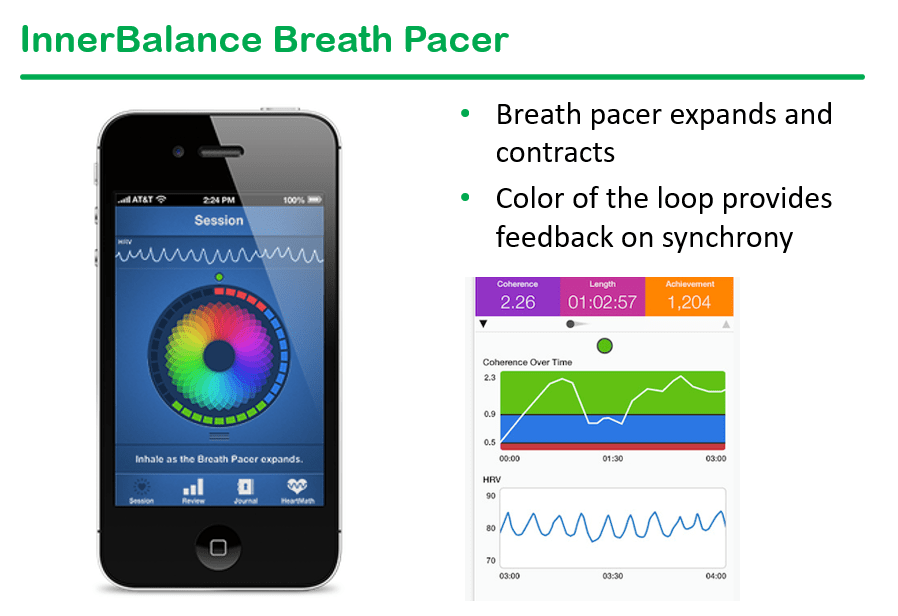
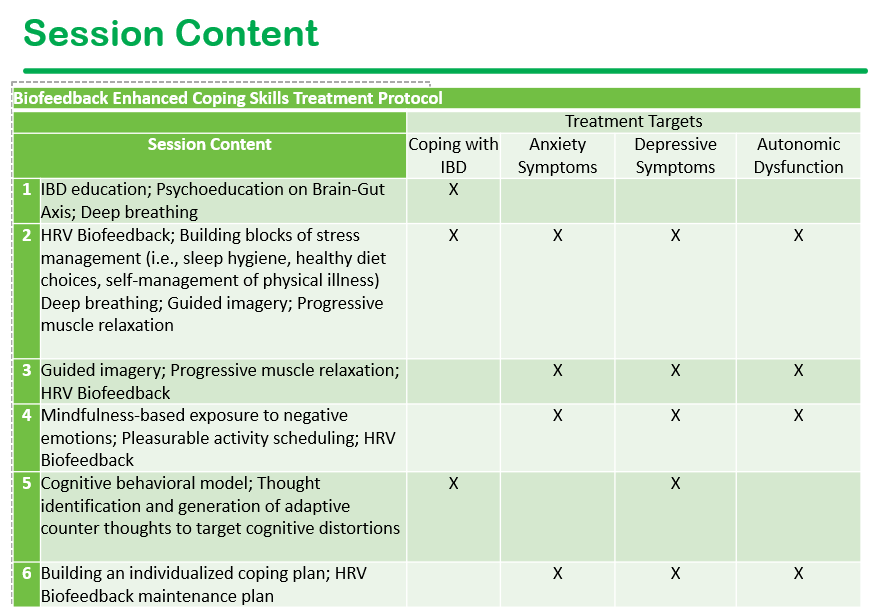

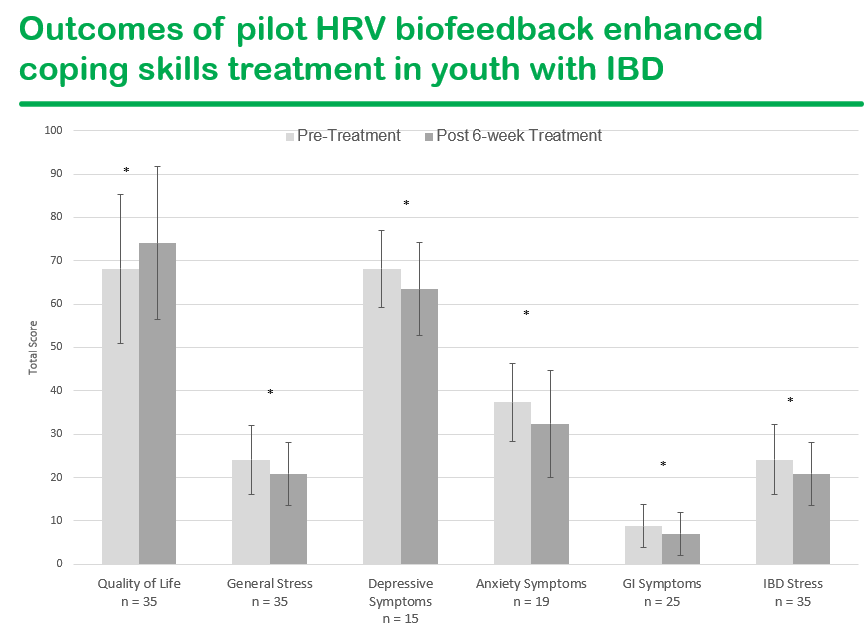

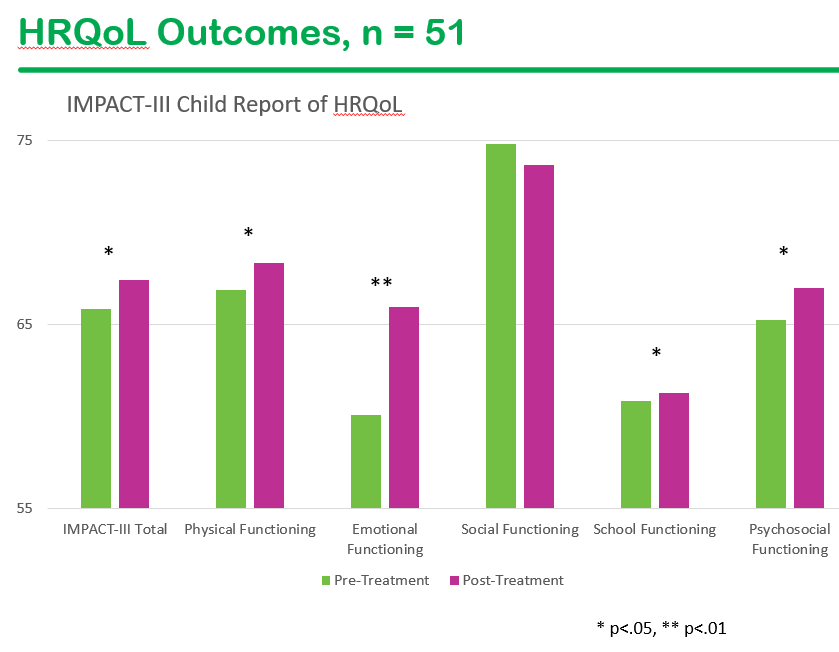
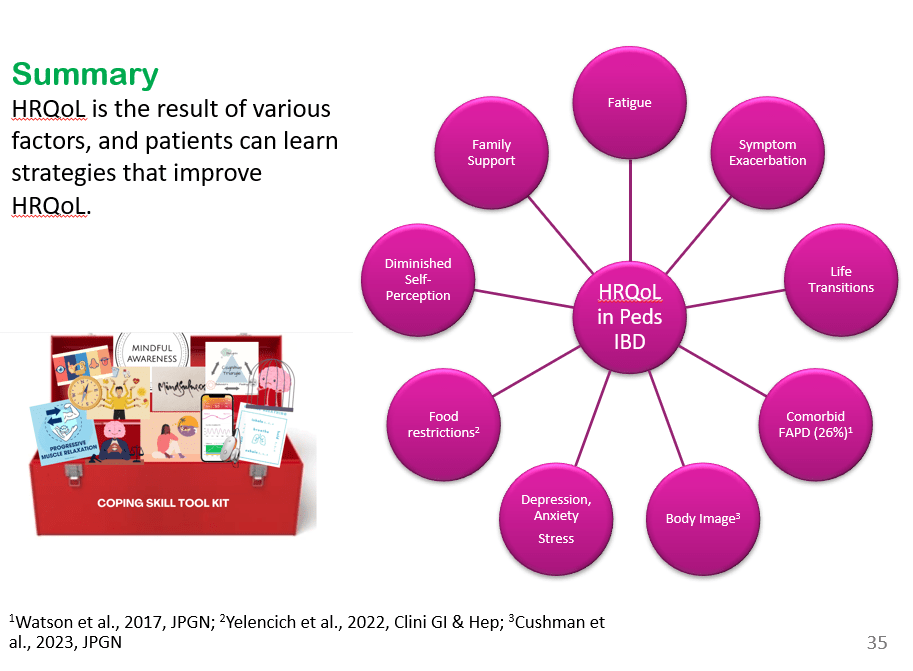
Related blog posts:
JW Jacobs et al. Transfusion 2026;1–9. Open Access! Directed donations for unvaccinated blood: A departure from evidence-based medicine associated with clinical harm, resource waste, and oversight gaps in a two-year single-center series
Background: “Vaccination status cannot be verified through standard blood supply channels, patients seeking “unvaccinated” blood have increasingly resorted to directed donation from known donors…directed donations demonstrate higher rates of infectious disease marker reactivity compared with repeat community donors, particularly among first-time parental donors.25, 26“
Methods: This was retrospective review of directed donations (n=15 including 9 pediatric patients) received at Vanderbilt University Medical Center between January1, 2024 and December 31, 2025
Key findings:
Conclusion by authors: “Directed donation pursued for “unvaccinated” blood concerns occurred across pediatric and adult settings in both elective and urgent clinical scenarios. These requests were associated with clinical deterioration, care delays, and resource inefficiencies when standard inventory products were refused.”
My take: Many families do not understand that directed donation is less safe than using the blood bank.
Fox News report on this article by M Rudy (4/13/26): More patients demand ‘unvaccinated’ blood, doctors warn of growing health risks

JMJ van der Zande et al. J Pediatr Gastroenterol Nutr. 2026;82:980–987. Interrater reliability in pediatric high-resolution anorectal manometry recordings
This study analyzed the interpretation of 10 pediatric high-resolution anorectal manometry (HR-ARM) by 10 pediatric experts in neurogastroenterology, including one of my partners, Dr. Jose Garza.
Key findings:
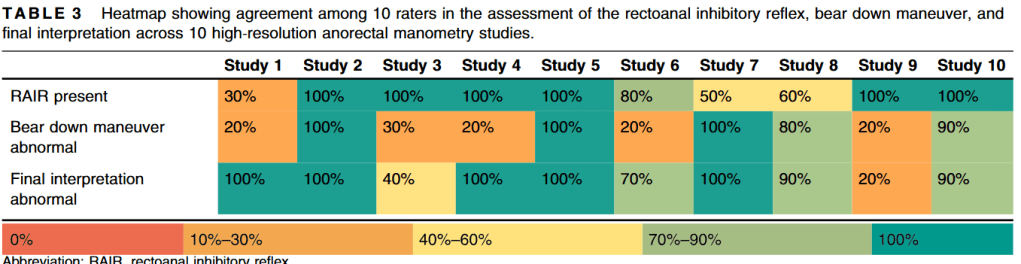
My take: This is a cautionary study indicating a high-rate of variability in the interpretation of key components of HR-ARM; the results are similar to a 2012 study of colonic manometry (see below). Thus, clinical context and supporting data (eg. histology) are quite important as well for clinical decision-making.
Related blog posts:
SO Larsen et al. J Pediatr Gastroenterol Nutr. 2026;82:425–433. Efficacy of low volume transanal irrigation in children with retentive fecal incontinence: A randomized controlled trial
Methods: Two-arm randomized controlled trial, including children (N=41) aged 4–14 suffering from retentive fecal incontinence. All included children were refractory to at least 2 months treatment with stool softening oral laxatives. Treatment duration was 6 weeks. The control group continued oral laxative therapy. The intervention group received low-volume transanal irrigation (L-TAI) as add-on.
Irrigations: “Irrigations were performed once daily with the Qufora IrriSedo MiniGo® Small system. According to the manufacturer the MiniGo® water pump contains 180 mL of water, with an estimated 20 mL residual water per use. Families were instructed to use 1 pump full of water to irrigate. If this did not result in defecation, they were instructed to refill and irrigate again.”
Key Findings:
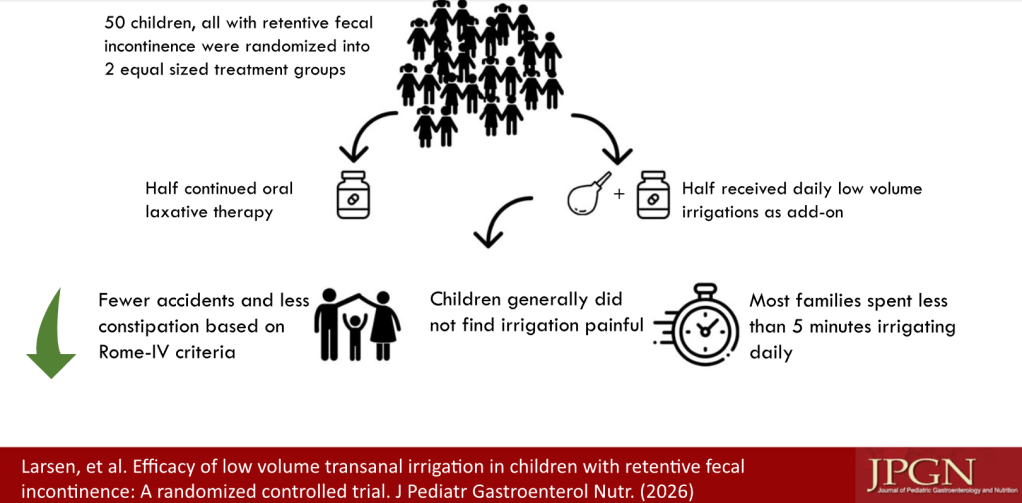
One limitation of this study, besides its sample size, is the type of laxative used prior to enrollment. Patients had been treated with PEG, magnesia or lactulose. These agents are not recommended for refractory constipation; high dose sennosides or bisacodyl have been recommended per recent position paper.
My take: Willie Sutton was a notorious bank robber who was famous for his quote (which he later denied) that he robbed banks “because that’s where the money is.” Similarly, for constipation, targeting treatment at the site of the “deposit” turns out to be an effective strategy. (I have no financial ties to the irrigation company.)
Related article: L Rodriguez et al. Clin Gastroenterol Hepatol 2026 (ahead of print). Open Access! American Gastroenterological Association-North American Society for Pediatric Gastroenterology, Hepatology and Nutrition Pediatric Functional Constipation Clinical Care Pathway
Related blog posts:
There are several insightful and concise articles that highlight the reasons for increased U.S. healthcare costs as well as challenges: corporitization of health care, private equity, and health-harming corporations.
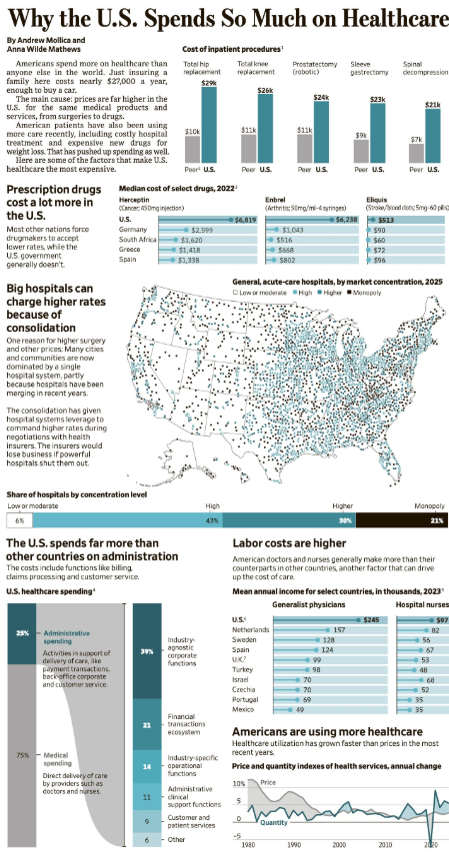
A Mollica, AW Mathews. WSJ 4/6/26: Why the U.S. Spends So Much on Healthcare
An excerpt:
“Americans spend more on healthcare than anyone else in the world. Just insuring a family here costs nearly $27,000 a year, enough to buy a car. The main cause: Prices are far higher in the U.S. for the same medical products and services, from surgeries to drugs.
American patients have also been using more care recently, including costly hospital treatment and expensive new drugs for weight loss.”
LP Casalino. N Engl J Med 2026;394:1249-1251. Physicians, Corporatization, and the Unmeasured Quality of Care
This article notes that historically, in economic experiments, physicians have acted more altruistically than members of the general population and this results in better outcomes for patients. However, “extreme size and corporate ownership are leading to the widgetization of care. It is difficult or impossible for a large organization, even one with well-intentioned leaders, to avoid treating its physicians and staff like interchangeable widgets whose behavior can be monitored and controlled to maximize profit….Physicians who feel like widgets are more likely to behave like widgets…there is evidence that corporatization is leading to higher prices, higher health care spending, and unchanged quality or poorer quality.”
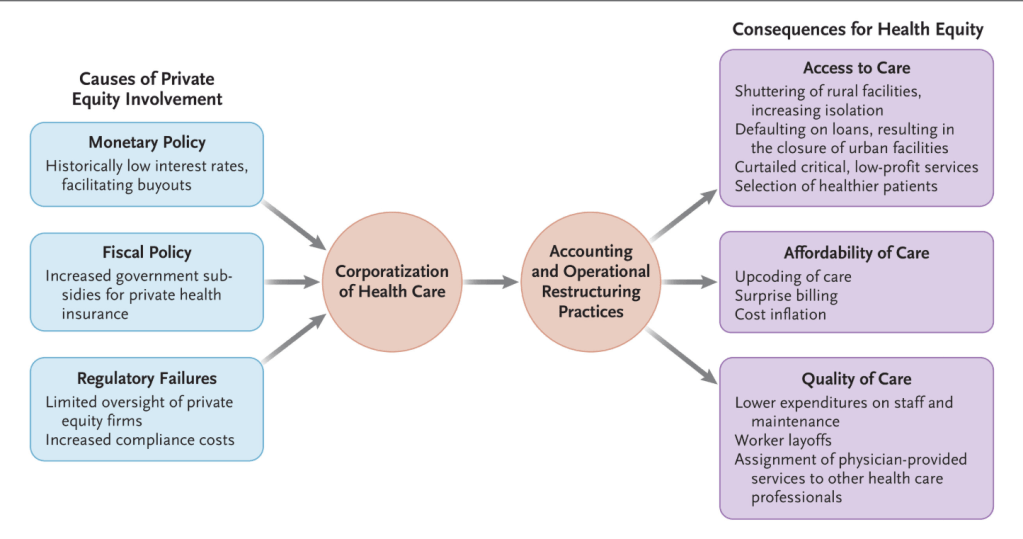
R Yearby, M Alson. N Engl J Med 2026;394:937-940. Private Equity’s Transformation of American Medicine — Implications for Health Equity
An excerpt:
“Accumulating evidence presented in scholarly articles and government reports indicates that the proliferation of PE in health care has reduced access to care, increased costs, and compromised quality of care…PE firms often extract value using tactics that obscure a health care system’s profitability while maximizing financial returns for the firm and its investors. These tactics include sale–leaseback transactions, in which facilities are sold to entities affiliated with a firm and then leased back to the seller at inflated rates. Another strategy is dividend recapitalization, whereby fund managers take on additional debt to pay partners instead of putting money toward staff, critical maintenance, or supplies…
PE investors achieve cost savings by laying off workers, reducing salaries and the number of full-time employees, assigning services previously provided by physicians to other health care professionals, and cutting critical but low-profit services…
Cream skimming — selectively caring for healthier (i.e., lower-cost) patients — is another widely used PE practice. This tactic limits access to care for older and sicker patients, leaving them worse off after PE investment.2 Despite this behavior, hospital acquisitions by PE firms have been associated with increases in emergency department deaths and deaths after emergency surgeries.3,5“
Consortium of the Center to End Corporate Harm, University of California, San Francisco. N Engl J Med 2026;394:1231-1237. Corporate Vectors of Chronic Disease — Using Internal Industry Documents to Craft Counterstrategies
An excerpt:
“Health-harming corporations use common tactics to corrupt scientific data, including influencing research questions, attacking and discrediting independent science and scientists who do not support the industry’s position, suppressing scientific data on the health harms of their products, and sponsoring research that downplays those harms.27,28
For example, the primary U.S. manufacturers of perfluoroalkyl and polyfluoroalkyl substances (PFAS) — DuPont and 3M — used multiple tactics to downplay evidence of PFAS toxicity, including successfully suppressing for more than 20 years internal studies showing adverse effects of PFAS…
Corporations have various tactics for influencing the public’s beliefs about their products’ benefits and harms. These include sophisticated and pervasive advertising and marketing campaigns; use of public relations companies, front groups, and think tanks; and capture of consumer groups.
For example, opioid manufacturers deployed particularly insidious advertising strategies for marketing opioids to vulnerable populations, such as recruiting youth coaches and school nurses to encourage opioid use by children, developing unbranded initiatives encouraging adolescents to ask clinicians for pain medications, promoting “safe opioids” for untreated pain in women, and distorting policy discussions of unmet needs for pain medication…
Make America Healthy Again initiative highlights the roles of toxic chemicals and pesticides, ultraprocessed foods, and corporate influence on science in harming children’s health.47 But…the administration has appointed former lobbyists and scientists from the chemical and petroleum industries to lead EPA offices responsible for regulating air pollution, toxic chemicals, and pesticides48,49 — and plans to eliminate regulatory and other measures, which will lead to increased exposure to toxic chemicals and air pollutants, thereby increasing child health risks.50,51
My take: Poorly-regulated capitalism is not good for patients. Insurers, private equity, hospitals, pharmaceutical companies and many providers may prioritize profits over care.
Related blog posts:
Z Younossi et al. Liver Transplantation 2026; 32(4):p 549-557. Steatotic liver disease is the dominant indication for liver transplantation in both Europe and the United States: Trends and outcomes in the past 2 decades
Methods: Adult data from 2 large LT registries: the US Scientific Registry of Transplant Recipients (SRTR) and the European Liver Transplant Registry (ELTR), years 2000–2022, were compared. There were 109,048 recipients of transplant from ELTR (30 countries), and 128,765 from SRTR.
Key findings:
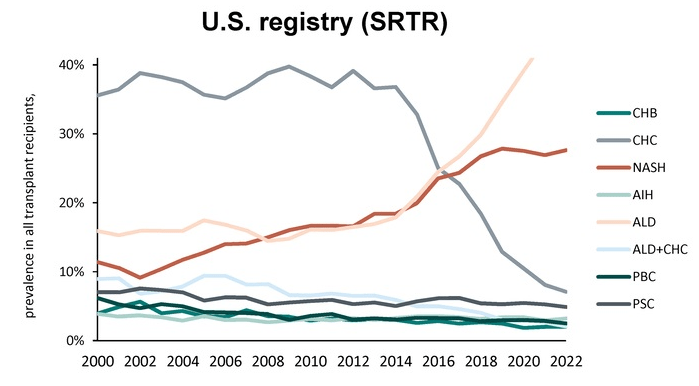
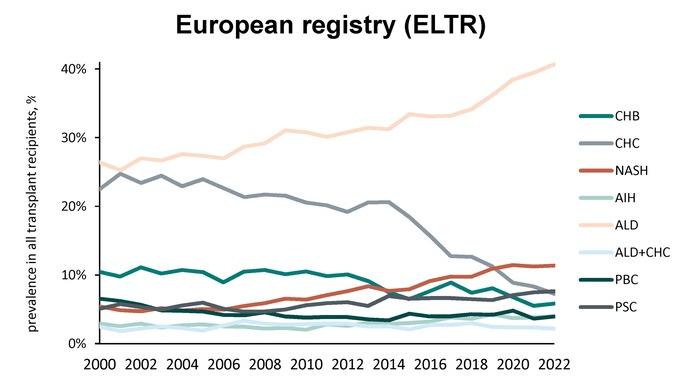
Related blog posts:
C Thellin, Y Urrutia. Gastroenterology and Endoscopy News, March 17, 2026. Open Acess! ‘Uncorking’ the Esophageal Food Impaction: Why Twisting Works Better Than Pulling
An excerpt from this recent article:
Yet, just as one wouldn’t simply yank a cork from a bottle, it may be time to rethink how we approach food bolus removal. Torsional stress—twisting rather than pulling—may provide a more efficient and atraumatic method for managing these impactions, a technique we refer to as the “corkscrew method.”
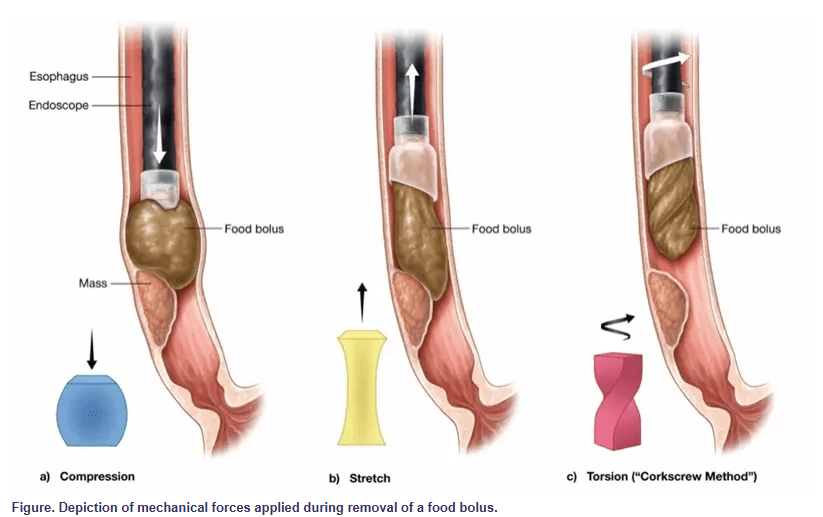
Less force is required because rotation gradually disengages the bolus, decreasing the need for strong pulling or pushing. This method also poses a lower risk for mucosal injury. Shear forces are distributed rather than focused, reducing trauma.
My take: This is probably worth a try for the next food impaction requiring endoscopic removal.
Related blog posts:
Disclaimer: This blog, gutsandgrowth, assumes no responsibility for any use or operation of any method, product, instruction, concept or idea contained in the material herein or for any injury or damage to persons or property (whether products liability, negligence or otherwise) resulting from such use or operation. These blog posts are for educational purposes only. Specific dosing of medications (along with potential adverse effects) should be confirmed by prescribing physician. Because of rapid advances in the medical sciences, the gutsandgrowth blog cautions that independent verification should be made of diagnosis and drug dosages. The reader is solely responsible for the conduct of any suggested test or procedure. This content is not a substitute for medical advice, diagnosis or treatment provided by a qualified healthcare provider. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a condition.
MV Vestergaard et al. Gastroenterol 2026; 170: 721-734. Open Access! Genetic Risk of Inflammatory Bowel Disease Is Associated With Disease Course Severity
Methods: The authors calculated IBD polygenic scores (PGS) for 3732 Danish patients with Crohn’s disease (CD) and 4535 patients with ulcerative colitis (UC), and investigated their association with disease outcomes.
“The Danish National Biobank stores neonatal blood spots from almost all Danes born since 1982…The North Denmark IBD (NorDIBD) cohort is a population-based cohort of all patients from the North Denmark Region with a confirmed IBD diagnosis from 1978–2020.17 From this cohort, 940 patients with IBD and a further 973 blood donors with no IBD diagnosis were genotyped from whole blood…applied variant loadings for calculating PGS for CD and UC susceptibility generated by Middha et al19 to our dataset. These variant loadings were derived from summary statistics from the IIBDGC.” This cohort had a young age. Only 2% and 3% of CD and UC patients, respectively, were 40+.
Severe Disease Definitions:
Key findings:
The graphs below show the differences in hospitalization and surgery based on PGS for CD (A & B) and then for UC (C & D)
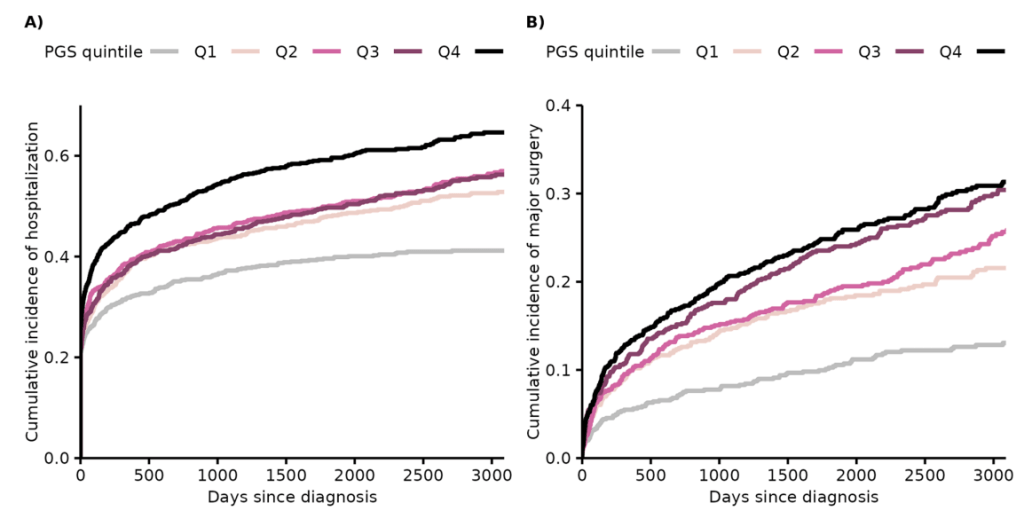
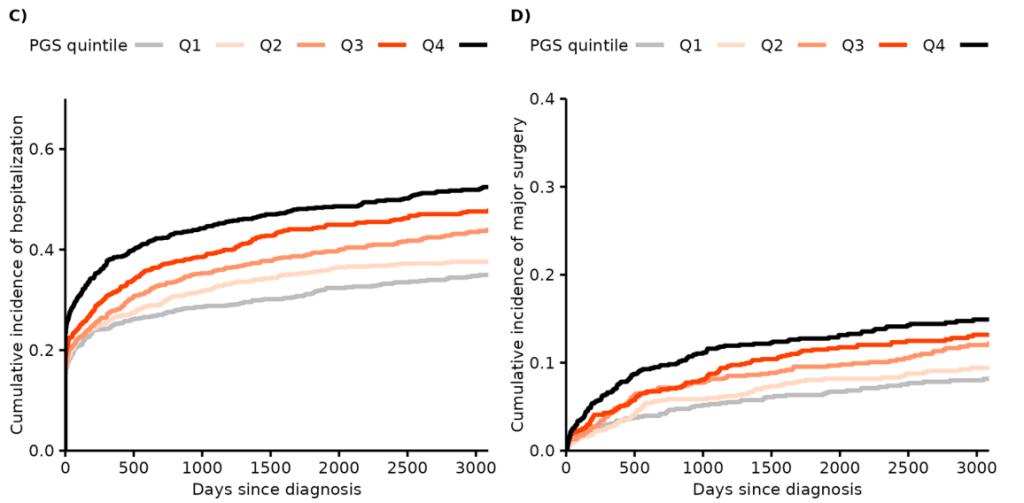
Discussion:
My take: It is intuitive that those with more genetic risk factors would be more likely to have severe disease. This study shows this assumption is correct in this cohort; in fact, there is a dose-response relationship. Those with higher PGS had more hospitalizations, major surgeries and medical treatments.
Related blog posts: