SA Kahn et al. NEJM 2023; 388: 358-368. Case 3-2023: A 16-Year-Old Girl with Abdominal Pain and Bloody Diarrhea
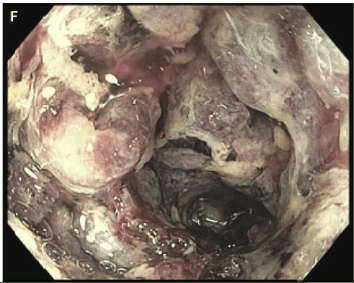
This case report of a girl presenting with abdominal pain and diarrhea identifies a rare etiology, catastrophic antiphospholipid syndrome (CAPS). CAPS can result in ischemic colitis. This patient underwent a colonoscopy which was normal in rectum but then became abnormal in the sigmoid colon:
“CAPS is thought to result from the binding of antiphospholipid antibodies to cell surfaces, which activates endothelial cells, monocytes, and platelets and leads to inflammation, complement activation, and thrombosis. The formation of thrombi in patients with antiphospholipid antibodies is thought to be a multihit process.7 The presence of antiphospholipid antibodies in the blood is the first event, but the antibodies typically do not cause disease until another event occurs.”
Management of antiphospholipid antibodies: “Thrombotic disease manifestations are important in guiding therapy. For patients with antiphospholipid antibodies and no history of clotting, anticoagulation for primary thromboprophylaxis is generally not recommended…For patients with antiphospholipid antibodies and a history of unprovoked thrombosis (e.g., patients with APS), long-term thromboprophylaxis is recommended…For patients with more severe presentations — such as this patient, who had CAPS with thrombosis in multiple organs — treatment is more aggressive and involves targeting multiple steps within the CAPS cascade.”
From ChatGPT

My take: This 16 yo had a severe presentation and the case is a reminder that there are multiple reasons besides IBD for a teenager to have bloody diarrhea and abdominal pain.
Related blog posts: