Notice: At this time, gutsandgrowth intends to post blogs 2-3 times per week rather than daily.
——————————————————————————————————————
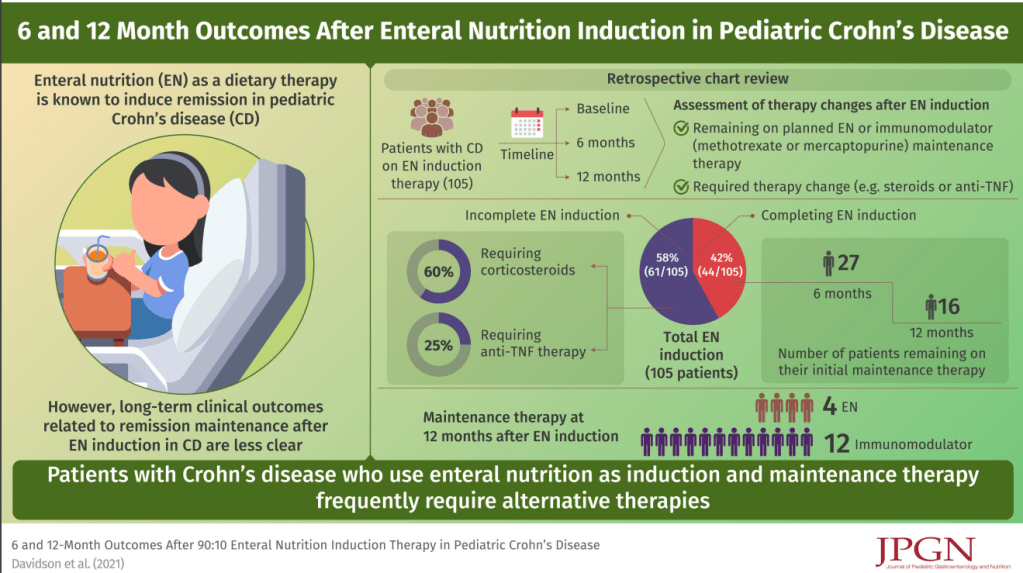
N Davidson et al. JPGN 2022; 75: 70-75. 6- and 12-Month Outcomes after 90:10 Enteral Nutrition Induction Therapy in Pediatric Crohn’s Disease
In this retrospective study (2013-2018), the authors examined outcomes in 105 children treated with a 90:10 enteral feeds (90% formula).
Key findings:
- 44/105 (42%) patients completed 8–12 weeks
- After induction, 18 continued EN maintenance with a 80:20 then 70:30 protocol; however, only 10 remained on EN at 6 months and 4 remained on EN at 12 months
The associated editorial (pg: 1-2) make several points:
- While EEN is effective and safe, this study and others have shown poor adherence
- It is unclear how exclusive enteral nutrition needs to be in order to be effective. And, many patients instructed to receive 90% of their calories as formula are likely consuming higher amounts of table foods
- We still are working out which foods need to be excluded
My take: This study shows that EEN is NOT a practical option for most patients beyond induction. Only 4 patients remained on EEN at 12 months.
Related blog posts:
- Mediterranean Diet vs Specific Carbohydrate Diet for Crohn’s Disease
- CDED + PEN: An Alternative Diet to Exclusive Enteral Nutrition?
- Dietary Therapy for Inflammatory Bowel Disease This is a good lecture review on dietary therapy
- Trial by Diet for Pediatric Crohn’s Disease
- Can IBD Be Treated with Diet Alone?
- Good Food and Bad Food for Crohn’s Disease -No Agreement | gutsandgrowth
- Pushing the Boundaries on Dietary Therapy for Crohn’s Disease: CD-TREAT
- Position Paper: Nutrition in Pediatric IBD
- CDED Diet for IBD/IBD Updates
- Specific Carbohydrate Diet | gutsandgrowth