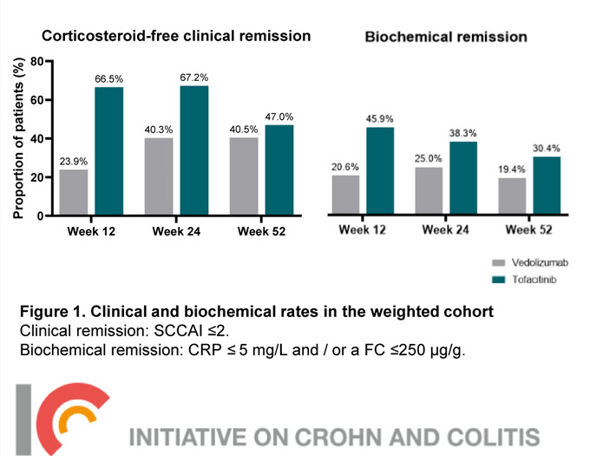
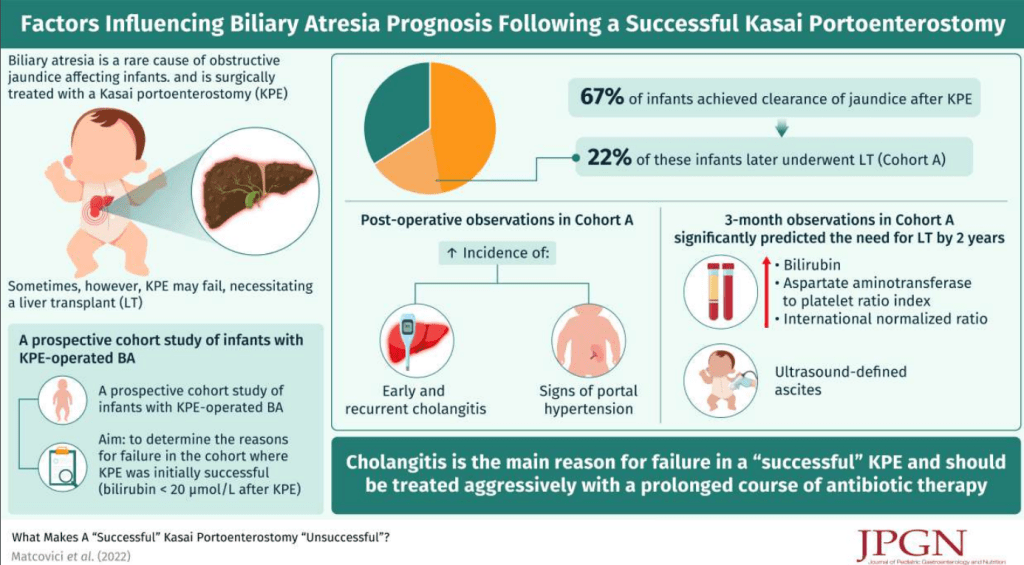
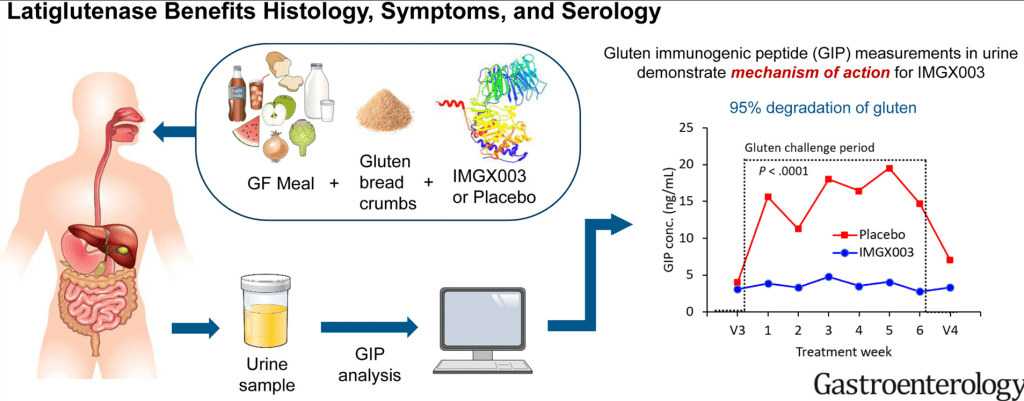
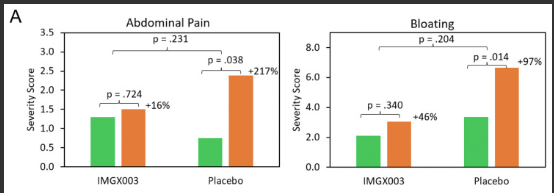
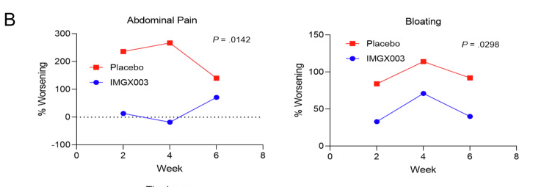
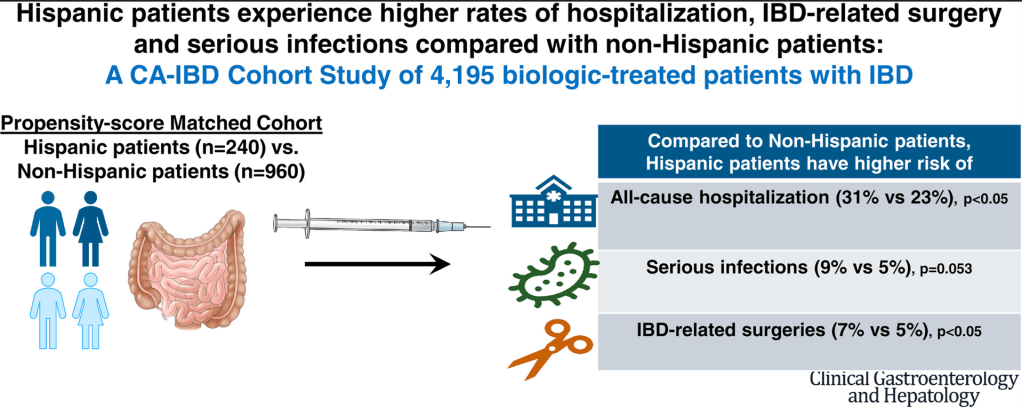
R Kuang et al. Inflamm Bowel Dis 2023; 29: 140-150. Is Salt at Fault? Dietary Salt Consumption and Inflammatory Bowel Disease
This review looks at the potential role of salt in relation to the epidemiology of inflammatory bowel disease. The general focus is that the prevalence/incidence of IBD has been increasing and there must be environmental/dietary factors involved. Could salt be one of those causal factors or is it merely a temporal association?
Key points:
- Ultra-processed foods make up more than half of the daily caloric intake in developed countries such as the United States! and Canada and between one-third to one-fifth of diets in middle-income countries such as Brazil and Mexico.. Ultra-processed foods involve “fractioning of whole foods into substances, chemical modifications of these substances, frequent use of cosmetic additives and sophisticated packaging that allow producers to create highly profitable, convenient, and hyperpalatable products.” Ultra-processed foods are typically high in sugar, unhealthy fats, and salt and low in dietary fiber, protein, vitamins, and minerals. They are also calorie dense. For Americans, the primary source of sodium in the diet is from commercially processed foods.
- At present, the typical American consumes over 40% more salt on a daily basis than is re-commended. Added salt is a key component of UPFs, whose increased consumption has been closely linked to this rise in the IBD incidence. Even though salt is a key component of UPFs, it has received limited attention in the investigation of IBD...Excess salt contributes to greater monocyte and T-cell-driven inflammation and a parallel loss of immunoregulatory mechanisms involving M2 macrophages and Tregs in the Th17 axis.
- The authors argue that improvement in IBD with exclusive enteral nutrition is another factor indicating a potential role for salt reduction as beneficial. “Although these ultra-processed liquid nutrition formulas were high in sugars, emulsifiers, and carrageenan, they were very low in sodium content.”
My take: It is not clear what impact salt has on IBD. However, too much salt causes problems well beyond hypertension and may contribute to several inflammatory conditions, including IBD, asthma, and rheumatoid arthritis.
Related blog posts:
- IBD Updates: Dietary Patterns and Disease Activity, Ustekinumab in SUSTAIN study, INSPECT Study for Perianal Fistulas
- Dietary Therapy for Inflammatory Bowel Disease –Useful Update
- NASPGHAN 2021 Nutrition Highlights
- Can IBD Be Treated with Diet Alone?
- What is An Emulsifier and Are They Safe in Our Diets?
- Dietary Therapy for Adults with Crohn’s Disease
- CDED + PEN: An Alternative Diet to Exclusive Enteral Nutrition?
- The Search for a Dietary Culprit in IBD
Unrelated website information: IBD-EII is a website which has tried to organize/summarize some of the more important IBD articles including a timeline of these publications and evidence for specific medications.

Atlanta Botanical Gardens. Garden Nights, Holiday Lights exhibit