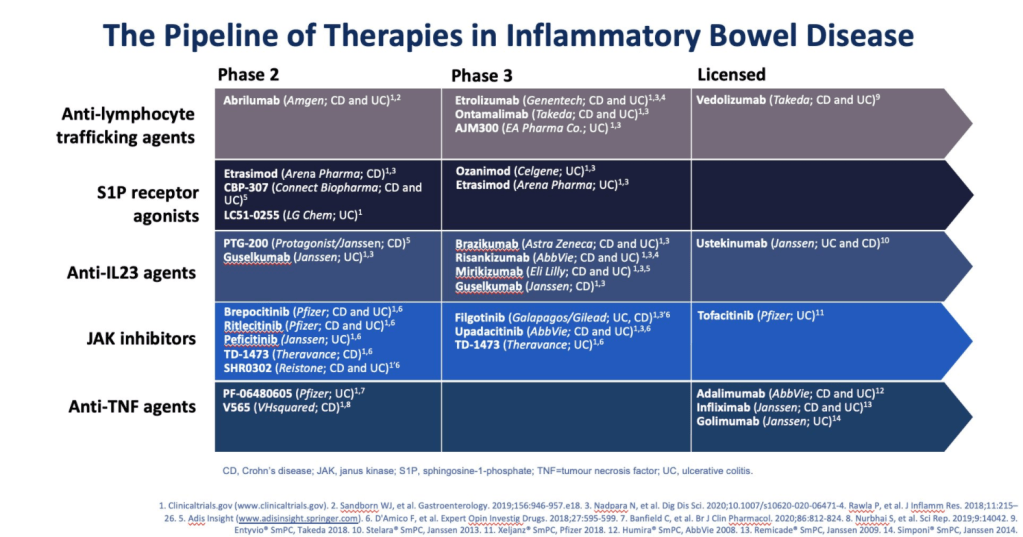
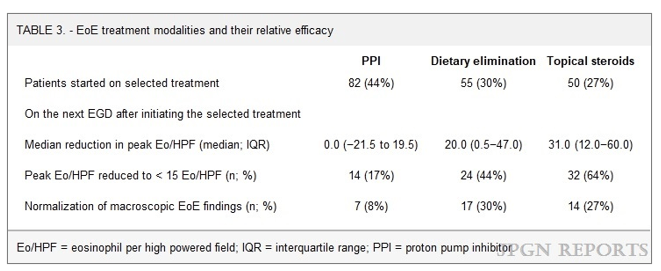
T Gonska et al. Pediatrics 2021; 148: e2021051740. Outcomes of Cystic Fibrosis Screening-Positive Infants With Inconclusive Diagnosis at School Age
Background: : Cystic fibrosis (CF) screen-positive infants with an inconclusive diagnosis (CFSPID) are infants in whom sweat testing and genetic analysis does not resolve a CF diagnosis
Methods: Prospective, longitudinal, multicenter, Canada-wide cohort study of CFSPID for a mean of 7.7 years
Key findings:
- A CF diagnosis was established for 24 of the 115 children with CFSPID (21%) — either because of reinterpretation of the cystic fibrosis transmembrane conductance regulator genotype or because of increase in sweat chloride concentration ≥60 mmol/L. Those with initial sweat chloride concentration ≥40 mmol/L were most likely to receive a diagnosis of cystic fibrosis.
- Children with CFSPID were pancreatic sufficient and showed normal growth until school age and had good pulmonary outcomes (similar to healthy controls)
- In the associated commentary by P Chakraborty et al (Maximizing Benefits and Minimizing Harms: Diagnostic Uncertainty Arising From Newborn Screening), the authors note that while newborn screening (NBS) offers benefit of early diagnosis, some families can be harmed by false-positive tests or inconclusive results. Furthermore, “these issues of uncertainty are increasingly important to consider as the scope of NBS programs and their use of genomic technologies expands.”
My take: With CF, this study shows the need to monitor those with inconclusive studies. More broadly, the use of genomic testing is leading to more frequent inconclusive results in many areas and sometimes leaving more questions than answers.
Related blog posts:
- CHOA Nutrition Support -CF in the age of “Miracle Drug”
- Aspen Webinar 2021 Part 7 -Cystic Fibrosis Liver Disease
- Data on Immobilized Lipase Cartridge for Patients with CF
- Cystic Fibrosis Expert Update 2017
- Nutrition Pearls: Fiber and Cystic Fibrosis
- Big Advance for Cystic Fibrosis -Who Will Benefit?
- Nutrition Guidelines for Cystic Fibrosis
- Complex Family of CFTR-Associated Disorders | gutsandgrowth