Briefly noted –all of these articles are open access:
A Volkers et al. AP&T 2022; https://doi.org/10.1111/apt.17153 Open access: Real-world experience of switching from intravenous to subcutaneous vedolizumab maintenance treatment for inflammatory bowel disease. In this prospective cohort study, patients (n=135) with IBD who had ≥4 months IV vedolizumab were switched to SC vedolizumab.
Key findings:
- 4 patients with Crohn’s disease had loss of response.
- 9% of patients were switched back to IV vedolizumab due to adverse events or fear of needles.
- Median clinical and biochemical disease activity remained stable after the switch. Median vedolizumab serum concentrations increased from 19 μg/ml at the time of the switch to 31 μg/ml 12 weeks after the switch (p < 0.005).
Related blog posts:
- Vedolizumab vs Adalimumab: Histology Outcomes from Varsity Trial
- Vedolizumab versus Adalimumab for Ulcerative Colitis (part 2)
- Vedolizumab More Effective Than Adalimumab for Ulcerative Colitis
- Real-World Vedolizumab: Better Than Expected | gutsandgrowth
- Comparative Efficacy: Vedolizumab vs Anti-TNF Agents | gutsandgrowth
- Vedolizumab vs. Adalimumab for Infliximab Failure in Ulcerative Colitis: Which is better?
- Is Vedolizumab the Best First Line Biologic in Ulcerative Colitis? | gutsandgrowth
HC Kaplan et al. Am J Gastroenterol 2022 Jun 1;117(6):902-917. Open access: Personalized Research on Diet in Ulcerative Colitis and Crohn’s Disease: A Series of N-of-1 Diet Trials. In this study, 21 patients (completed trial) were randomized to 1 of 2 sequences of 4 alternating 8-week SCD (specific carbohydrate diet) and MSCD (modified specific carbohydrate diet) periods.
Key findings: “SCD and MSCD did not consistently improve symptoms or inflammation.” “Some individuals had improvement in symptoms and fecal calprotectin compared with their UD, whereas others did not.” The authors note that it took 18 months to recruit 54 patients for this study across 19 research sites.
Related blog posts:
- Mediterranean Diet vs Specific Carbohydrate Diet for Crohn’s Disease
- Trial by Diet Approach for Crohn’s disease in children (this blog post has link to 16 other dietary therapy posts)
EL Barnes et al. Inflamm Bowel Dis 2022; 28: 983-987. Open access: Racial and Ethnic Distribution of Inflammatory Bowel Disease in the United States The authors electronic health records from 337 centers from January 2013 to December 2018 with nearly 40 million patients in U.S.
Key findings:
- Black adult patients were significantly less likely than White patients to have a diagnosis of CD (odds ratio [OR], 0.53) or UC (OR, 0.41). Pediatric Black patients were also less likely to have a diagnosis of CD (OR, 0.41) or UC (OR, 0.38)
- Adult Hispanic patients were less likely to have a diagnosis of CD (OR, 0.33) or UC (OR, 0.45) compared with non-Hispanic patients. Similarly, pediatric Hispanic patients were less likely to have a diagnosis of CD (OR, 0.34) or UC (OR, 0.50).
- Thus, these data suggest that CD and UC are modestly less prevalent among patients of non-White races and Hispanic ethnicity
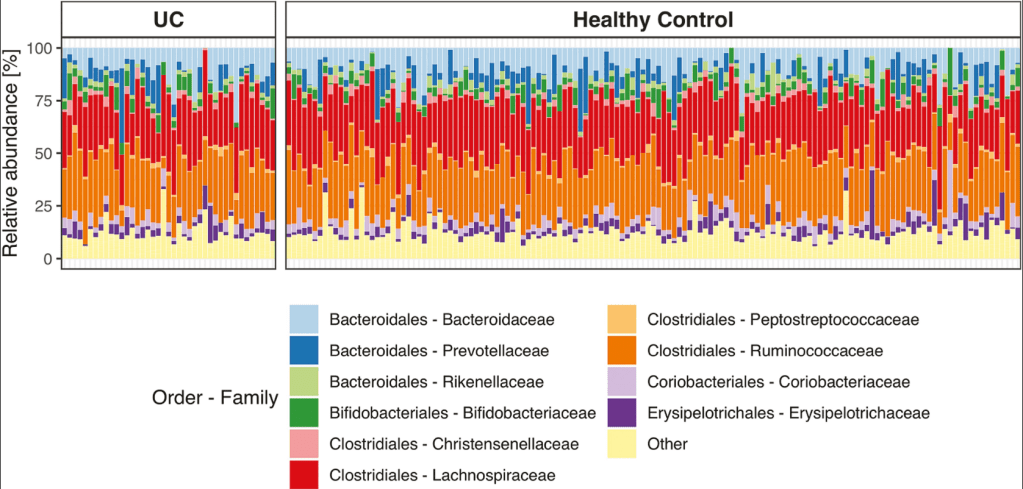
M Frioirksmork et al. Inflamm Bowel Dis 2022; 28: 1081-1089. Open access: Similar Gut Bacterial Composition Between Patients With Ulcerative Colitis and Healthy Controls in a High Incidence Population: A Cross-sectional Study of the Faroe Islands IBD Cohort. This cross-sectional study from the Faroe Islands (which has very high incidence of IBD) consisted of 41 patients with established ulcerative colitis and 144 age- and sex-matched healthy controls.
Key findings: There was a similarity in bacterial community composition and absence of the beneficial Akkermansia genus in both groups.